Advanced One-Step Synthesis of Geraniol Thioacetate for Industrial Flavor Applications
The chemical industry is witnessing a significant paradigm shift in the synthesis of sulfur-containing terpenoids, driven by the urgent need for greener, more efficient manufacturing protocols. A pivotal development in this domain is disclosed in patent CN110156652B, which details a novel preparation method for geraniol thioacetate (also known as thioglycollate geraniol ester). This compound serves as a critical intermediate in the flavors, fragrances, and pharmaceutical sectors, valued for its unique aromatic profile and biological activity. The patented technology fundamentally reimagines the synthetic route by replacing complex, multi-step sequences with a streamlined, one-step esterification process. By leveraging pyridine as a catalyst in a dichloromethane solvent system, the inventors have achieved a reaction that proceeds smoothly at room temperature, thereby circumventing the energy-intensive and hazardous conditions typical of legacy methods. This breakthrough not only enhances the economic viability of producing high-purity flavor intermediates but also aligns with modern sustainability goals by reducing waste and energy consumption. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a competitive edge in the supply of high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfur-containing terpene derivatives like geraniol thioacetate has been plagued by inefficiencies and operational complexities that hinder large-scale adoption. Traditional literature, such as reports involving citral as a starting material, describes a cumbersome three-step sequence that begins with a 1,4-addition reaction followed by a reduction and finally an acetylation. This conventional pathway necessitates the use of harsh reagents, including sodium borohydride at cryogenic temperatures (0°C), which significantly escalates energy costs and safety risks. Furthermore, the high reactivity of intermediates like acetyl halides often leads to uncontrolled side reactions, resulting in a complex impurity profile that is difficult to separate. The cumulative yield of such multi-step processes is inherently low due to material losses at each stage, and the generation of substantial chemical waste poses environmental compliance challenges. These factors collectively render traditional methods economically unattractive for modern manufacturers seeking to optimize their cost structures and minimize their carbon footprint in the production of specialty chemicals.
The Novel Approach
In stark contrast to the arduous traditional pathways, the method disclosed in patent CN110156652B introduces a remarkably elegant and efficient one-step synthesis. This innovative approach utilizes geraniol and thioacetic acid as directly accessible raw materials, reacting them in the presence of pyridine within a dichloromethane medium. The reaction proceeds under mild conditions at room temperature for a duration of 4 to 10 hours, eliminating the need for extreme heating or cooling. This simplification drastically reduces the operational burden on manufacturing facilities, allowing for simpler reactor configurations and lower utility consumption. The direct esterification avoids the formation of unstable intermediates, thereby minimizing byproduct generation and facilitating a cleaner reaction profile. As illustrated in the reaction scheme below, the transformation is direct and atom-economical, representing a significant leap forward in process chemistry for sulfur-containing terpenoids.
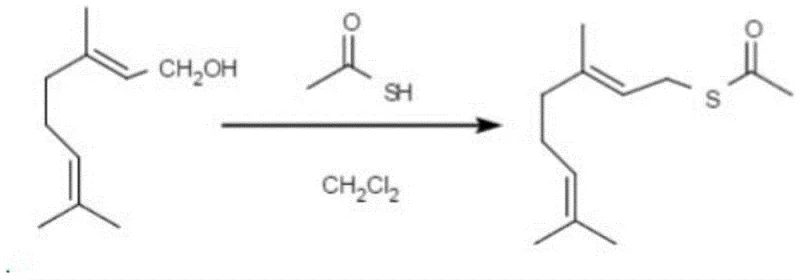
Mechanistic Insights into Pyridine-Catalyzed Esterification
The success of this novel synthesis lies in the precise mechanistic role played by pyridine, which acts as both a base and a nucleophilic catalyst to drive the esterification forward. In this system, pyridine activates the thioacetic acid by forming a reactive acylpyridinium intermediate, which is significantly more electrophilic than the parent acid. This activation lowers the energy barrier for the nucleophilic attack by the hydroxyl group of geraniol, facilitating the formation of the thioester bond under ambient conditions. The choice of dichloromethane as the solvent is equally critical, as it provides excellent solubility for both the lipophilic terpene alcohol and the polar catalyst, ensuring a homogeneous reaction mixture that maximizes molecular collisions. Furthermore, the mild acidity of the byproduct (pyridinium salt) prevents the acid-catalyzed polymerization or rearrangement of the sensitive geraniol backbone, a common degradation pathway in stronger acidic environments. This mechanistic finesse ensures that the structural integrity of the terpene is preserved while efficiently installing the sulfur functionality.
From an impurity control perspective, this mechanism offers distinct advantages over reduction-based routes. By avoiding strong reducing agents like sodium borohydride, the process eliminates the risk of over-reduction or the formation of sulfide byproducts that often contaminate sulfur chemistry. The reaction stoichiometry, typically maintained at a molar ratio of geraniol to thioacetic acid between 1:1 and 1:2, ensures complete conversion of the alcohol while minimizing excess reagent waste. Post-reaction neutralization with dilute hydrochloric acid effectively quenches the pyridine catalyst, allowing for easy separation during the aqueous workup. The subsequent purification via silica gel column chromatography, using a petroleum ether and ethyl acetate gradient, efficiently removes any trace unreacted starting materials or minor side products. This rigorous control over the reaction environment and workup procedure guarantees a final product purity exceeding 98.00%, meeting the stringent specifications required for high-end flavor and fragrance applications.
How to Synthesize Geraniol Thioacetate Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high yield. The process begins with the precise dissolution of geraniol and thioacetic acid in dichloromethane, followed by the controlled addition of pyridine to initiate the catalytic cycle. Maintaining the reaction at room temperature for the specified 4 to 10 hour window is crucial; extending the time beyond this range offers diminishing returns, while shortening it may lead to incomplete conversion. Following the reaction, the quenching step using 5-20% hydrochloric acid must be performed carefully to reach a neutral pH without generating excessive heat that could degrade the product. The extraction and drying phases are standard but vital for removing water and inorganic salts before concentration. For detailed operational parameters, stoichiometric ratios, and purification gradients, please refer to the standardized protocol outlined below.
- Dissolve geraniol and thioacetic acid in dichloromethane with pyridine catalyst and stir at room temperature for 4-10 hours.
- Neutralize the reaction mixture to pH 7 using dilute hydrochloric acid and extract the organic layer with diethyl ether.
- Dry the organic phase, concentrate to obtain crude product, and purify via silica gel column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple chemical yield. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced supply chain resilience. By collapsing a three-step synthesis into a single pot reaction, manufacturers can significantly decrease the turnaround time for production batches, thereby improving responsiveness to market demand fluctuations. The elimination of cryogenic conditions and hazardous reducing agents also lowers the barrier for entry for contract manufacturing organizations, expanding the pool of potential suppliers and reducing dependency on specialized facilities. This democratization of production capability fosters a more competitive sourcing environment, ultimately driving down costs for downstream users in the flavor and pharmaceutical industries.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of expensive reagents and energy-intensive unit operations. Traditional routes require costly reducing agents and strict temperature controls, whereas this method operates at ambient temperature using inexpensive, commodity-grade solvents like dichloromethane and pyridine. The one-step nature of the reaction reduces labor costs, equipment occupancy time, and utility consumption, leading to a substantially lower cost of goods sold (COGS). Furthermore, the high selectivity of the reaction minimizes the loss of valuable raw materials to byproducts, maximizing the atom economy and ensuring that every kilogram of input contributes effectively to the final output. These factors combine to create a highly cost-competitive manufacturing model that allows for aggressive pricing strategies without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of sourcing specialized intermediates or managing hazardous logistics. This synthesis relies on geraniol and thioacetic acid, both of which are widely available bulk chemicals with stable global supply chains. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or utility shortages, as it does not rely on complex refrigeration or high-pressure systems. Additionally, the simplified workup and purification process reduces the lead time from raw material intake to finished goods shipment. This agility enables suppliers to maintain leaner inventory levels while still meeting just-in-time delivery requirements, providing a critical buffer against market volatility and ensuring a steady flow of high-purity intermediates to customers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the linear nature of this esterification makes it inherently scalable from gram to ton quantities. The absence of exothermic hazards or gas evolution simplifies reactor design and safety protocols, facilitating a smoother transition from pilot plant to full commercial production. From an environmental standpoint, the process generates significantly less waste compared to multi-step alternatives, as there are fewer solvent exchanges and purification stages. The use of standard organic solvents allows for efficient recovery and recycling, further reducing the environmental footprint. This alignment with green chemistry principles not only mitigates regulatory risks but also enhances the brand value of the final product for eco-conscious consumers in the food and personal care sectors.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled answers to the most pressing questions regarding the synthesis and application of geraniol thioacetate. These insights are derived directly from the experimental data and technical disclosures within patent CN110156652B, ensuring accuracy and relevance for industrial application. Understanding these details is crucial for making informed decisions about process adoption and supplier qualification. The following section addresses key concerns regarding yield optimization, purity standards, and scalability.
Q: What are the primary advantages of this new synthesis method over traditional citral-based routes?
A: The new method utilizes a direct one-step esterification of geraniol at room temperature, eliminating the harsh conditions, multi-step reductions, and low yields associated with traditional citral-based pathways.
Q: What purity levels can be achieved with this pyridine-catalyzed process?
A: Following silica gel column purification, the process consistently yields geraniol thioacetate with a purity exceeding 98.00%, suitable for high-end flavor and fragrance applications.
Q: Is this process scalable for industrial production of sulfur-containing terpenoids?
A: Yes, the use of readily available raw materials, ambient temperature conditions, and standard solvent systems makes this protocol highly amenable to commercial scale-up and continuous manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Geraniol Thioacetate Supplier
As the global demand for high-quality sulfur-containing terpenoids continues to rise, partnering with a technically proficient manufacturer is paramount for ensuring product consistency and supply security. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in process chemistry to deliver superior intermediates for the flavor, fragrance, and pharmaceutical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of geraniol thioacetate meets or exceeds the >98% purity benchmark established by leading patents.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through our advanced synthetic capabilities. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us be your strategic partner in navigating the complexities of fine chemical sourcing and driving innovation in your product development pipeline.
