Advanced Solvent-Free Synthesis of 3,7-Dimethyl-3-Acetylmercapto-6-Octenal for Commercial Scale-Up
The chemical landscape for high-value terpene derivatives is undergoing a significant transformation, driven by the urgent need for greener and more cost-effective manufacturing processes. A pivotal development in this sector is detailed in patent CN112142632A, which discloses a novel preparation method for 3,7-dimethyl-3-acetylmercapto-6-octenal, a critical intermediate widely utilized in the flavor, fragrance, and pharmaceutical industries. This innovation addresses long-standing inefficiencies in the synthesis of C13-norisoprenoid derivatives by transitioning from traditional solvent-heavy protocols to a streamlined, solvent-free approach. By leveraging the inherent reactivity of citral and thioacetic acid under mild thermal conditions, this technology not only simplifies the operational workflow but also drastically reduces the environmental footprint associated with volatile organic compound (VOC) emissions. For R&D directors and procurement strategists, this represents a tangible opportunity to optimize supply chains for complex sulfur-containing terpenes, ensuring both regulatory compliance and economic viability in large-scale production scenarios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this technological breakthrough, the synthesis of 3,7-dimethyl-3-acetylmercapto-6-octenal was fraught with significant operational and environmental challenges that hindered efficient commercialization. Historical literature, such as the work by Canon et al. (2015), relied heavily on the use of pyridine as a base catalyst and required substantial volumes of organic solvents to facilitate the reaction. These conventional methods necessitated cryogenic reaction conditions, typically maintained at 0°C, which imposed a heavy energy burden on manufacturing facilities and required specialized cooling infrastructure. Furthermore, the reliance on pyridine introduced severe toxicity concerns, complicating waste disposal and worker safety protocols, while the extensive use of solvents necessitated complex and energy-intensive recovery systems. The combination of harsh conditions, toxic reagents, and multi-step workup procedures often resulted in lower overall yields and a cumbersome purification process, making the cost of goods sold (COGS) prohibitively high for many downstream applications in the fine chemical sector.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent data utilizes a direct, one-step solvent-free reaction that fundamentally redefines the process economics of this valuable intermediate. By eliminating the need for external solvents and toxic amine bases, the new methodology allows the reaction between citral and thioacetic acid to proceed smoothly at a mild temperature of 50°C. This shift not only removes the capital and operational expenditures associated with solvent storage, recovery, and disposal but also significantly enhances the safety profile of the manufacturing process. The absence of a solvent medium increases the effective concentration of reactants, potentially accelerating reaction kinetics and improving space-time yield. Moreover, the simplified workflow reduces the number of unit operations required, minimizing material handling and the risk of product loss during transfer, thereby delivering a robust and scalable solution that aligns perfectly with modern principles of green chemistry and sustainable manufacturing.
Mechanistic Insights into Solvent-Free 1,4-Addition Reaction
The core of this synthetic advancement lies in the precise control of a 1,4-addition reaction, also known as a Michael-type addition, between the alpha,beta-unsaturated aldehyde moiety of citral and the nucleophilic sulfur atom of thioacetic acid. In the absence of a solvent, the interaction between these two reactants is governed by their intrinsic electronic properties and the thermal energy provided at 50°C. The sulfur atom in thioacetic acid acts as a soft nucleophile, selectively attacking the beta-carbon of the conjugated system in citral rather than the carbonyl carbon, which ensures high regioselectivity for the desired 3-substituted product. This selectivity is crucial for minimizing the formation of 1,2-addition by-products or polymerization side reactions that often plague aldehyde chemistry. The solvent-free environment likely creates a unique micro-environment where hydrogen bonding and dipole interactions between the reactants are maximized, facilitating the transition state without the interference of solvating molecules that might stabilize competing pathways.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity flavor and fragrance intermediates. The mild thermal conditions prevent the degradation of the sensitive aldehyde group and the isomerization of the double bonds, which are common issues in harsher synthetic routes. The patent data indicates that the reaction yields a crude product with a single dominant species, significantly simplifying the downstream purification burden. By avoiding strong bases like pyridine, the process mitigates the risk of base-catalyzed aldol condensations or racemization, ensuring that the stereochemical integrity of the chiral centers in the citral backbone is preserved to the greatest extent possible. This mechanistic clarity provides R&D teams with the confidence that the process is robust and reproducible, capable of consistently delivering material that meets stringent quality specifications for sensitive applications in food and pharma.
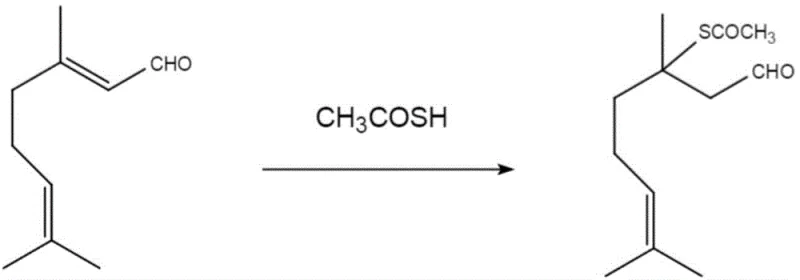
How to Synthesize 3,7-Dimethyl-3-Acetylmercapto-6-Octenal Efficiently
The implementation of this synthesis route is designed for seamless integration into existing chemical manufacturing infrastructure, requiring minimal modification to standard reactor setups. The process begins with the precise metering of citral and thioacetic acid, followed by a controlled heating phase that drives the conversion to completion without the need for inert atmosphere protection in many cases. The simplicity of the procedure belies its sophistication, as the careful optimization of molar ratios and temperature ensures that the reaction proceeds with high efficiency. For technical teams looking to adopt this methodology, the following guide outlines the critical operational parameters derived directly from the patent examples, serving as a foundational framework for process validation and scale-up activities.
- Mix citral and thioacetic acid in a molar ratio ranging from 1: 1 to 1:4 under solvent-free conditions.
- Stir the reaction mixture at a controlled temperature between 40°C and 60°C, optimally at 50°C, for 4 to 10 hours.
- Purify the resulting crude product via silica gel column chromatography using a petroleum ether and ethyl acetate eluent system to obtain the final pure compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solvent-free technology translates into immediate and tangible strategic benefits that extend far beyond simple chemical conversion. The elimination of organic solvents removes a major variable from the cost equation, eradicating the expenses associated with purchasing, storing, and recovering vast quantities of volatile liquids. This reduction in material complexity directly lowers the operational overhead, allowing for a more competitive pricing structure for the final synthetic flavor intermediate. Furthermore, the use of readily available and inexpensive raw materials like citral and thioacetic acid ensures a stable supply base, reducing the risk of bottlenecks that can occur with specialized or scarce reagents. The streamlined nature of the process also implies a shorter manufacturing cycle time, enabling faster response to market demand fluctuations and improved inventory turnover rates.
- Cost Reduction in Manufacturing: The most significant economic driver of this new method is the complete removal of solvent-related costs, which traditionally account for a substantial portion of variable manufacturing expenses in fine chemical synthesis. By operating under solvent-free conditions, the process eliminates the need for expensive solvent recovery distillation columns and the associated energy consumption for heating and cooling large liquid volumes. Additionally, the avoidance of pyridine, a costly and hazardous reagent, further reduces the raw material bill of materials (BOM). The simplified workup procedure, which requires fewer separation steps, reduces labor costs and minimizes product loss during isolation, collectively contributing to a substantially lower cost of goods sold without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on commodity chemicals that are produced on a massive global scale. Citral is a ubiquitous terpene derived from natural essential oils or petrochemical sources, ensuring a consistent and reliable feedstock supply that is less susceptible to geopolitical disruptions. Thioacetic acid is similarly a standard industrial chemical with a mature supply network. This contrasts sharply with methods relying on exotic catalysts or custom-synthesized reagents that may have long lead times. The robustness of the reaction conditions, which do not require cryogenic cooling or strictly anhydrous environments, also reduces the dependency on specialized utility infrastructure, making the production feasible in a wider range of manufacturing locations globally.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is inherently safer and more straightforward due to the absence of flammable solvent vapors, which significantly lowers the explosion hazard rating of the facility. This safety profile facilitates easier permitting and regulatory approval, accelerating the time-to-market for new capacity. From an environmental standpoint, the process aligns with increasingly stringent global regulations regarding VOC emissions and hazardous waste disposal. The reduction in waste generation simplifies effluent treatment requirements and lowers the carbon footprint of the manufacturing operation. This sustainability advantage is not merely a compliance checkbox but a powerful marketing asset for downstream customers in the food and pharmaceutical sectors who are under pressure to source ingredients from green and sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel intermediate. These insights are derived from the specific technical disclosures within the patent documentation, providing clarity on process capabilities and product specifications. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this material into their existing product portfolios or manufacturing lines.
Q: What are the primary advantages of the solvent-free method over traditional synthesis?
A: The solvent-free method eliminates the need for toxic organic solvents and pyridine bases required in conventional routes, significantly reducing environmental impact and operational costs while simplifying post-reaction processing.
Q: What is the optimal reaction temperature for this synthesis?
A: The patent specifies an optimal reaction temperature of 50°C, which avoids the harsh cryogenic conditions (0°C) of previous methods, thereby enhancing energy efficiency and process safety.
Q: How is high purity achieved in the final product?
A: High purity exceeding 98% is achieved through a straightforward silica gel column chromatography purification step using a specific ratio of petroleum ether to ethyl acetate, effectively removing unreacted starting materials and by-products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,7-Dimethyl-3-Acetylmercapto-6-Octenal Supplier
As the global demand for high-quality sulfur-containing terpenes continues to rise, partnering with a technically proficient manufacturer is critical for securing a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in process chemistry to deliver superior intermediates like 3,7-dimethyl-3-acetylmercapto-6-octenal. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required for flavor, fragrance, and pharmaceutical applications.
We invite you to collaborate with us to explore how this advanced solvent-free technology can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality targets. Please contact us today to request specific COA data, discuss route feasibility assessments, or initiate a dialogue about how we can support your long-term strategic goals with reliable, high-performance chemical solutions.
