Advanced Nickel-Catalyzed Resolution for High-Purity L-Glufosinate-Ammonium Production
Advanced Nickel-Catalyzed Resolution for High-Purity L-Glufosinate-Ammonium Production
The global demand for high-efficiency, low-toxicity herbicides has placed L-glufosinate-ammonium at the forefront of agrochemical innovation. However, traditional manufacturing routes have long struggled with the economic and environmental burdens of separating chiral isomers. A groundbreaking technical solution is presented in patent CN112979701A, which discloses a highly efficient preparation method utilizing a nickel-salt catalyzed complexation strategy. This technology represents a paradigm shift from expensive noble metal catalysis and complex enzymatic processes to a robust, base-metal-driven resolution system. By leveraging specific chiral ligands in conjunction with nickel salts under alkaline conditions, this method achieves a seamless conversion of racemic glufosinate derivatives into the biologically active L-isomer. The process not only delivers exceptional stereocontrol but also addresses critical supply chain pain points regarding catalyst cost and ligand recovery, positioning it as a superior choice for large-scale industrial application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure L-glufosinate-ammonium has been plagued by significant technical and economic inefficiencies inherent in legacy technologies. For instance, early methods relying on quinine crystallization, as reported in U.S. Pat. No. 5,5767309, suffer from inherently low yields, typically capping around 86%, and necessitate the use of expensive resolving agents like 3,5-dinitrosalicylaldehyde which are difficult to recover economically. Similarly, asymmetric hydrogenation routes utilizing chiral rhodium-phosphorus catalysts, while offering mild conditions, introduce prohibitive costs due to the scarcity of rhodium and the complexity of ligand synthesis. Enzymatic approaches, though selective, often face challenges with cofactor regeneration (such as NADPH), long reaction times extending up to 14 days, and the difficulty of recycling expensive biological catalysts. These conventional pathways frequently result in the wasteful discarding of the D-isomer, effectively halving the potential yield from racemic starting materials and generating substantial chemical waste that complicates environmental compliance.
The Novel Approach
In stark contrast to these legacy constraints, the novel approach detailed in CN112979701A introduces a streamlined nickel-mediated resolution that fundamentally alters the cost structure of L-glufosinate manufacturing. This method employs a sophisticated coordination chemistry strategy where (L,D)-glufosinate-ammonium derivatives form a transient metal complex with a nickel salt and a specifically designed chiral ligand under alkaline conditions. The brilliance of this system lies in its ability to dynamically convert the inactive D-isomer into the desired L-form during the reaction sequence, thereby breaking the 50% yield barrier typical of classical resolutions. Following complexation, a controlled acid hydrolysis step releases the high-purity L-glufosinate-ammonium while precipitating the chiral ligand for immediate recovery and reuse. This closed-loop capability significantly reduces raw material consumption and simplifies the downstream purification process, offering a scalable pathway that aligns perfectly with modern green chemistry principles and cost-reduction mandates in agrochemical manufacturing.
Mechanistic Insights into Nickel-Catalyzed Chiral Resolution
The core of this technological breakthrough resides in the precise stereoelectronic interactions between the nickel center and the chiral ligand, which creates a highly differentiated environment for the substrate. The reaction initiates with the formation of a stable metal-ligand complex in the presence of a base, such as sodium hydroxide or potassium carbonate, within polar protic solvents like ethanol or methanol. The chiral ligand, characterized by a pyrrolidine carboxamide backbone substituted with benzoylphenyl and benzyl groups, imposes a rigid steric constraint around the nickel ion. This chiral pocket selectively coordinates with the amino acid derivative, favoring the stabilization of one enantiomeric transition state over the other. The subsequent acidification triggers a hydrolytic cleavage of the metal-amino acid bond, releasing the L-glufosinate-ammonium into the aqueous phase while the ligand, now protonated or in a less soluble salt form, precipitates out of the solution. This elegant mechanism ensures that the chiral information is efficiently transferred to the product without the need for stoichiometric amounts of expensive chiral auxiliaries.
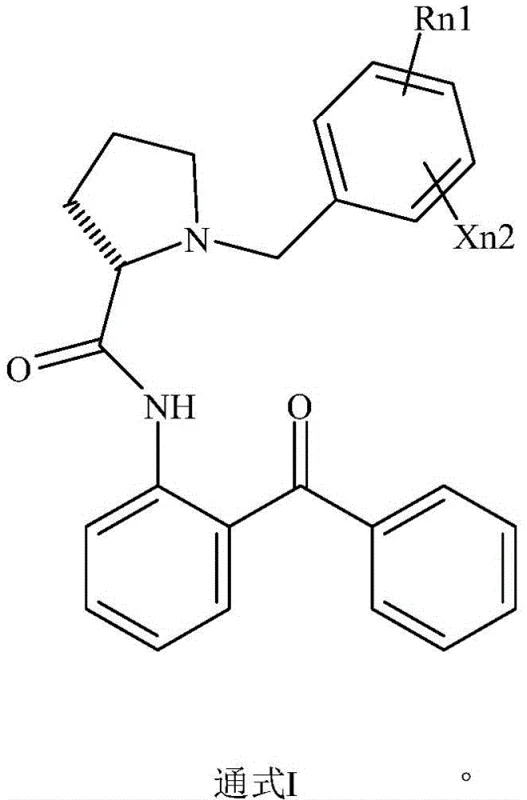
Furthermore, the impurity profile of the final product is rigorously controlled through the selectivity of the complexation step. The specific geometry of the ligand, as illustrated in the general formula, prevents the coordination of unwanted by-products or the D-isomer in a manner that would lead to co-crystallization. The hydrolysis conditions, typically maintained between 20°C and 60°C, are mild enough to prevent racemization of the newly formed chiral center, ensuring that the enantiomeric excess (ee) remains consistently above 95.0%. The use of nickel, a first-row transition metal, also eliminates the risk of toxic heavy metal contamination often associated with palladium or rhodium residues, simplifying the regulatory clearance for the final agrochemical active ingredient. This mechanistic robustness allows for a broader operating window regarding temperature and solvent composition, making the process highly tolerant to minor variations in industrial scale-up scenarios.
How to Synthesize L-Glufosinate-Ammonium Efficiently
Implementing this advanced synthesis route requires careful attention to the stoichiometry of the nickel salt and the chiral ligand to maximize complexation efficiency. The process begins by charging a reactor with the racemic glufosinate derivative, the specific chiral ligand (such as (2S)-N-(2-benzoylphenyl)-1-(4-methylbenzyl)-2-pyrrolidinecarboxamide), and a nickel source like nickel acetate tetrahydrate. The mixture is heated in an alcoholic solvent under alkaline conditions to facilitate the formation of the diastereomeric complex. Once the complexation is complete, the reaction mixture is subjected to acid hydrolysis, which liberates the target L-isomer into the solution. The detailed standardized synthetic steps, including specific molar ratios, temperature profiles, and workup procedures required to achieve >95% yield and ee, are outlined below.
- Complexation Reaction: React (L,D)-glufosinate-ammonium derivatives with a specific chiral ligand and nickel salt in the presence of alkali and solvent at 40-80°C.
- Hydrolysis: Add acid to the metal complex reaction solution to induce hydrolysis, releasing the L-glufosinate-ammonium into the solution.
- Purification: Perform post-treatment including desolventizing, filtration to recover the ligand, and ammoniation to isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this nickel-catalyzed process offers transformative economic benefits that extend far beyond simple yield improvements. The substitution of precious metals like rhodium or palladium with abundant nickel salts results in a drastic reduction in raw material costs, insulating the production budget from the volatile pricing of noble metals. Moreover, the operational simplicity of the ligand recovery—achieved merely through filtration after acidification—eliminates the need for complex chromatographic separations or energy-intensive distillation columns often required to recover chiral catalysts. This streamlined workflow translates directly into reduced utility consumption and lower labor costs per kilogram of finished product. Additionally, the ability to utilize the entire racemic feedstock by converting the D-isomer effectively doubles the throughput capacity of existing reactors without requiring capital expenditure on new infrastructure, thereby enhancing overall asset utilization rates.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the high recovery rate of the chiral ligand create a substantially leaner cost structure. By avoiding the use of stoichiometric chiral resolving agents that are consumed in the process, the variable cost per unit is significantly lowered. The process operates under relatively mild temperatures and pressures, reducing energy demands for heating and cooling compared to high-pressure hydrogenation methods. Furthermore, the simplified downstream processing reduces the consumption of solvents and auxiliary chemicals, contributing to a more economical production model that enhances margin potential in competitive agrochemical markets.
- Enhanced Supply Chain Reliability: Reliance on nickel and common organic solvents like ethanol or methanol ensures a stable and secure supply chain, free from the geopolitical risks often associated with rare earth or platinum group metals. The robustness of the chemical process allows for flexible sourcing of raw materials, as the system tolerates a variety of nickel salts and solvent mixtures. This flexibility mitigates the risk of production stoppages due to single-source supplier failures. The high yield and consistency of the reaction also mean that production schedules can be met with greater certainty, reducing the need for safety stock and enabling a more responsive just-in-time delivery model for downstream formulators.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-gram to kilogram scales with consistent results, indicating readiness for multi-ton commercial production. From an environmental perspective, the reduction in heavy metal usage and the ability to recycle the chiral ligand significantly lower the E-factor (mass of waste per mass of product). The aqueous workup and lack of toxic organometallic residues simplify wastewater treatment protocols, ensuring easier compliance with stringent environmental regulations. This green chemistry profile not only reduces disposal costs but also aligns with the sustainability goals of major global agrochemical corporations, enhancing the marketability of the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed resolution technology. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is critical for R&D teams planning technology transfer and for procurement specialists assessing long-term supply viability.
Q: What is the primary advantage of the nickel-catalyzed method over rhodium-based asymmetric hydrogenation?
A: The nickel-catalyzed method described in CN112979701A offers a significant cost advantage because nickel salts are substantially cheaper than chiral rhodium-phosphorus ligand catalysts. Furthermore, the chiral ligand in this process can be easily recovered via filtration after acidification, whereas rhodium catalysts often require complex and expensive recovery processes.
Q: How does this process handle the unwanted D-isomer of glufosinate?
A: Unlike traditional resolution methods that discard the D-isomer, this technology effectively converts D-glufosinate-ammonium into the biologically active L-glufosinate-ammonium. This dynamic kinetic resolution capability ensures that the theoretical yield can approach 100%, drastically improving atom economy compared to standard crystallization methods.
Q: What represent the typical purity and optical purity specifications achievable with this route?
A: According to the patent data, this method consistently achieves an L-glufosinate-ammonium yield of more than 95%, a chemical content of more than 95%, and an enantiomeric excess (ee) value exceeding 95.0%. These specifications meet the rigorous standards required for high-efficiency herbicide formulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Glufosinate-Ammonium Supplier
The technical superiority of the nickel-catalyzed resolution method underscores the immense potential for optimizing the global supply of L-glufosinate-ammonium. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying stringent purity specifications, including the critical ee values required for high-performance herbicides. We understand that consistency is key in the agrochemical sector, and our dedicated process engineering team is prepared to adapt this advanced chemistry to meet your specific volume and quality requirements with precision.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly manufacturing route. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current supply chain dynamics. We are ready to provide specific COA data from pilot batches and comprehensive route feasibility assessments to demonstrate how our expertise can drive value for your organization. Let us be your partner in securing a sustainable and economical supply of high-purity agrochemical intermediates.
