Industrial Scale Production of BTG-1675A via Novel Oxidative Cyclization Technology
Industrial Scale Production of BTG-1675A via Novel Oxidative Cyclization Technology
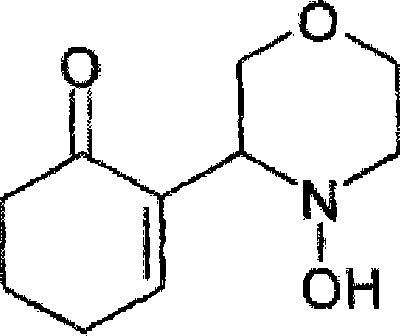
The pharmaceutical industry continuously seeks robust manufacturing pathways for central nervous system (CNS) active ingredients, particularly those addressing depression and anxiety disorders. Patent CN101384564A introduces a transformative methodology for the preparation of 2-(4-hydroxy-3-morpholinyl)-2-cyclohexenone, widely recognized in the sector as BTG-1675A. This compound has been identified in international literature, specifically WO2004/111021, as a potent therapeutic agent for treating anxiety induced by benzodiazepine withdrawal or cessation of substances like nicotine and alcohol. The technical breakthrough detailed in this patent lies in its ability to transition from a low-yield laboratory curiosity to a viable industrial process capable of producing hundreds of grams to multi-ton quantities. By re-engineering the oxidative cyclization steps, the inventors have solved critical bottlenecks related to yield, purity, and operational safety, making it a highly attractive candidate for reliable pharmaceutical intermediate supplier partnerships aiming to secure the supply chain for next-generation CNS medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations disclosed in CN101384564A, the synthesis of BTG-1675A was plagued by severe inefficiencies that rendered it commercially unviable for large-scale adoption. The conventional route, as described in earlier international filings, relied on the in situ generation of nitrones via catalytic oxidation using hydrogen peroxide and sodium tungstate, followed by a prolonged reaction with cyclohexenone. This legacy approach suffered from a catastrophic yield of merely 14%, necessitating complex and costly chromatographic purification steps to isolate the target molecule. Furthermore, the reaction mixture analysis revealed significant issues such as incomplete oxidation of the morpholine precursor, minimal conversion of the cycloaddition intermediate, and the rapid decomposition of starting materials during distillation. These technical failures resulted in substantial waste generation and made the process economically prohibitive for any organization seeking cost reduction in API manufacturing, as the resource intensity far outweighed the value of the final product output.
The Novel Approach
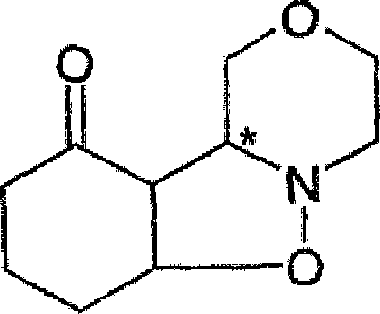
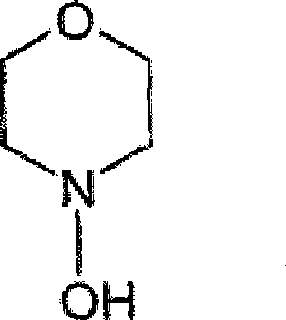
The patented process fundamentally alters the synthetic landscape by introducing a highly efficient two-step sequence that bypasses the pitfalls of the traditional nitrone route. Instead of relying on unstable in situ nitrone generation, the new method reacts N-hydroxymorpholine directly with cyclohexenone in the presence of specific, mild oxidizing agents, most notably azodicarboxylic acid derivatives like azodicarbonamide. This strategic shift allows for the formation of a stable isoxazolidine intermediate, depicted in the structural diagrams, which can be isolated in high purity through simple filtration rather than chromatography. The conversion of this intermediate to the final enone product is then achieved through basic catalysis followed by trituration in aromatic hydrocarbons like toluene. This approach not only boosts the initial yield to over 50% but enables an overall recovery of approximately 90% when unconverted materials are recycled, representing a monumental leap in process efficiency and scalability for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Azodicarbonamide-Mediated Oxidative Cyclization
The core of this technological advancement resides in the precise selection of the oxidizing agent and the mechanistic pathway it facilitates. In the first step, N-hydroxymorpholine undergoes oxidation in the presence of cyclohexenone. While metal oxides like mercuric oxide or activated manganese dioxide were considered, they present significant handling and toxicity challenges; mercuric oxide generates toxic metallic mercury, and manganese dioxide creates difficult-to-filter sludges. The patent highlights the surprising efficacy of azodicarbonamide, which acts as a superior oxidant. Mechanistically, the azodicarbonamide accepts electrons from the hydroxylamine, facilitating the formation of the reactive nitrone species which immediately undergoes a 1,3-dipolar cycloaddition with the electron-deficient double bond of the cyclohexenone. This tandem oxidation-cycloaddition sequence occurs rapidly at temperatures between 40°C and 100°C, preferably around 70°C, completing in less than one hour. The reduced form of the oxidant, hydrazodicarboxamide, precipitates as a practically insoluble solid, allowing for its effortless removal via filtration, thereby driving the equilibrium forward and preventing side reactions that typically degrade product quality.
Impurity control is rigorously managed through the physical properties of the reaction components and the subsequent workup procedure. The second step involves the conversion of the isoxazolidine intermediate into the target 2-(4-hydroxy-3-morpholinyl)-2-cyclohexenone. This transformation is promoted by basic catalysis, where reagents like triethylamine in hot methanol induce an equilibrium favoring the product. However, to achieve pharmaceutical-grade purity, the process utilizes a trituration technique in toluene. Since the isoxazolidine intermediate is significantly more soluble in hot toluene than the final product, repeated grinding and cooling cycles effectively wash away unreacted starting materials and byproducts. This physical purification method eliminates the need for silica gel chromatography, which is a major source of yield loss and solvent waste in fine chemical synthesis. The result is a crystalline solid with a melting point of 127-128°C and purity exceeding 98%, demonstrating that the mechanistic design inherently supports high-purity pharmaceutical intermediate production without compromising throughput.
How to Synthesize 2-(4-hydroxy-3-morpholinyl)-2-cyclohexenone Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the oxidant and the thermal management of the exothermic oxidation step. The process begins with the suspension of crude or purified N-hydroxymorpholine and cyclohexenone in a solvent like ethyl acetate, followed by the controlled addition of azodicarbonamide. The reaction is heated until spontaneous reflux occurs, indicating the onset of the oxidative cycle, and then maintained under reflux for approximately four hours to ensure complete conversion. Following the filtration of the spent oxidant, the intermediate is crystallized from methanol at low temperatures. The final conversion step utilizes a base-catalyzed rearrangement in methanol, followed by the critical toluene trituration to polish the crystal lattice and remove impurities. For detailed operational parameters, safety data, and exact stoichiometric ratios required for GMP compliance, please refer to the standardized synthesis protocol below.
- React N-hydroxymorpholine with cyclohexenone in the presence of azodicarbonamide at 70°C to form the isoxazolidine intermediate.
- Filter the insoluble hydrazodicarboxamide byproduct and isolate the intermediate via crystallization.
- Convert the isoxazolidine to the final enone product using basic catalysis in methanol followed by trituration in toluene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers profound strategic benefits that extend beyond mere technical feasibility. The elimination of toxic heavy metal catalysts such as mercury and the avoidance of complex chromatographic purification significantly streamline the manufacturing workflow. This simplification translates directly into reduced operational expenditures, as the facility requirements for waste treatment and solvent recovery are drastically minimized. Furthermore, the ability to recycle the oxidant byproduct back into its active form through electrochemical or chemical oxidation presents a unique opportunity for circular economy integration within the plant, further enhancing the sustainability profile of the supply chain. These factors collectively contribute to a more resilient and cost-effective sourcing strategy for high-value CNS intermediates.
- Cost Reduction in Manufacturing: The substitution of expensive and hazardous metal oxides with commodity-grade azodicarbonamide results in substantial raw material cost savings. Additionally, the replacement of chromatography with crystallization and filtration reduces solvent consumption and labor hours associated with column packing and fraction collection. The high yield of the process, reaching up to 90% with recycling, ensures that the cost per kilogram of the active intermediate is significantly lower than legacy methods, providing a competitive edge in pricing negotiations for long-term contracts.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a range of temperatures and do not require cryogenic cooling or ultra-high vacuum, makes the process highly scalable from pilot plants to multi-ton reactors. The use of stable, commercially available starting materials like morpholine and cyclohexenone mitigates the risk of raw material shortages. Moreover, the simplified purification train reduces the likelihood of batch failures due to equipment fouling or column channeling, ensuring consistent on-time delivery and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process is markedly superior. The absence of mercury eliminates the need for specialized hazardous waste disposal protocols, while the solid nature of the spent oxidant simplifies containment and handling. The ability to recover and reuse solvents like toluene and ethyl acetate, combined with the high atom economy of the cycloaddition step, aligns with modern green chemistry principles. This compliance facilitates easier regulatory approval in stringent markets and reduces the carbon footprint of the manufacturing operation, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and advantageous effects claimed in the patent documentation, providing clarity on yield expectations, safety profiles, and purification strategies. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What is the primary advantage of using azodicarbonamide over metal oxides?
A: Azodicarbonamide avoids the toxicity issues associated with mercury and manganese oxides, and its reduced form is a solid that can be easily filtered and potentially recycled, significantly simplifying downstream processing.
Q: How does this process improve yield compared to previous methods?
A: While prior art methods yielded only about 14% requiring complex chromatography, this novel route achieves yields greater than 50% initially, and up to 90% when accounting for recovered unconverted materials, without the need for chromatographic purification.
Q: Is the N-hydroxymorpholine starting material stable for large-scale use?
A: Yes, the patent describes a robust method for generating N-hydroxymorpholine in situ or using purified forms, ensuring high purity (up to 95% yield in precursor synthesis) which is critical for consistent final product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-hydroxy-3-morpholinyl)-2-cyclohexenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality intermediates for the development of life-saving CNS therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of BTG-1675A meets the exacting standards required for pharmaceutical applications. Our infrastructure is designed to handle complex oxidative chemistries safely, leveraging the insights from patents like CN101384564A to optimize yield and minimize environmental impact.
We invite global partners to collaborate with us to leverage this advanced manufacturing technology for your pipeline projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
