Advanced Synthesis of Brinzolamide Key Intermediate for Commercial Scale-Up
Advanced Synthesis of Brinzolamide Key Intermediate for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and cost-effective pathways for producing critical active pharmaceutical ingredients (APIs) and their precursors. Patent CN113354665B introduces a significant technological breakthrough in the synthesis of the key intermediate for Brinzolamide, a potent carbonic anhydrase inhibitor widely used in treating glaucoma and ocular hypertension. This novel method addresses long-standing inefficiencies in traditional manufacturing routes by streamlining the synthesis of Compound V, a pivotal precursor. By shifting away from complex chiral resolution steps and expensive reagents, this technology offers a streamlined alkylation and cyclization strategy that enhances both economic viability and operational safety. For global stakeholders, this represents a shift towards more sustainable and reliable pharmaceutical intermediate supply chains, ensuring consistent quality for downstream drug formulation.
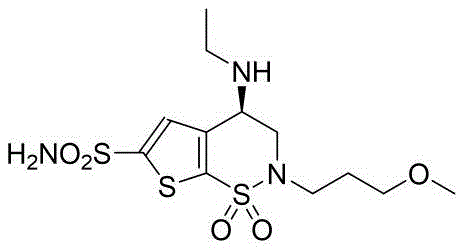
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Brinzolamide intermediates has been plagued by significant technical and economic hurdles that hinder efficient large-scale production. As detailed in prior art such as patent CA2114877C, traditional routes often rely heavily on the use of diisopinocampheylchloroborane, an exceptionally expensive chiral reducing agent required to establish the necessary stereochemistry. Furthermore, these legacy processes mandate rigorous low-temperature conditions, often dropping to -30°C, which imposes severe energy demands and limits reactor throughput. Alternative routes reported by other entities involve multi-step sequences starting from racemic mixtures followed by oxidation and chiral reduction, which not only complicates the process flow but also introduces additional purification challenges. These factors collectively result in elevated production costs, extended lead times, and a higher environmental footprint due to the intensive resource consumption required for cryogenic cooling and specialized reagent handling.
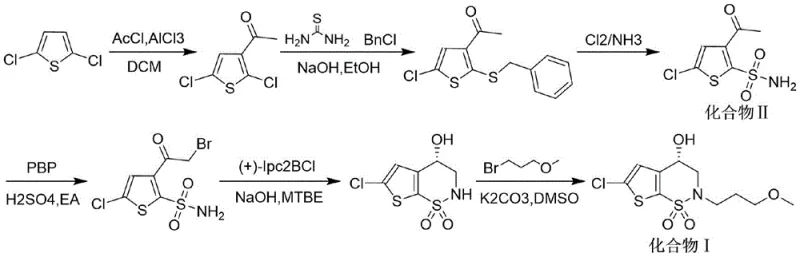
The Novel Approach
In stark contrast to these cumbersome legacy methods, the methodology disclosed in CN113354665B presents a radically simplified and economically superior pathway. This innovative approach initiates with Compound II, utilizing a direct alkylation reaction with 1-bromo-3-methoxypropane to form Compound III, thereby bypassing the need for early-stage chiral induction. The subsequent bromination and intramolecular cyclization steps proceed under mild thermal conditions, typically between 60°C and 80°C, eliminating the necessity for energy-intensive cryogenic systems. By replacing costly borane reagents with accessible alkylating agents and standard bases like potassium carbonate or sodium tert-butoxide, the new route drastically reduces raw material expenditures. This strategic redesign of the synthetic logic not only accelerates the reaction timeline but also simplifies the isolation and purification of intermediates, making it an ideal candidate for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios.
Mechanistic Insights into Alkylation and Cyclization Strategy
The core of this technological advancement lies in the precise orchestration of nucleophilic substitution and ring-closing reactions that construct the complex thieno[3,2-e]-1,2-thiazine scaffold. The initial alkylation step involves the nucleophilic attack of the sulfonamide nitrogen in Compound II upon the terminal carbon of 1-bromo-3-methoxypropane. This transformation is facilitated by the presence of phase transfer catalysts such as benzyltriethylammonium chloride and inorganic bases like potassium carbonate in polar aprotic solvents like dimethyl sulfoxide. The reaction kinetics are optimized at temperatures of 70-80°C, ensuring complete conversion while minimizing side reactions. Following this, the bromination of the acetyl group in Compound III to form the alpha-bromo ketone (Compound IV) is achieved using reagents like dibromohydantoin in the presence of an acid catalyst, creating a highly reactive electrophilic center primed for the final cyclization event.
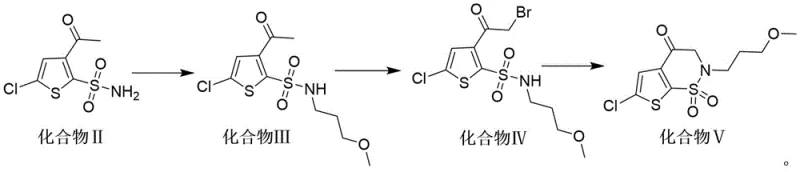
The final and most critical mechanistic step is the intramolecular cyclization of Compound IV to yield the target Compound V. This transformation is driven by a strong base, such as sodium tert-butoxide, which deprotonates the sulfonamide nitrogen, triggering a nucleophilic attack on the adjacent alpha-bromo carbonyl carbon. This intramolecular SN2 reaction effectively closes the six-membered thiazine ring, establishing the core architecture of the Brinzolamide intermediate. The choice of tetrahydrofuran as the solvent for this step is crucial, as it provides the necessary solubility for the organic substrates while maintaining stability under basic conditions. The entire sequence is designed to maximize atom economy and minimize impurity formation, resulting in a product with high structural integrity and purity profiles that meet stringent pharmaceutical standards without the need for complex chromatographic separations.
How to Synthesize Brinzolamide Key Intermediate Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to ensure optimal yield and purity. The process begins with the alkylation of Compound II, where precise control of stoichiometry and temperature is essential to drive the reaction to completion. Following the isolation of Compound III, the bromination step must be monitored to prevent over-bromination or degradation of the sensitive thiophene ring. Finally, the cyclization step demands anhydrous conditions and controlled addition of the base to manage the exotherm and ensure clean ring closure. For detailed operational protocols and specific reagent grades required for GMP compliance, please refer to the standardized synthesis guide below.
- Perform alkylation of Compound II with 1-bromo-3-methoxypropane using potassium carbonate and a phase transfer catalyst in DMSO at 70-80°C to yield Compound III.
- Conduct bromination of Compound III using dibromohydantoin and p-toluenesulfonic acid in methanol under reflux conditions to obtain Compound IV.
- Execute intramolecular cyclization of Compound IV using sodium tert-butoxide in tetrahydrofuran at 65-80°C to finalize the synthesis of Compound V.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. By fundamentally altering the raw material profile, this method eliminates the dependency on volatile and high-cost chiral boranes, which are often subject to supply shortages and price fluctuations. The shift to commodity chemicals like 1-bromo-3-methoxypropane and standard inorganic bases ensures a stable and predictable supply chain, reducing the risk of production stoppages. Furthermore, the ability to conduct reactions at near-ambient or moderately elevated temperatures significantly lowers utility costs associated with refrigeration, contributing to substantial cost savings in API manufacturing. This efficiency gain allows manufacturers to offer more competitive pricing while maintaining healthy margins, a critical factor in the highly regulated ophthalmic drug market.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and the reduction of reaction steps lead to a drastic decrease in overall production costs. By utilizing cheaper starting materials and avoiding cryogenic operations, the process significantly lowers both raw material expenditure and energy consumption, enhancing the economic feasibility of large-scale production.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial chemicals rather than specialized reagents ensures a robust supply chain that is less susceptible to geopolitical or logistical disruptions. This stability guarantees consistent delivery schedules for downstream partners, fostering stronger long-term business relationships and reducing inventory holding risks.
- Scalability and Environmental Compliance: The simplified workflow and mild reaction conditions facilitate seamless scale-up from pilot batches to multi-ton commercial production without the need for specialized equipment. Additionally, the reduced use of hazardous reagents and lower energy demand align with modern green chemistry principles, simplifying waste treatment and ensuring compliance with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is vital for stakeholders evaluating its integration into their supply networks. The following questions address common inquiries regarding process safety, scalability, and quality control, derived directly from the technical disclosures in the patent literature. These insights are intended to provide clarity on how this method compares to existing technologies and what expectations partners should have regarding performance metrics and regulatory compliance.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: The primary advantage is the elimination of expensive chiral reducing agents like diisopinocampheylchloroborane and the avoidance of harsh low-temperature conditions (-30°C), significantly lowering production costs and energy consumption.
Q: What is the expected purity and yield of the final Compound V?
A: According to the patent data, the process achieves high purity levels exceeding 97.8% and molar yields ranging from 85% to 91% across multiple experimental examples, ensuring suitability for pharmaceutical applications.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes readily available raw materials, standard solvents like DMSO and THF, and mild reaction temperatures (60-80°C), making it highly scalable and safe for commercial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brinzolamide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement the novel alkylation-cyclization route described in CN113354665B positions us as a strategic partner capable of meeting the demanding volume and quality requirements of top-tier ophthalmic drug manufacturers.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
