Revolutionizing Rosuvastatin Calcium Intermediate Production via High-Selectivity Substitution Strategies
The pharmaceutical industry continuously seeks robust synthetic pathways that maximize stereochemical purity while minimizing downstream processing burdens. A pivotal advancement in this domain is detailed in patent CN113754650A, which discloses a high-selectivity synthesis method for a critical rosuvastatin calcium intermediate. This technology addresses a persistent challenge in statin manufacturing: the control of cis-isomer impurities that often plague traditional routes. By strategically modifying the starting thiol reagents, the inventors have achieved a dramatic reduction in stereoisomeric byproducts, shifting the impurity profile from an unacceptable 1-2% range down to less than 0.1%. For R&D directors and process chemists, this represents a significant leap forward in quality by design, offering a pathway to higher purity active pharmaceutical ingredients with reduced reliance on complex chromatographic separations. The methodology leverages well-established organic transformations but optimizes them through precise substrate engineering, ensuring that the final olefination step proceeds with exceptional geometric control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of rosuvastatin intermediates has relied heavily on the condensation of chloromethyl dioxolane derivatives with unsubstituted mercaptobenzothiazole. While this conventional route is chemically feasible, it suffers from inherent stereoselectivity issues that manifest late in the synthesis. Specifically, the use of unsubstituted thiols leads to a significant formation of cis-isomers during the subsequent Julia-Kocienski olefination, typically resulting in impurity levels between 1% and 2%. These cis-isomers are structurally similar to the desired trans-product, making them notoriously difficult and expensive to remove via standard crystallization techniques. Consequently, manufacturers face increased operational costs due to lower overall yields and the necessity for rigorous purification steps to meet pharmacopeial standards. Furthermore, the presence of these isomers can complicate regulatory filings, as impurity qualification becomes more arduous when dealing with genotoxic or structurally alerting contaminants that persist through multiple synthetic steps.
The Novel Approach
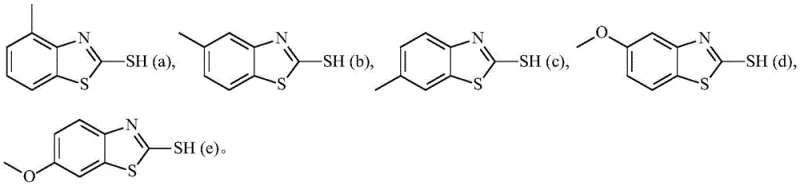
The innovative strategy presented in the patent data fundamentally alters the electronic and steric environment of the reaction by introducing substituents on the benzene ring of the mercaptobenzothiazole starting material. Instead of the plain heterocycle, the process utilizes derivatives such as 5-methyl-2-mercaptobenzothiazole or 6-methoxy-2-mercaptobenzothiazole. This subtle structural modification exerts a profound influence on the transition state of the subsequent olefination reaction, effectively suppressing the formation of the unwanted cis-alkene geometry. As demonstrated in the experimental data, switching to these substituted reagents reduces the cis-isomer content to negligible levels, often below the detection limit of 0.1%. This approach not only streamlines the purification workflow but also enhances the overall atom economy of the process by maximizing the yield of the desired E-isomer. The versatility of this method is further highlighted by the compatibility with various substituents, allowing process chemists to fine-tune reactivity based on specific supply chain availability.
Mechanistic Insights into Julia-Kocienski Olefination Selectivity
The core of this technological breakthrough lies in the mechanistic nuances of the Julia-Kocienski olefination, a reaction widely favored for constructing E-alkenes in complex molecule synthesis. In this specific application, the sulfone intermediate derived from the substituted benzothiazole acts as the nucleophilic partner against the pyrimidine aldehyde. The presence of electron-donating or sterically bulky groups on the benzothiazole ring appears to stabilize the specific conformation of the betaine intermediate or the transition state leading to the E-alkene. By carefully controlling the reaction temperature, specifically maintaining cryogenic conditions between -50°C and 0°C during the base addition, the kinetic control of the elimination step is maximized. This precise thermal management, combined with the electronic effects of the substituents, ensures that the thermodynamic preference for the trans-geometry is overwhelmingly favored. Such mechanistic understanding allows for the reproducible scaling of the reaction, as the parameters are robust enough to tolerate minor fluctuations while maintaining high stereoselectivity.
Furthermore, the oxidation step converting the sulfide to the sulfone is critical for the success of the olefination. The patent specifies the use of hydrogen peroxide in conjunction with molybdenum-based catalysts, such as ammonium molybdate or phosphomolybdic acid. This catalytic system is highly efficient, operating under mild conditions (30-50°C) to prevent the degradation of the sensitive acetal protecting groups present in the molecule. The choice of oxidant is also commercially prudent, as hydrogen peroxide generates water as the only byproduct, aligning with green chemistry principles. The resulting sulfone possesses the necessary acidity at the alpha-position to facilitate smooth deprotonation by strong bases like sodium tert-butoxide. This sequence ensures that the sulfur moiety acts as an effective leaving group during the elimination phase, driving the reaction to completion with minimal side reactions such as epoxide formation or aldehyde self-condensation.
How to Synthesize Rosuvastatin Intermediate Efficiently
Implementing this high-selectivity synthesis requires strict adherence to the optimized reaction parameters outlined in the patent documentation. The process begins with a phase-transfer catalyzed condensation, followed by a controlled oxidation, and culminates in a low-temperature olefination. Each step has been refined to balance reaction rate with selectivity, ensuring that the final product meets the rigorous demands of API manufacturing. The following guide summarizes the critical operational phases required to achieve the reported purity and yield profiles. For detailed standard operating procedures and specific stoichiometric ratios, please refer to the technical breakdown below.
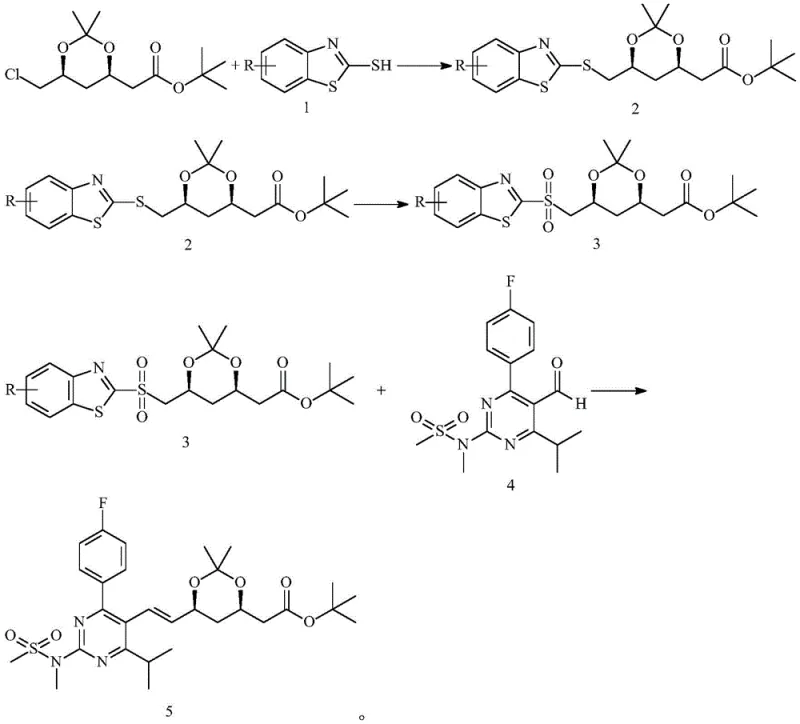
- Condense (4R-cis)-6-chloromethyl-2,2-dimethyl-1,3-dioxolane-4-tert-butyl acetate with benzene ring-substituted mercaptobenzothiazole using inorganic base and phase transfer catalyst.
- Oxidize the resulting sulfide intermediate to the corresponding sulfone using hydrogen peroxide and a molybdenum-based catalyst.
- Perform Julia-Kocienski olefination between the sulfone and pyrimidine aldehyde at low temperature using organic base to yield the final alkene intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this high-selectivity synthesis route offers substantial strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. By reducing the cis-isomer impurity from percent-level quantities to trace amounts, the need for extensive recrystallization cycles or preparative chromatography is virtually eliminated. This reduction in downstream processing directly translates to shorter batch cycle times and increased throughput capacity within existing manufacturing facilities. For supply chain heads, this means a more reliable and consistent supply of the intermediate, as the risk of batch failure due to out-of-specification impurity profiles is significantly mitigated. The robustness of the process also implies a lower dependency on specialized purification equipment, allowing for greater flexibility in manufacturing site selection.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of yield losses associated with removing difficult cis-isomers. In traditional processes, a significant portion of the crude material is often discarded or relegated to rework loops to achieve the necessary purity. By preventing the formation of these impurities at the source, the overall material efficiency is markedly improved. Additionally, the use of commodity oxidants like hydrogen peroxide and readily available substituted thiols avoids the need for exotic or prohibitively expensive reagents. This combination of higher yield and lower reagent costs results in a significantly reduced cost of goods sold (COGS), providing a competitive edge in the pricing of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reliance on stable, bench-top reagents enhances the resilience of the supply chain. Substituted mercaptobenzothiazoles are commercially accessible and do not suffer from the supply volatility often seen with highly specialized chiral catalysts. Furthermore, the mild reaction conditions reduce the stress on reactor hardware and minimize safety risks associated with high-pressure or high-temperature operations. This operational safety profile facilitates smoother regulatory audits and reduces the likelihood of unplanned shutdowns. For procurement managers, this translates to a more predictable lead time and a secure supply of critical intermediates, ensuring continuity of production for the final rosuvastatin calcium drug product.
- Scalability and Environmental Compliance: Scaling this process to commercial volumes is facilitated by the absence of complex unit operations. The reaction workups involve standard extraction and crystallization techniques that are easily transferable from pilot plant to multi-ton production scales. Moreover, the environmental footprint of the process is favorable; the use of aqueous hydrogen peroxide and the generation of benign byproducts align with increasingly stringent environmental regulations. The reduction in solvent usage for purification further contributes to a greener manufacturing profile. This sustainability aspect is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility goals and reduce waste disposal costs associated with hazardous solvent streams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and claims within the patent literature, providing a clear understanding of the technology's capabilities. Understanding these details is essential for evaluating the feasibility of integrating this route into your existing manufacturing portfolio.
Q: How does the new synthesis method improve impurity profiles compared to prior art?
A: By utilizing benzene ring-substituted mercaptobenzothiazoles instead of unsubstituted variants, the stereoselectivity is significantly enhanced, reducing cis-isomer impurities from 1-2% down to below 0.1%.
Q: What are the critical reaction conditions for the oxidation step?
A: The oxidation requires mild temperatures between 10-50°C using hydrogen peroxide as the oxidant and catalysts like ammonium molybdate or phosphomolybdic acid to ensure high conversion without over-oxidation.
Q: Why is controlling the cis-isomer content crucial for rosuvastatin production?
A: Minimizing cis-isomer impurities at the intermediate stage prevents their carryover into the final API, thereby simplifying downstream purification and ensuring the final drug product meets stringent regulatory purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high-selectivity benefits of this patent can be fully realized at an industrial level. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation. This commitment to quality ensures that every batch of rosuvastatin intermediate we deliver meets the exacting standards required for global regulatory submissions, providing our partners with peace of mind and supply security.
We invite you to leverage our technical capabilities to optimize your statin supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis methods can drive value for your organization. Let us collaborate to bring high-quality, cost-effective cardiovascular medications to the market faster and more efficiently.
