Scalable Green Synthesis of Glyceraldehyde Acetonide via C60/ZnO/Se Catalytic Oxidation
The landscape of chiral intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways. Patent CN108752309B introduces a groundbreaking methodology for the synthesis of glyceraldehyde acetonide, a critical chiral building block widely utilized in the pharmaceutical and agrochemical sectors. This innovation leverages a novel fullerene-zinc-selenium (C60/ZnO/Se) composite material as a heterogeneous catalyst, enabling the selective aerobic oxidation of glycerol acetonide. By utilizing molecular oxygen as the terminal oxidant under mild room temperature conditions, this technology effectively circumvents the severe environmental and safety limitations associated with traditional stoichiometric oxidants. For R&D directors and procurement strategists, this represents a pivotal shift towards sustainable chemistry that does not compromise on yield or purity, offering a robust solution for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of glyceraldehyde and its protected derivatives has relied heavily on chemical oxidation methods that are fraught with significant operational and environmental drawbacks. Traditional protocols frequently employ strong oxidizing agents such as hydrogen peroxide, peroxy acids, dilute nitric acid, chromium oxide, activated manganese dioxide, or dimethyl sulfoxide (DMSO). While these reagents can effect the necessary transformation, they introduce substantial risks; for instance, hydrogen peroxide and peroxy acids are inherently unstable and pose explosion hazards during transportation and storage. Furthermore, the use of heavy metal oxidants like chromium oxide generates toxic waste streams that require costly and complex disposal procedures, violating modern green chemistry principles. Alternative routes, such as the multi-step synthesis starting from D-mannitol involving Lewis acid catalysis and periodate cleavage, suffer from high raw material costs and excessive procedural complexity, rendering them economically unviable for large-scale industrial application.
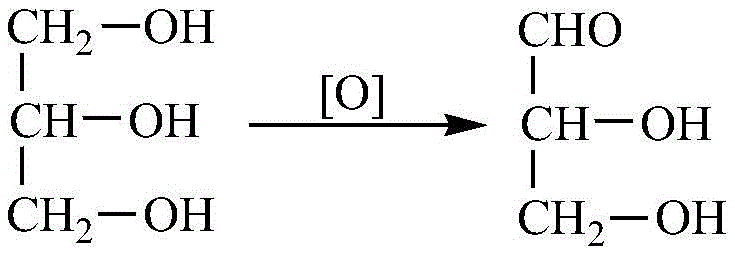
Another conventional pathway involves the indirect electrooxidation of glycerol, which, despite offering simple reaction steps, is plagued by high energy consumption and the necessity for specialized membrane separation technologies. These legacy methods collectively create a bottleneck in the supply chain, characterized by high production costs, safety liabilities, and environmental compliance challenges. The reliance on stoichiometric amounts of hazardous reagents not only inflates the cost of goods sold but also complicates the purification process, often leading to lower overall yields and inconsistent product quality. Consequently, the industry has long sought a catalytic system that could utilize benign oxidants like oxygen while maintaining high selectivity and conversion rates.
The Novel Approach
The technology disclosed in CN108752309B offers a transformative solution by employing a C60/ZnO/Se composite catalyst to drive the oxidation using molecular oxygen. This approach fundamentally alters the economic and environmental profile of the synthesis. Unlike the complex multi-step sequence required for the D-mannitol route, which involves protection, oxidation, and deprotection steps, the new method achieves the transformation in a single catalytic step under ambient conditions. The use of acetone as a solvent and oxygen gas as the oxidant ensures that the only byproduct is water, drastically simplifying the workup procedure. The catalyst can be easily recovered via centrifugation, allowing for potential reuse and further reducing material costs. This streamlined process eliminates the need for hazardous reagent handling and expensive waste treatment, positioning it as a superior alternative for the cost reduction in chiral building block manufacturing.
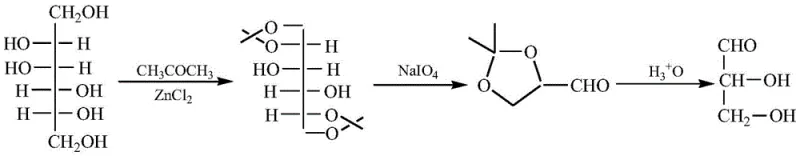
Mechanistic Insights into C60/ZnO/Se Catalytic Oxidation
The core of this technological breakthrough lies in the unique electronic and structural properties of the fullerene-zinc-selenium composite material. The C60 fullerene component acts as an electron acceptor and transporter, facilitating the activation of molecular oxygen on the surface of the zinc oxide and selenium sites. This synergistic interaction creates active oxygen species that are highly selective for the oxidation of primary hydroxyl groups to aldehydes, while leaving secondary hydroxyl groups and acetonide protecting groups intact. The mechanism likely involves the adsorption of the glycerol acetonide substrate onto the catalyst surface, followed by the abstraction of hydrogen atoms from the primary alcohol moiety by the activated oxygen species. This precise selectivity is crucial for preventing over-oxidation to carboxylic acids or the degradation of the sensitive acetonide ring, which are common side reactions in non-selective oxidation processes.
From an impurity control perspective, the heterogeneous nature of the C60/ZnO/Se catalyst provides a distinct advantage over homogeneous systems. Since the catalytic activity is confined to the solid surface, the reaction environment remains free of dissolved metal ions that could catalyze unwanted side reactions or contaminate the final product. The patent data reports HPLC purities exceeding 98.6% for the resulting glyceraldehyde acetonide, demonstrating the exceptional chemoselectivity of this system. Furthermore, the mild reaction conditions (room temperature) minimize thermal degradation pathways that often lead to the formation of polymeric byproducts or racemization of the chiral center. This high level of control over the reaction trajectory ensures a clean impurity profile, which is a critical requirement for regulatory approval in pharmaceutical applications and significantly reduces the burden on downstream purification units.
How to Synthesize Glyceraldehyde Acetonide Efficiently
The synthesis protocol outlined in the patent is remarkably straightforward, designed to facilitate easy adoption in both laboratory and pilot plant settings. The process begins with the dissolution of glycerol acetonide in acetone, followed by the addition of the optimized C60/ZnO/Se composite catalyst. Oxygen gas is then introduced into the reaction mixture at a controlled flow rate, typically between 180 to 200 mL/min, while maintaining the system at room temperature. The reaction proceeds until the starting material is fully consumed, a process that generally takes approximately 20 to 24 hours depending on the specific catalyst loading and batch size. Upon completion, the solid catalyst is removed via simple centrifugation, and the clear supernatant is concentrated under reduced pressure and dried to yield the high-purity product. For detailed standardized operating procedures and safety guidelines, please refer to the technical documentation below.
- Dissolve glycerol acetonide in acetone and add the C60/ZnO/Se composite catalyst.
- Introduce oxygen gas at room temperature and react until the starting material is consumed.
- Remove the catalyst by centrifugation, then concentrate and dry the supernatant to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic oxidation technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The shift from stoichiometric hazardous oxidants to a catalytic aerobic system fundamentally reshapes the cost structure and risk profile of the supply chain. By eliminating the need for expensive and dangerous reagents like chromium oxide or periodic acid, manufacturers can achieve substantial cost savings in raw material procurement and waste disposal. The simplicity of the workup, which requires only filtration and evaporation rather than complex extractions or chromatography, significantly reduces processing time and solvent consumption. This efficiency gain allows for faster turnaround times and increased production throughput, directly addressing the need for reducing lead time for high-purity pharmaceutical intermediates in a competitive market.
- Cost Reduction in Manufacturing: The implementation of the C60/ZnO/Se catalyst drives down manufacturing costs through multiple mechanisms. Firstly, the use of molecular oxygen, which is abundant and inexpensive, replaces costly stoichiometric oxidants, thereby lowering the direct material cost per kilogram of product. Secondly, the heterogeneous nature of the catalyst allows for its recovery and potential reuse, minimizing catalyst consumption expenses over time. Thirdly, the elimination of toxic heavy metals removes the financial burden associated with specialized waste treatment and environmental compliance fees. Finally, the high selectivity of the reaction minimizes the formation of byproducts, leading to higher effective yields and reducing the loss of valuable starting materials, which collectively contributes to a more lean and cost-effective production model.
- Enhanced Supply Chain Reliability: Supply chain stability is often compromised by the availability and regulatory restrictions of hazardous chemicals. By transitioning to a process that relies on oxygen and a stable solid catalyst, manufacturers mitigate the risk of supply disruptions caused by the strict transportation and storage regulations governing strong oxidizers. The robustness of the C60/ZnO/Se composite ensures consistent performance across different batches, reducing the variability that can lead to production delays. Furthermore, the mild reaction conditions reduce the dependency on specialized high-pressure or high-temperature equipment, allowing for greater flexibility in manufacturing site selection and capacity expansion. This resilience ensures a continuous and reliable flow of critical intermediates to downstream customers, safeguarding their own production schedules against upstream volatility.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this green oxidation method is inherently scalable. The exothermicity of the reaction is manageable under the described conditions, reducing the risk of thermal runaway incidents that are common in large-scale oxidations. The absence of toxic effluents aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against evolving compliance standards. The simple unit operations involved—dissolution, gas sparging, filtration, and evaporation—are standard in the fine chemical industry, facilitating seamless technology transfer from pilot to commercial scale. This ease of scale-up enables rapid response to market demand surges without the need for extensive capital investment in new infrastructure, ensuring long-term sustainability and operational agility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this process for integration into their supply chains. Understanding these details is essential for assessing the feasibility of adopting this green synthesis route for the commercial production of chiral intermediates.
Q: What are the advantages of the C60/ZnO/Se catalyst over traditional oxidants?
A: Unlike traditional oxidants such as chromium oxide or dimethyl sulfoxide which pose environmental and safety hazards, the C60/ZnO/Se composite uses molecular oxygen as the oxidant, generating no toxic waste and operating under mild room temperature conditions.
Q: What is the purity level achievable with this catalytic oxidation method?
A: The patent data indicates that the method yields glyceraldehyde acetonide with an HPLC purity exceeding 98.6% after simple concentration and drying, eliminating the need for complex purification steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly suitable for scale-up due to its simple operation, use of inexpensive oxygen gas, mild reaction conditions, and the ease of catalyst recovery via centrifugation, which simplifies downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glyceraldehyde Acetonide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic technologies to deliver superior value to our global partners. With our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we are uniquely positioned to leverage innovations like the C60/ZnO/Se catalytic oxidation method to enhance our portfolio of chiral building blocks. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of glyceraldehyde acetonide meets the exacting standards required for pharmaceutical synthesis. By integrating this green and efficient process, we reinforce our capability to provide a reliable pharmaceutical intermediates supplier service that balances cost, quality, and sustainability.
We invite you to explore how this technological advancement can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our upgraded production capabilities can support your long-term strategic goals in the development of chiral medicines and fine chemicals.
