Advanced Manufacturing of 2,5-Dihalogeno Benzoic Acid via Efficient Acid Hydrolysis Technology
The chemical industry is constantly seeking more efficient pathways for synthesizing critical building blocks, and patent CN1740135A presents a significant advancement in the production of 2,5-dihalogeno benzoic acid derivatives. This intellectual property discloses a novel preparation process that shifts away from traditional oxidative methods, instead utilizing the direct acid-catalyzed hydrolysis of 2,5-dihalogeno trifluoro toluene. For R&D directors and procurement specialists in the fine chemical sector, this represents a pivotal opportunity to optimize supply chains for pharmaceutical and agrochemical intermediates. The technology leverages the reactivity of the trifluoromethyl group under strong acidic conditions, specifically employing oleum, to achieve high yields with a drastically simplified operational footprint. By adopting this methodology, manufacturers can bypass the complexities associated with composite catalysts, thereby enhancing both the economic viability and the technical robustness of producing these essential halogenated aromatic acids.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenyl-dihalide formic acids has relied heavily on the air oxidation of dihalo toluene derivatives, a process fraught with inherent inefficiencies and operational challenges. Traditional routes typically necessitate the use of complex composite catalysts to facilitate the oxidation, which not only increases the raw material costs but also introduces significant complications in downstream purification. The presence of heavy metal catalysts often requires extensive removal steps to meet the stringent purity specifications demanded by the pharmaceutical industry, leading to increased waste generation and longer processing times. Furthermore, air oxidation processes can be difficult to control precisely, often resulting in variable yields and the formation of unwanted byproducts that compromise the overall quality of the final intermediate. These factors collectively contribute to a higher production cost and a less reliable supply chain for critical dihalogenated benzoic acid precursors.
The Novel Approach
In stark contrast to the oxidative limitations, the novel approach detailed in the patent utilizes a direct hydrolysis strategy that transforms 2,5-dihalogeno trifluoro toluene into the target benzoic acid with remarkable efficiency. This method capitalizes on the susceptibility of the trifluoromethyl group to hydrolysis in the presence of strong acids, specifically sulfuric acid containing 20% SO3, known as oleum. By operating at moderate temperatures ranging from 50 to 100 degrees Celsius, the process avoids the extreme conditions often required for oxidation, thereby reducing energy consumption and equipment stress. The simplicity of this one-step synthesis eliminates the need for expensive catalytic systems and allows for a straightforward workup procedure involving ice quenching and filtration. This technological shift not only lowers the barrier to entry for manufacturing but also ensures a more consistent product profile, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Acid-Catalyzed Hydrolysis of Trifluoromethyl Groups
The core of this innovative synthesis lies in the mechanistic pathway of acid-catalyzed hydrolysis, where the electron-withdrawing nature of the halogen substituents plays a crucial role in facilitating the reaction. Under the influence of strong acidic media like oleum, the trifluoromethyl group undergoes hydration, eventually cleaving the carbon-fluorine bonds to form the carboxylic acid functionality. The use of oleum, rather than dilute sulfuric acid, provides a highly dehydrating environment that drives the equilibrium towards the formation of the acid while minimizing side reactions. This specific choice of reagent ensures that the reaction proceeds smoothly at relatively low temperatures, typically between 55 and 65 degrees Celsius, which is vital for maintaining the integrity of the halogen substituents on the aromatic ring. Understanding this mechanism allows process chemists to fine-tune reaction parameters to maximize conversion rates while minimizing the degradation of sensitive functional groups.
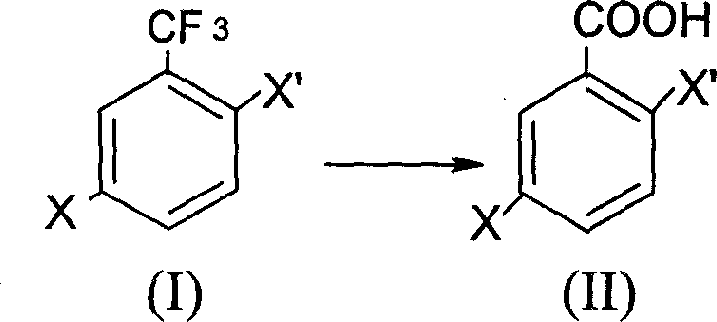
Impurity control is another critical aspect where this mechanistic understanding translates into commercial value, particularly regarding the removal of unreacted starting materials and acidic residues. The patent specifies a purification protocol involving recrystallization from organic solvents such as toluene, which is highly effective in isolating the target benzoic acid from the reaction matrix. The solubility differences between the halogenated benzoic acid and potential impurities in hot versus cold toluene allow for the growth of high-quality crystals with purity levels exceeding 99 percent. This level of purity is essential for downstream applications in drug synthesis, where trace impurities can affect reaction kinetics or final product safety. By integrating this recrystallization step, the process ensures that the final intermediate meets the rigorous quality standards required by global regulatory bodies, thereby reducing the risk of batch rejection and ensuring supply continuity.
How to Synthesize 2-Chloro-5-bromo-benzoic Acid Efficiently
To implement this synthesis effectively, operators must adhere to precise procedural guidelines that balance reaction kinetics with safety considerations, particularly when handling corrosive reagents like oleum. The process begins with the careful charging of 2-chloro-5-bromobenzotrifluoride and oleum into a reactor equipped with efficient agitation and temperature control systems to manage the exothermic nature of the mixing. Maintaining the reaction temperature within the optimal window of 58 to 65 degrees Celsius for a duration of approximately 12 hours is critical to ensuring complete conversion without promoting decomposition. Following the reaction, the mixture is cooled and quenched into trash ice to precipitate the crude product, which is then isolated via filtration. The detailed standardized synthetic steps for scaling this process are outlined in the guide below.
- Charge 2,5-dihalogeno trifluoro toluene and oleum (containing 20% SO3) into a reactor equipped with agitation and temperature control.
- Heat the reaction mixture to a controlled temperature range of 55-65°C and maintain stirring for approximately 12 hours to ensure complete hydrolysis.
- Quench the reaction by pouring into crushed ice, filter the crude precipitate, and purify via recrystallization using toluene to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrolysis-based technology offers substantial strategic benefits that extend beyond simple yield improvements. By eliminating the dependency on complex composite catalysts and high-pressure oxidation equipment, manufacturers can significantly reduce capital expenditure and operational overheads. The use of widely available raw materials, such as substituted trifluorotoluenes and sulfuric acid derivatives, ensures a stable supply base that is less susceptible to market volatility compared to specialized catalytic systems. Furthermore, the simplified workflow reduces the number of unit operations required, which directly correlates to shorter production cycles and faster time-to-market for downstream customers. This operational efficiency translates into a more resilient supply chain capable of meeting fluctuating demand without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated removal processes results in a drastic simplification of the production cost structure. Without the need for catalyst recovery or heavy metal scavenging steps, the overall consumption of auxiliary chemicals is reduced, leading to substantial cost savings in raw material procurement. Additionally, the moderate reaction temperatures lower energy consumption requirements for heating and cooling, further enhancing the economic profile of the process. These cumulative efficiencies allow suppliers to offer more competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like oleum and trifluorotoluenes mitigates the risk of supply disruptions often associated with proprietary or scarce catalytic reagents. Since the process does not require specialized high-pressure reactors typically used for air oxidation, it can be executed in standard glass-lined or stainless steel facilities, increasing the pool of qualified contract manufacturing organizations. This flexibility ensures that production can be scaled up or shifted between sites with minimal friction, guaranteeing continuous availability of critical intermediates for global clients. Consequently, partners can rely on a more robust and predictable supply network for their long-term manufacturing needs.
- Scalability and Environmental Compliance: The straightforward nature of the hydrolysis reaction facilitates easy scale-up from laboratory benchtop to multi-ton commercial production without significant re-engineering of the process. The waste stream primarily consists of diluted sulfuric acid, which is easier to treat and neutralize compared to the complex heavy metal waste generated by oxidative methods. This alignment with greener chemistry principles reduces the environmental footprint of the manufacturing process and simplifies compliance with increasingly stringent environmental regulations. By adopting this cleaner technology, companies can enhance their sustainability credentials while avoiding the costly remediation efforts associated with traditional catalytic processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrolysis technology, derived directly from the patent specifications and industry best practices. These insights are designed to clarify the operational feasibility and quality assurances associated with the production of 2,5-dihalogeno benzoic acid derivatives. Understanding these details is crucial for stakeholders evaluating the integration of this method into their existing manufacturing portfolios. The answers reflect the specific advantages of the oleum-mediated pathway over conventional oxidation techniques.
Q: What are the primary advantages of this hydrolysis method over traditional air oxidation?
A: Unlike traditional air oxidation which requires complex composite catalysts and harsh conditions, this hydrolysis method utilizes readily available oleum, significantly simplifying the process flow and reducing equipment corrosion risks associated with heavy metal catalysts.
Q: How is high purity (>99%) achieved in the final product?
A: High purity is secured through a robust workup procedure involving precipitation in ice water followed by recrystallization in organic solvents like toluene, which effectively removes unreacted starting materials and acidic byproducts.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process operates at moderate temperatures (50-100°C) and uses standard acid-handling infrastructure, making it highly scalable for industrial manufacturing without requiring specialized high-pressure oxidation reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Dihalogeno Benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this acid hydrolysis technology in delivering high-value intermediates to the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from development to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2,5-dihalogeno benzoic acid meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging our technical expertise to optimize this process for your specific needs, ensuring both quality and consistency.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Let us collaborate to drive efficiency and innovation in your chemical manufacturing operations.
