Advanced One-Step Synthesis of Chiral 2-Carbonyl Oxazoline for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral heterocycles, which serve as critical scaffolds in modern drug discovery. Patent CN102643246A introduces a groundbreaking approach for the synthesis of chiral 2-carbonyl oxazoline, specifically targeting the production of (S)-4-isopropyl oxazolinyl-2-ketone. This compound is not merely a chemical curiosity but a vital intermediate utilized in the synthesis of potent cancer therapy drugs, addressing a high-value niche in oncology research. The disclosed method distinguishes itself by employing a one-step reflux reaction involving 7,7,8,8-tetracyanoquinodimethane and L-valinol, catalyzed by zinc chloride in chlorobenzene. This innovation represents a significant leap forward for a reliable pharmaceutical intermediates supplier, as it simplifies what was traditionally a multi-step, labor-intensive process into a streamlined operation. By leveraging the unique reactivity of tetracyanoquinodimethane under Lewis acid conditions, the patent offers a pathway that enhances both atom economy and operational simplicity, crucial factors for modern green chemistry initiatives in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral oxazolidinones and related oxazoline structures has relied on methodologies that often suffer from significant operational drawbacks. Traditional routes frequently necessitate the use of phosgene or its equivalents, which pose severe safety hazards and environmental concerns due to their high toxicity. Furthermore, existing literature, such as the references cited in the background of the patent, indicates that many conventional syntheses require stringent anhydrous conditions, expensive chiral auxiliaries, or complex protection-deprotection sequences. These factors collectively contribute to elevated production costs and extended lead times, creating bottlenecks for cost reduction in API manufacturing. The reliance on multiple isolation steps also increases the risk of yield loss and racemization, compromising the optical purity essential for biological activity. Consequently, procurement managers have long faced challenges in securing high-purity chiral building blocks without incurring prohibitive expenses associated with waste disposal and specialized safety infrastructure.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN102643246A offers a transformative solution by utilizing a tandem reaction sequence initiated by the decomposition of 7,7,8,8-tetracyanoquinodimethane. This novel approach ingeniously bypasses the need for external carbonyl sources like formic acid or phosgene, as the precursor itself generates the necessary reactive species in situ under the influence of zinc chloride. The reaction proceeds in a single pot using chlorobenzene as a solvent, which allows for high-temperature reflux, driving the equilibrium towards the desired cyclic product. This simplification drastically reduces the number of unit operations required, thereby minimizing solvent consumption and labor hours. For supply chain heads, this translates to a more resilient production model where the commercial scale-up of complex pharmaceutical intermediates becomes significantly more feasible. The ability to obtain the target (S)-enantiomer directly from readily available L-valinol ensures that the stereochemical integrity is maintained throughout the process, delivering a product that meets the rigorous quality standards demanded by top-tier pharmaceutical developers.
Mechanistic Insights into ZnCl2-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the intricate mechanistic pathway facilitated by the zinc chloride catalyst. As elucidated in the patent documentation, the reaction initiates with the instability of 7,7,8,8-tetracyanoquinodimethane in the presence of air and the Lewis acid catalyst. This instability triggers a decomposition sequence where cyano groups are hydrolyzed or transformed, ultimately generating formic acid as a transient intermediate. This in situ generation of formic acid is pivotal, as it immediately engages in a condensation reaction with the excess L-valinol present in the reaction matrix. The zinc chloride acts as a powerful Lewis acid, coordinating with the oxygen and nitrogen atoms of the amino alcohol and the formic acid derivative. This coordination activates the carbonyl carbon towards nucleophilic attack by the amine group, forming an amide linkage. Subsequently, the hydroxyl group of the valinol moiety attacks the activated carbonyl, leading to the elimination of two molecules of water and the closure of the five-membered oxazoline ring. This cascade ensures that the chiral center derived from the L-valinol is preserved, resulting in the exclusive formation of the (S)-configured product.
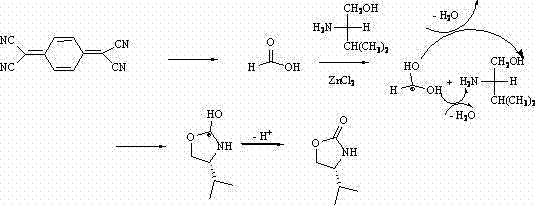
Furthermore, the mechanistic understanding provides critical insights into impurity control, a key concern for R&D directors focused on purity profiles. The use of a large excess of L-valinol (approximately 18 equivalents relative to the TCNQ precursor) drives the reaction kinetics towards the desired product, effectively suppressing side reactions that might arise from incomplete conversion or alternative decomposition pathways of the tetracyanoquinodimethane. The specific choice of chlorobenzene as a solvent is also mechanistically significant; its high boiling point facilitates the removal of water formed during the cyclization via azeotropic distillation or simply by maintaining a high thermal energy state that favors dehydration. This thermodynamic drive ensures that the equilibrium shifts decisively towards the cyclic oxazoline, minimizing the presence of open-chain amide intermediates in the final crude mixture. Consequently, the downstream purification via column chromatography becomes more efficient, yielding a product with high optical purity and minimal structural impurities, which is essential for subsequent coupling reactions in drug synthesis.
How to Synthesize (S)-4-Isopropyl oxazolinyl-2-ketone Efficiently
Implementing this synthesis requires careful attention to stoichiometry and reaction conditions to maximize yield and purity. The protocol dictates the use of anhydrous zinc chloride as the catalyst, which must be handled under inert conditions to prevent premature deactivation by atmospheric moisture. The reaction mixture, comprising the catalyst, solvent, TCNQ, and L-valinol, is subjected to prolonged reflux, typically lasting around 72 hours, to ensure complete transformation of the stable TCNQ precursor. Following the reaction, the workup involves solvent removal and extraction, followed by purification using a specific ratio of petroleum ether and dichloromethane. Detailed standardized synthesis steps see the guide below.
- Charge a reactor with anhydrous zinc chloride (105 mol%) and chlorobenzene solvent, followed by the addition of 7,7,8,8-tetracyanoquinodimethane and excess L-valinol.
- Maintain the reaction mixture at reflux temperature for approximately 72 hours to ensure complete conversion and cyclization.
- Upon completion, remove the solvent under reduced pressure, extract the residue with dichloromethane, and purify the crude product via column chromatography using petroleum ether and dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this zinc chloride-catalyzed route offers substantial strategic advantages for organizations managing the procurement of chiral intermediates. The elimination of hazardous reagents like phosgene not only reduces regulatory compliance burdens but also lowers the capital expenditure required for specialized containment equipment. This shift towards safer chemistry aligns with global sustainability goals, enhancing the corporate social responsibility profile of the supply chain. Moreover, the simplicity of the one-pot design reduces the overall processing time and labor intensity, directly contributing to significant cost savings in manufacturing operations without compromising on the quality of the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the consolidation of multiple synthetic steps into a single operation. By avoiding the purchase and handling of discrete carbonylating agents and reducing the number of isolation stages, manufacturers can achieve a drastic simplification of the production workflow. The use of zinc chloride, a ubiquitous and inexpensive Lewis acid, further minimizes raw material costs compared to processes relying on precious metal catalysts. Additionally, the high atom efficiency derived from the in situ generation of reactants reduces waste generation, leading to lower disposal costs and a more favorable environmental footprint for the facility.
- Enhanced Supply Chain Reliability: Securing a consistent supply of high-quality chiral building blocks is often hindered by the complexity of their synthesis. This method mitigates such risks by utilizing L-valinol, a commercially abundant chiral pool material, as the primary source of stereochemistry. The robustness of the reaction conditions, which tolerate standard industrial heating and stirring capabilities, ensures that production schedules are less susceptible to delays caused by sensitive operational parameters. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing downstream drug developers to maintain their clinical and commercial timelines with greater confidence.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its reliance on standard reflux techniques in chlorobenzene, a solvent widely used in industrial organic synthesis. The absence of cryogenic steps or high-pressure reactors means that existing manufacturing infrastructure can be easily adapted for this chemistry, facilitating rapid technology transfer from the laboratory to the pilot plant and eventually to full commercial production. Furthermore, the reduction in hazardous waste streams simplifies effluent treatment processes, ensuring that the manufacturing site remains compliant with increasingly stringent environmental regulations while maintaining high throughput capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. Understanding these nuances is essential for process chemists and technical buyers evaluating the feasibility of integrating this intermediate into their supply chains. The answers provided are derived directly from the experimental data and mechanistic rationalizations presented in the patent literature.
Q: What is the primary advantage of using TCNQ in this oxazoline synthesis?
A: The use of 7,7,8,8-tetracyanoquinodimethane (TCNQ) serves as a unique precursor that decomposes under Lewis acid catalysis to generate formic acid in situ, which then acts as the carbonyl source for the cyclization, eliminating the need for handling hazardous formic acid directly.
Q: How does the zinc chloride catalyst influence the stereoselectivity?
A: Zinc chloride acts as a potent Lewis acid that coordinates with the amino alcohol and the in situ generated carbonyl species, facilitating a highly stereoselective cyclization that preserves the chirality of the L-valinol starting material to yield the (S)-enantiomer.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes standard reflux conditions in chlorobenzene and avoids cryogenic temperatures or ultra-high pressure, making it inherently scalable for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-Isopropyl oxazolinyl-2-ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trials and market launch. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and enantiomeric excess. Our capability to adapt the zinc chloride-catalyzed synthesis described in CN102643246A allows us to offer a competitive and sustainable source of this valuable oxazoline derivative.
We invite potential partners to engage with our technical procurement team to discuss how this innovative chemistry can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this streamlined manufacturing route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
