Advanced Synthesis of Glufosinate-Ammonium Intermediates for Commercial Scale-Up
Advanced Synthesis of Glufosinate-Ammonium Intermediates for Commercial Scale-Up
The global demand for high-efficiency, low-toxicity herbicides continues to drive innovation in agrochemical intermediate manufacturing. A pivotal advancement in this sector is detailed in patent CN110590836B, which discloses a novel synthetic method for producing glufosinate-ammonium intermediates. This technology addresses long-standing challenges in the production of glufosinate-ammonium aminonitrile, a critical precursor for the widely used non-selective herbicide glufosinate-ammonium. By shifting away from traditional cyaniding reagents that generate problematic waste streams, this process offers a cleaner, more economically viable pathway for commercial scale-up of complex organophosphorus compounds. For R&D directors and procurement strategists, understanding this shift is essential for optimizing supply chains and reducing environmental liabilities in herbicide manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of glufosinate-ammonium intermediates has relied heavily on sodium cyanide or potassium cyanide as the primary cyaniding agents. While chemically effective, these reagents introduce a significant downstream burden: the generation of mixed salts, specifically ammonium chloride alongside sodium chloride or potassium chloride. Separating these mixed salts is technically arduous and economically draining, often requiring extensive washing and purification steps that lower overall yield. Furthermore, alternative methods utilizing trimethylsilyl cyanide (TMSCN) have been explored to bypass salt issues, but TMSCN is prohibitively expensive for bulk industrial applications and suffers from poor atom economy. These conventional limitations create bottlenecks in cost reduction in herbicide manufacturing, forcing producers to balance between high raw material costs and expensive waste treatment protocols.
The Novel Approach
The methodology outlined in patent CN110590836B presents a transformative solution by employing ammonium cyanide (NH4CN) as the cyaniding reagent. This approach fundamentally alters the reaction landscape by ensuring that the only ammonium salt generated is ammonium chloride, which can be easily managed or recycled, completely eliminating the formation of intractable mixed sodium/ammonium salts. The process begins with the preparation of methyl propionaldehyde ethyl phosphate via a Michael addition and subsequent hydrolysis, followed by a Strecker reaction with ammonium cyanide in the presence of ammonia water and ammonium chloride. This strategic substitution not only utilizes a significantly cheaper reagent compared to TMSCN but also streamlines the purification process, making it highly suitable for reliable agrochemical intermediate supplier operations aiming for high throughput and minimal environmental impact.
Mechanistic Insights into the Phosphine-Aldehyde Strecker Reaction
The core of this synthesis lies in a two-stage mechanistic sequence that ensures high fidelity and purity of the final product. The first stage involves the construction of the carbon-phosphorus backbone. Diethyl methylphosphite undergoes a Michael addition with acrolein in an ethanol solvent system. This addition is highly regioselective, placing the phosphorus group at the beta-position relative to the carbonyl functionality after subsequent hydrolysis. The resulting acetal intermediate is then subjected to acidic hydrolysis, cleaving the ethoxy groups to reveal the reactive aldehyde functionality, yielding methyl propionaldehyde ethyl phosphate. This precursor is crucial as it sets the stereochemical and structural foundation for the final herbicide active ingredient.
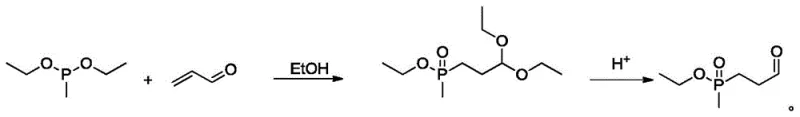
Following the formation of the phosphine-aldehyde, the process proceeds to the critical Strecker reaction phase. In this step, the aldehyde reacts with ammonium cyanide and ammonia to form an alpha-aminonitrile. The presence of ammonium chloride acts as a buffer and catalyst, facilitating the formation of the imine intermediate which is then trapped by the cyanide ion. The use of aqueous ammonium cyanide, prepared in situ from sodium cyanide and ammonium bicarbonate, ensures a steady supply of the nucleophile while precipitating out sodium bicarbonate, further purifying the reaction medium before the main coupling occurs. This mechanism avoids the introduction of extraneous cations that would otherwise complicate the salt profile of the final mixture.
Impurity control is inherently built into this mechanistic design. By avoiding sodium-based cyanides in the final coupling step, the process prevents the co-crystallization of sodium salts with the product, which is a common issue in traditional routes. The reaction conditions are maintained at mild temperatures, typically between 15°C and 30°C, which minimizes thermal degradation of the sensitive nitrile and phosphonate groups. Post-reaction, the mixture is treated with hydrochloric acid to hydrolyze any remaining imines and then neutralized with ammonia gas, allowing for the selective precipitation of ammonium chloride. This precise control over pH and temperature ensures that the resulting high-purity glufosinate intermediate meets stringent specifications required for downstream pharmaceutical and agrochemical applications.
How to Synthesize Glufosinate-Ammonium Aminonitrile Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize yield and minimize byproduct formation. The process is designed to be scalable, moving seamlessly from laboratory benchtop to pilot plant operations. The initial preparation of the ammonium cyanide solution is a key prerequisite, ensuring that the cyanide source is free of sodium contaminants before it ever touches the phosphine aldehyde. Operators must monitor the exothermic nature of the Michael addition and the subsequent acid hydrolysis to prevent runaway reactions. Detailed standard operating procedures regarding the molar ratios of ammonia, ammonium chloride, and the aldehyde are critical for reproducibility.
- Perform Michael addition of diethyl methylphosphite with acrolein in ethanol, followed by acidic hydrolysis to generate methyl propionaldehyde ethyl phosphate.
- Prepare an aqueous ammonium cyanide solution by reacting sodium cyanide with ammonium bicarbonate and filtering off sodium bicarbonate precipitate.
- React the phosphine aldehyde with ammonium cyanide, ammonium chloride, and ammonia water at 15-30°C, followed by acidification and recrystallization to obtain the final aminonitrile intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers tangible strategic benefits beyond simple chemical efficiency. The primary advantage lies in the drastic simplification of the waste stream profile. By eliminating the generation of mixed sodium and ammonium chloride salts, manufacturers can significantly reduce the costs associated with wastewater treatment and solid waste disposal. This reduction in environmental compliance burden translates directly into lower operational expenditures and a smaller carbon footprint, aligning with modern sustainability goals. Furthermore, the reliance on ammonium cyanide, which can be generated from commodity chemicals like sodium cyanide and ammonium bicarbonate, insulates the supply chain from the volatility of specialized reagent markets.
- Cost Reduction in Manufacturing: The substitution of expensive trimethylsilyl cyanide with readily available ammonium cyanide results in substantial raw material cost savings. Unlike TMSCN, which requires specialized handling and generates silanol byproducts, ammonium cyanide is a commodity reagent that integrates smoothly into existing infrastructure. Additionally, the elimination of complex salt separation steps reduces the consumption of solvents and energy required for purification. This streamlined workflow allows for a more lean manufacturing process, where resources are focused on product formation rather than waste remediation, driving down the overall cost per kilogram of the intermediate.
- Enhanced Supply Chain Reliability: Sourcing high-purity intermediates is often bottlenecked by the availability of specialized reagents. This method relies on basic industrial chemicals that are widely produced and stocked globally, ensuring a robust and resilient supply chain. The ability to prepare the cyaniding agent in situ further reduces the logistical burden of transporting hazardous cyanide salts, as the precursors are more stable and easier to handle. This flexibility enables suppliers to maintain consistent production schedules even during periods of raw material market fluctuation, ensuring reducing lead time for high-purity agrochemical intermediates for their downstream clients.
- Scalability and Environmental Compliance: The process is inherently designed for large-scale industrial production, with reaction conditions that are easily controlled in standard stainless steel reactors. The mild temperature range (15-30°C) reduces the need for extreme cooling or heating utilities, lowering energy costs. From an environmental perspective, the 'source reduction' of mixed salts means that the facility faces fewer regulatory hurdles regarding effluent discharge. This compliance advantage accelerates the permitting process for new production lines and ensures long-term operational continuity without the risk of shutdowns due to waste treatment capacity limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their current manufacturing portfolios.
Q: How does this new synthesis method improve waste management compared to traditional routes?
A: Traditional methods using sodium cyanide generate significant amounts of mixed ammonium chloride and sodium chloride salts, which are difficult and costly to separate and treat. This patented process utilizes ammonium cyanide, effectively avoiding the formation of sodium salts entirely, thereby solving the waste salt treatment problem at the source and simplifying downstream purification.
Q: Why is ammonium cyanide preferred over trimethylsilyl cyanide (TMSCN) for this application?
A: While TMSCN is a viable cyaniding reagent, it is significantly more expensive and exhibits poorer atom economy, making it less suitable for large-scale industrial production. The use of ammonium cyanide provides a much more cost-effective alternative without compromising the yield or purity of the glufosinate-ammonium aminonitrile intermediate.
Q: What are the critical reaction conditions for the Strecker step in this process?
A: The Strecker reaction is conducted under mild conditions, typically between 15°C and 30°C. The molar ratio of the phosphine aldehyde to ammonium cyanide is optimized between 1:1.5 and 1:2.5, ensuring high conversion rates while minimizing excess reagent usage and facilitating easier workup procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glufosinate-Ammonium Aminonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, sustainable synthesis routes in the modern agrochemical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of organophosphorus chemistry, ensuring that every batch of glufosinate intermediate meets stringent purity specifications. With our rigorous QC labs and commitment to process safety, we provide a secure supply base for global herbicide manufacturers looking to optimize their upstream supply chains.
We invite you to collaborate with us to leverage this advanced synthesis technology for your production needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your operational efficiency and product quality.
