Advanced Manufacturing of 4-Cyano-4'-Hydroxybiphenyl: A Breakthrough in Liquid Crystal Intermediate Production
The global demand for high-performance liquid crystal materials continues to drive innovation in the synthesis of key intermediates, specifically 4-cyano-4'-hydroxybiphenyl. A pivotal advancement in this domain is detailed in Chinese Patent CN100460385C, which discloses a robust, three-step synthetic route starting directly from biphenyl. This methodology represents a significant departure from conventional strategies that rely on biphenol derivatives, offering a streamlined pathway that circumvents the complexities of hydroxyl protection and the notorious challenges associated with iodination reactions. For R&D directors and procurement specialists alike, this patent outlines a process that not only enhances product purity by eliminating iodine-containing impurities but also optimizes the economic feasibility of large-scale production through the use of readily available commodity feedstocks.
The strategic value of this technology lies in its ability to deliver a high-purity intermediate essential for next-generation display technologies while simultaneously addressing critical supply chain vulnerabilities. By shifting the synthetic entry point to biphenyl, manufacturers can leverage a more stable and cost-effective raw material base, reducing dependency on specialized phenolic precursors that often suffer from price volatility. This report provides a deep technical analysis of the acylation-bromination-oxidation-substitution sequence, evaluating its mechanistic advantages and its potential to redefine cost structures in the electronic chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 4-cyano-4'-hydroxybiphenyl has relied heavily on routes initiating from 4,4'-biphenol or its mono-protected derivatives. As illustrated in the traditional reaction schemes, these methods invariably require a protection-deprotection strategy to manage the reactivity of the hydroxyl groups, inherently adding at least two extra synthetic steps to the overall process. More critically, many of these established pathways necessitate an iodination step to introduce the functionality required for subsequent cyanation. The presence of iodine in the reaction matrix poses severe quality control risks, as trace iodinated byproducts are notoriously difficult to separate completely from the final API or intermediate, potentially compromising the electro-optical performance of the final liquid crystal mixture.
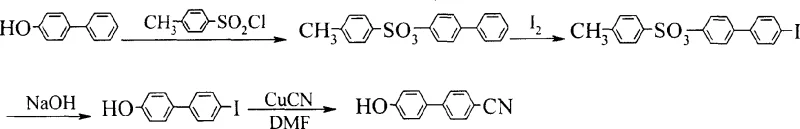
Furthermore, the reliance on protected biphenols introduces significant logistical and cost burdens. The synthesis of these protected precursors often involves hazardous reagents and generates substantial waste streams, complicating environmental compliance and increasing the overall cost of goods sold (COGS). The multi-step nature of these conventional routes also amplifies the risk of yield loss at each stage, resulting in lower overall throughput and extended production cycles that struggle to meet the rapid scaling demands of the modern electronics industry.
The Novel Approach
In stark contrast, the novel approach disclosed in CN100460385C utilizes unsubstituted biphenyl as the starting material, executing a clever tandem acylation-bromination sequence that functionalizes the aromatic ring in a single operational pot. This strategy effectively bypasses the need for hydroxyl protection entirely, as the hydroxyl group is introduced later in the sequence via oxidation of an acetyl intermediate. By avoiding the iodination step altogether, the process inherently guarantees a cleaner impurity profile, free from the persistent halogenated contaminants that plague older methods. This direct functionalization not only shortens the synthetic timeline but also dramatically simplifies the downstream purification requirements.
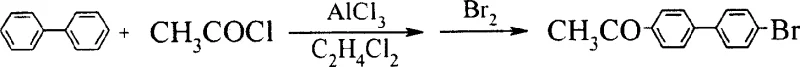
The core innovation of this route is the seamless integration of the acylation and bromination steps. Instead of isolating the acetylated intermediate, the reaction mixture is treated directly with bromine in the presence of a Lewis acid catalyst. This telescoped operation minimizes solvent usage, reduces handling time, and prevents the degradation of sensitive intermediates that can occur during isolation. The subsequent oxidation step converts the acetyl moiety into the requisite hydroxyl group with high efficiency, setting the stage for the final cyanation. This logical progression from a commodity chemical to a high-value intermediate exemplifies process intensification, offering a compelling alternative for manufacturers seeking to optimize their production capabilities.
Mechanistic Insights into the Acylation-Bromination-Oxidation Sequence
The success of this synthetic route hinges on the precise control of electrophilic aromatic substitution and oxidative transformation mechanisms. The initial step involves a classic Friedel-Crafts acylation where biphenyl reacts with acetyl chloride in the presence of anhydrous aluminum trichloride (AlCl3). The reaction is conducted at low temperatures, typically between 0°C and 20°C, to ensure regioselectivity and prevent poly-acylation. Crucially, the patent describes a one-pot protocol where, upon completion of the acylation, bromine is introduced directly into the reaction mixture. The acetyl group acts as a moderate deactivator but directs the incoming bromine electrophile to the para-position of the other phenyl ring due to steric and electronic factors, yielding 4-bromo-4'-acetylbiphenyl with high specificity. This tandem operation is a masterclass in atom economy and operational efficiency.
Following the formation of the bromo-acetyl intermediate, the process employs a sophisticated oxidation strategy to install the hydroxyl group. Using oxidants such as ammonium persulfate ((NH4)2S2O8) or potassium persulfate in an acetic acid-sulfuric acid medium, the acetyl group is converted to a hydroxyl functionality. This transformation likely proceeds through a Baeyer-Villiger type oxidation mechanism or a radical-mediated pathway facilitated by the persulfate species, ultimately cleaving the carbon-carbon bond of the acetyl group or rearranging it to form the phenol. The reaction is maintained at mild temperatures of 15°C to 40°C over a period of 10 to 20 hours to ensure complete conversion while minimizing side reactions. This step is critical for establishing the correct substitution pattern required for the final liquid crystal properties.
The final stage involves a Rosenmund-von Braun type substitution, where the aryl bromide is converted to the nitrile using cuprous cyanide (CuCN). This reaction is performed in high-boiling polar aprotic solvents such as N-methylpyrrolidone (NMP) or dimethylformamide (DMF) at elevated temperatures ranging from 150°C to 200°C. The use of strictly anhydrous conditions is paramount here to prevent the hydrolysis of the nitrile group or the deactivation of the copper catalyst. The excess of CuCN (at least 16% molar excess) drives the equilibrium towards the product, ensuring high yields of 4-cyano-4'-hydroxybiphenyl. The rigorous control of moisture and temperature in this step is essential for maintaining the integrity of the cyano group, which is the defining functional feature of this liquid crystal intermediate.
How to Synthesize 4-Cyano-4'-Hydroxybiphenyl Efficiently
The synthesis of 4-cyano-4'-hydroxybiphenyl via this patented route offers a clear roadmap for industrial implementation, balancing reaction efficiency with safety and scalability. The process is defined by three distinct operational phases: the tandem acylation-bromination, the oxidative hydroxylation, and the thermal cyanation. Each phase requires specific attention to stoichiometry, temperature control, and solvent management to achieve the reported yields and purity levels. The following guide summarizes the critical operational parameters derived from the patent examples, providing a foundational framework for process engineers looking to adopt this technology.
- Perform a one-pot Friedel-Crafts acylation and bromination on biphenyl using acetyl chloride and aluminum trichloride, followed by direct bromination without intermediate isolation.
- Execute an oxidative conversion of the acetyl group to a hydroxyl group using persulfate oxidants in an acidic acetic acid medium at controlled temperatures.
- Complete the synthesis via a high-temperature substitution reaction with cuprous cyanide in a polar aprotic solvent to install the cyano group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biphenyl-based synthesis route offers tangible strategic benefits that extend beyond simple chemistry. The primary advantage lies in the drastic simplification of the raw material portfolio. By anchoring the synthesis on biphenyl, a bulk commodity chemical with a stable global supply, manufacturers insulate themselves from the volatility associated with specialized protected phenols. This shift not only stabilizes input costs but also diversifies the supplier base, reducing the risk of single-source bottlenecks that can disrupt production schedules. The elimination of the iodination step further contributes to cost reduction by removing the need for expensive iodine reagents and the complex waste treatment protocols required to handle iodine-containing effluents.
- Cost Reduction in Manufacturing: The telescoped acylation-bromination step significantly lowers processing costs by combining two reaction stages into a single vessel operation. This consolidation reduces solvent consumption, energy usage for heating and cooling cycles, and labor hours associated with intermediate isolation and drying. Furthermore, the avoidance of hydroxyl protection groups eliminates the cost of protecting reagents and the subsequent deprotection steps, streamlining the entire value chain. The cumulative effect of these efficiencies results in a substantially lower cost of goods sold, enhancing the competitiveness of the final liquid crystal material in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, particularly the tolerance of the oxidation step to various persulfate oxidants, provides flexibility in raw material sourcing. Manufacturers are not locked into a single proprietary reagent but can choose from a range of commercially available oxidants based on price and availability. Additionally, the simplified purification process, driven by the absence of difficult-to-remove iodine impurities, leads to faster batch release times. This agility allows supply chain teams to respond more rapidly to fluctuations in demand from downstream display panel manufacturers, ensuring consistent product availability.
- Scalability and Environmental Compliance: From an environmental perspective, this route offers a greener profile by reducing the generation of halogenated waste streams associated with iodination. The use of standard organic solvents like dichloromethane and acetic acid, which have well-established recovery and recycling protocols, facilitates compliance with increasingly stringent environmental regulations. The process is inherently scalable, as demonstrated by the use of standard reactor configurations (e.g., 2-liter three-neck flasks in examples, easily translatable to larger vessels) and moderate temperature ranges. This scalability ensures that production can be ramped up to meet commercial volumes without requiring exotic equipment or hazardous high-pressure conditions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in patent CN100460385C, offering clarity on the practical aspects of adopting this technology for commercial production.
Q: Why is the biphenyl-based route superior to traditional biphenol methods for 4-cyano-4'-hydroxybiphenyl?
A: The biphenyl-based route described in patent CN100460385C eliminates the need for hydroxyl protection and, crucially, avoids iodination reactions. Traditional methods often introduce iodine impurities that are difficult to remove and destabilize product quality, whereas the new acylation-bromination pathway ensures a cleaner impurity profile suitable for high-end electronic applications.
Q: What are the critical process controls for the oxidation step in this synthesis?
A: The oxidation step requires precise temperature control between 15°C and 40°C using oxidants like ammonium persulfate in an acetic acid-sulfuric acid medium. Maintaining these conditions is vital to ensure the complete conversion of the acetyl group to the hydroxyl functionality without over-oxidation or degradation of the bromo-substituted biphenyl backbone.
Q: How does this manufacturing process impact supply chain reliability for liquid crystal manufacturers?
A: By utilizing biphenyl, a widely available commodity chemical, instead of specialized protected biphenols, the process significantly reduces raw material sourcing risks. Furthermore, the elimination of the iodination step simplifies the purification workflow, leading to more consistent batch-to-batch quality and reduced lead times for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Cyano-4'-Hydroxybiphenyl Supplier
The technological advancements outlined in patent CN100460385C underscore the immense potential of optimizing intermediate synthesis for the electronics sector. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory processes into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the absence of critical impurities like iodine.
We invite global partners to collaborate with us to leverage this cost-effective and high-quality synthesis route. Whether you require a Customized Cost-Saving Analysis for your existing supply chain or need to validate the feasibility of this new route for your specific application, our technical procurement team is ready to assist. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value and reliability in your liquid crystal material supply chain.
