Scalable Production of High-Purity Deuterated Diazepam for Analytical Standards
Scalable Production of High-Purity Deuterated Diazepam for Analytical Standards
The demand for precise analytical standards in forensic toxicology and pharmaceutical quality control has never been more critical, driving the need for robust synthetic routes for isotopically labeled compounds. Patent CN103204819A introduces a groundbreaking methodology for the preparation of Deuterated Diazepam, a vital internal standard used to ensure accuracy in the detection of benzodiazepines. This technology addresses the historical reliance on imported, exorbitantly priced standards by offering a streamlined, cost-effective domestic production capability. By leveraging a specific H/D exchange mechanism under mild basic conditions, the process achieves exceptional isotopic enrichment without the need for cryogenic temperatures or exotic catalysts. 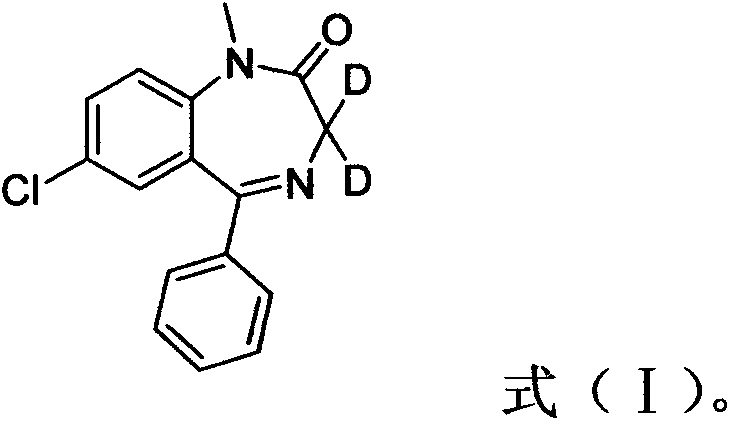 This structural integrity is paramount for mass spectrometry applications where the labeled compound must behave identically to the analyte during extraction and ionization. The innovation lies not just in the molecule itself, but in the accessibility of the reagents, transforming a niche laboratory curiosity into a commercially viable commodity for the global supply chain.
This structural integrity is paramount for mass spectrometry applications where the labeled compound must behave identically to the analyte during extraction and ionization. The innovation lies not just in the molecule itself, but in the accessibility of the reagents, transforming a niche laboratory curiosity into a commercially viable commodity for the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deuterated benzodiazepines has been plagued by significant economic and operational hurdles that hinder widespread adoption in routine analytical laboratories. Traditional protocols often rely on the use of deuterated methanol (CD3OD) in the presence of strong bases such as sodium hydroxide or potassium tert-butoxide to facilitate hydrogen-deuterium exchange. These reagents are not only prohibitively expensive on an industrial scale but also introduce severe safety hazards due to the hygroscopic and corrosive nature of the bases involved. Furthermore, the reaction conditions required for these legacy methods are often harsh, necessitating strict moisture-free environments and extended reaction times that degrade throughput. The purification of the resulting product frequently involves complex workups to remove residual alkali metals and solvent impurities, which can compromise the isotopic purity essential for high-precision mass spectrometry. Consequently, the final cost of goods for these standards remains excessively high, limiting their utility to well-funded research institutions while excluding smaller clinical or forensic labs.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a remarkably simple yet effective system comprising potassium carbonate, deuterated chloroform, and N,N-dimethylformamide (DMF). This approach fundamentally shifts the economic paradigm by replacing costly deuterated alcohols with deuterated chloroform, a reagent that is significantly more affordable and stable. The use of potassium carbonate as a mild base eliminates the need for rigorous anhydrous conditions, allowing the reaction to proceed smoothly even with trace moisture presence, thereby simplifying operational protocols.  By heating the reaction mixture to moderate temperatures ranging from 40°C to 120°C, the process achieves rapid equilibration, drastically reducing the energy consumption and reactor occupancy time compared to room temperature stirrings that require days to complete. The result is a high-yielding transformation that produces the target deuterated species with minimal side reactions, setting a new benchmark for efficiency in the manufacturing of isotopically labeled pharmaceutical intermediates.
By heating the reaction mixture to moderate temperatures ranging from 40°C to 120°C, the process achieves rapid equilibration, drastically reducing the energy consumption and reactor occupancy time compared to room temperature stirrings that require days to complete. The result is a high-yielding transformation that produces the target deuterated species with minimal side reactions, setting a new benchmark for efficiency in the manufacturing of isotopically labeled pharmaceutical intermediates.
Mechanistic Insights into Base-Catalyzed H/D Exchange
The core chemical transformation driving this synthesis is a base-catalyzed hydrogen-deuterium exchange occurring specifically at the C3 methylene position of the 1,4-benzodiazepine ring. In the presence of potassium carbonate, the relatively acidic protons at the C3 position, flanked by both an imine nitrogen and a carbonyl group, are abstracted to form a resonance-stabilized enolate intermediate. This nucleophilic species then attacks the deuterated chloroform (CDCl3), which acts as the deuterium donor, effectively swapping the protium atoms for deuterium. The choice of DMF as the solvent is critical, as its high boiling point allows the reaction to be driven at elevated temperatures up to 120°C, accelerating the kinetics of the exchange without decomposing the sensitive diazepine scaffold. This thermal activation ensures that the equilibrium favors the deuterated product, achieving near-complete substitution at the target site within a short timeframe of approximately 2 hours.
From an impurity control perspective, the mechanism is inherently clean because it avoids the use of strong nucleophiles that could attack the electrophilic centers of the diazepine ring, such as the C2 carbonyl or the C5 phenyl attachment point. The mildness of potassium carbonate ensures that the lactam ring remains intact, preventing hydrolysis or ring-opening degradation pathways that are common with stronger alkalis. Furthermore, the use of ethyl acetate for extraction capitalizes on the differential solubility of the organic product versus the inorganic salts, allowing for a sharp separation of the target molecule from the potassium chloride and excess carbonate byproducts. Subsequent purification via silica gel chromatography using a dichloromethane and methanol gradient further refines the product, removing any trace non-deuterated starting material to ensure the isotopic purity exceeds 99.5%, a specification mandatory for reliable quantitative analysis.
How to Synthesize Deuterated Diazepam Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling, making it accessible for both pilot-scale development and full commercial manufacturing. The process begins with the dissolution of the starting material, 7-chloro-1-methyl-5-phenyl-1,3-dihydro-1,4-benzodiazepin-2-one, in DMF, followed by the sequential addition of the base and deuterium source. Optimization studies indicate that maintaining a specific molar ratio of reagents is crucial; for every 142mg of substrate, approximately 138mg of potassium carbonate and 1mL of deuterated chloroform yield the best results.
- Dissolve 7-chloro-1-methyl-5-phenyl-1,3-dihydro-1,4-benzodiazepin-2-one in N,N-dimethylformamide (DMF) with stirring.
- Add potassium carbonate and deuterated chloroform, then heat the mixture to 120°C for approximately 2 hours.
- Extract the product with ethyl acetate, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process represents a strategic opportunity to optimize the cost structure of analytical standard production while securing a more resilient supply line. The shift away from imported, high-cost deuterated reagents to domestically sourced, commoditized chemicals directly impacts the bottom line by reducing the raw material expenditure significantly. Moreover, the simplified operational requirements mean that production can be executed in standard glass-lined reactors without the need for specialized cryogenic or high-pressure equipment, lowering the barrier to entry for contract manufacturing organizations. This accessibility fosters a competitive supplier landscape, ensuring that buyers are not locked into single-source monopolies for critical quality control materials.
- Cost Reduction in Manufacturing: The elimination of expensive deuterated methanol and strong organic bases like potassium tert-butoxide results in substantial raw material savings. By utilizing deuterated chloroform and inorganic potassium carbonate, the process leverages widely available industrial chemicals that are subject to less price volatility than specialty deuterated solvents. Additionally, the reduced reaction time from days to mere hours decreases utility costs and increases facility throughput, allowing for more batches to be produced within the same operational window. The straightforward workup involving ethyl acetate extraction avoids the need for expensive distillation columns or complex scavenging resins, further streamlining the cost of goods sold.
- Enhanced Supply Chain Reliability: Sourcing reliability is dramatically improved as all key reagents—DMF, potassium carbonate, and ethyl acetate—are bulk commodities with established global supply chains. Unlike proprietary catalysts or custom-synthesized precursors that may face long lead times, these materials can be procured from multiple vendors, mitigating the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, reducing the rate of batch failures and ensuring consistent delivery schedules to downstream analytical laboratories. This stability is crucial for maintaining the continuity of quality control operations in regulated pharmaceutical environments.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from gram to multi-gram scales with consistent yields. The use of ethyl acetate, a greener solvent compared to chlorinated alternatives like dichloromethane for extraction, aligns with modern environmental, health, and safety (EHS) guidelines, simplifying waste disposal and regulatory compliance. The absence of heavy metal catalysts removes the burden of extensive metal clearance testing and specialized waste treatment, reducing the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only lowers compliance costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of deuterated diazepam, derived directly from the experimental data and specifications outlined in the patent documentation. These insights are intended to clarify the feasibility of implementation and the quality attributes of the final product for potential partners and end-users. Understanding these details is essential for integrating this material into existing quality assurance workflows.
Q: What is the primary advantage of this deuteration method over traditional methods?
A: Unlike traditional methods requiring expensive deuterated methanol or strong bases like potassium tert-butoxide, this process utilizes inexpensive deuterated chloroform and mild potassium carbonate, significantly reducing raw material costs while maintaining high yields.
Q: What purity levels can be achieved with this synthesis route?
A: Following the optimized protocol involving ethyl acetate extraction and silica gel chromatography (DCM:MeOH = 100:10), the final deuterated diazepam product achieves a purity exceeding 99.5%, making it suitable for use as a certified analytical reference standard.
Q: Is this process scalable for commercial production of internal standards?
A: Yes, the process avoids harsh conditions and uses common solvents like DMF and ethyl acetate. The simple workup procedure involving liquid-liquid extraction and standard column chromatography facilitates easy scale-up from gram to kilogram quantities without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Diazepam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality internal standards play in ensuring the safety and efficacy of pharmaceutical products worldwide. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify isotopic enrichment and chemical purity for every batch released. Our commitment to excellence ensures that the deuterated diazepam supplied meets the exacting demands of forensic and pharmaceutical analysis.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific analytical needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your supply chain efficiency and reduce overall procurement costs.
