Scalable One-Step Reduction Route for High-Purity Benzimidazole Drug Intermediates
Scalable One-Step Reduction Route for High-Purity Benzimidazole Drug Intermediates
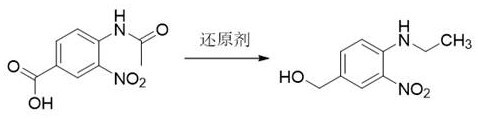
The pharmaceutical industry continuously seeks robust and cost-effective pathways for synthesizing critical heterocyclic intermediates, particularly those serving as precursors for benzimidazole-based therapeutics. Patent CN116217416A introduces a transformative preparation method for 3-nitro-4-ethylaminobenzyl alcohol, a pivotal building block in the synthesis of proton pump inhibitors and anthelmintic agents. This technology addresses long-standing challenges in the field by replacing harsh, energy-intensive protocols with a streamlined one-step reduction strategy. By utilizing 3-nitro-4-acetamidobenzoic acid as a versatile starting material, the process achieves simultaneous conversion of both carboxylic acid and acetamido functional groups under remarkably mild conditions. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates while drastically reducing dependency on specialized microwave equipment and expensive fluorinated reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-nitro-4-ethylaminobenzyl alcohol has relied on nucleophilic substitution reactions involving 3-nitro-4-fluorobenzyl alcohol and ethylamine. As detailed in the background art, this traditional route necessitates the use of excessive diisopropylethylamine as a base and requires microwave irradiation at elevated temperatures of 120°C for extended periods, typically around 3 hours. These harsh reaction conditions not only demand specialized and costly reactor equipment capable of sustaining high pressure and temperature but also pose significant safety risks during scale-up. Furthermore, the starting material, 3-nitro-4-fluorobenzyl alcohol, is inherently more expensive and less commercially available than simple benzoic acid derivatives, creating a bottleneck in cost reduction in API manufacturing. The reliance on microwave technology also limits the batch size, making continuous large-scale production difficult and economically unfeasible for bulk pharmaceutical ingredient suppliers.
The Novel Approach
In stark contrast, the innovative methodology disclosed in the patent leverages a direct reduction strategy that converts 3-nitro-4-acetamidobenzoic acid directly into the target alcohol in a single operational step. This approach utilizes powerful reducing agents such as borane, diisobutyl aluminum hydride, or red aluminum to effect the transformation of the carboxylic acid moiety to a primary alcohol and the acetamido group to an ethylamino group simultaneously. The reaction proceeds efficiently in common organic solvents like tetrahydrofuran or dichloromethane at temperatures ranging from 0°C to 40°C, completely eliminating the need for high-energy microwave inputs. This shift not only simplifies the equipment requirements to standard stainless steel reactors but also significantly enhances the safety profile of the manufacturing process. By adopting this route, manufacturers can achieve higher product yields with reduced raw material costs, establishing a more reliable pharmaceutical intermediate supplier framework for global markets.
Mechanistic Insights into Borane-Mediated Dual Functional Group Reduction
The core chemical innovation lies in the selective yet comprehensive reduction capability of borane complexes and related hydride sources towards multifunctional substrates. When 3-nitro-4-acetamidobenzoic acid is treated with borane-tetrahydrofuran complex, the boron atom acts as a Lewis acid, coordinating with the carbonyl oxygen of the carboxylic acid to facilitate hydride transfer, ultimately reducing the acid to a primary alcohol. Concurrently, the acetamido group undergoes deoxygenation and alkylation; the carbonyl oxygen of the amide is removed, and the acetyl methyl group is retained as part of the ethyl chain on the nitrogen, effectively converting the acetamido group (-NHCOCH3) into an ethylamino group (-NHCH2CH3). This dual transformation is highly sensitive to stoichiometry and temperature control, with the patent specifying a molar ratio of substrate to borane between 1:2.0 and 3.0 to ensure complete conversion without over-reduction of the nitro group, which remains intact throughout the process. The preservation of the nitro group is critical, as it serves as a key handle for subsequent cyclization reactions to form the benzimidazole core.
Impurity control in this mechanism is governed by the precise management of reaction exotherms and quenching protocols. The addition of the reducing agent is performed at low temperatures (0-10°C) to prevent runaway reactions that could lead to the reduction of the aromatic nitro group to an amine, a major impurity that would complicate downstream purification. Furthermore, the workup procedure involves careful quenching with methanol followed by dispersion in alkaline aqueous solutions, which helps to decompose boron-containing byproducts and facilitates the extraction of the neutral organic product. This meticulous control over the reaction environment ensures that the final crude product contains minimal amounts of de-nitrated byproducts or partially reduced intermediates, thereby streamlining the crystallization process and delivering a product with stringent purity specifications required for GMP manufacturing environments.
How to Synthesize 3-Nitro-4-Ethylaminobenzyl Alcohol Efficiently
To implement this synthesis effectively, operators must adhere to strict anhydrous conditions and temperature gradients to maximize yield and safety. The process begins with the dissolution of the starting acid in dry tetrahydrofuran, followed by the controlled addition of the borane complex while maintaining the internal temperature below 10°C to manage the initial exotherm. Once the addition is complete, the reaction mixture is allowed to warm gradually to ambient or slightly elevated temperatures (20-40°C) to drive the reduction to completion over a period of 2 to 5 hours, monitored via thin-layer chromatography. The detailed standardized synthetic steps, including specific quenching agents, extraction solvents, and crystallization parameters for various reducing agents, are outlined in the technical guide below to ensure reproducibility across different production scales.
- Dissolve 3-nitro-4-acetamidobenzoic acid in anhydrous tetrahydrofuran and cool the solution to 0-10°C under inert atmosphere.
- Slowly add the borane-tetrahydrofuran complex solution while maintaining the temperature between 0-10°C, ensuring a molar ratio of 1: 2.0 to 3.0.
- Warm the reaction mixture to 20-40°C and stir for 2-5 hours until TLC indicates completion, then quench carefully with methanol and alkaline solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented reduction technology offers substantial strategic benefits beyond mere technical feasibility. The transition from microwave-assisted nucleophilic substitution to a conventional thermal reduction process fundamentally alters the cost structure of producing this key intermediate. By removing the dependency on specialized microwave reactors and expensive fluorinated starting materials, the overall capital expenditure and operating costs are significantly reduced. This process optimization allows for the utilization of standard chemical infrastructure found in most fine chemical plants, thereby enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates. The ability to source raw materials like 3-nitro-4-acetamidobenzoic acid from a broader range of suppliers further mitigates the risk of supply disruptions, ensuring a steady flow of materials for downstream drug synthesis.
- Cost Reduction in Manufacturing: The elimination of high-cost raw materials such as 3-nitro-4-fluorobenzyl alcohol and the removal of energy-intensive microwave steps result in a drastic simplification of the cost model. The use of commodity reducing agents and common solvents like THF or toluene means that variable costs are kept low, allowing for competitive pricing in the global market. Additionally, the one-step nature of the reaction reduces labor hours and utility consumption associated with multi-step sequences or prolonged heating cycles, contributing to substantial cost savings in the overall manufacturing budget without compromising on quality.
- Enhanced Supply Chain Reliability: Sourcing 3-nitro-4-acetamidobenzoic acid is generally more straightforward and stable compared to specialized fluorinated benzyl alcohols, which may have limited suppliers. This abundance of starting material ensures that production schedules can be maintained consistently, even during periods of raw material volatility. Furthermore, the mild reaction conditions reduce the likelihood of equipment failure or safety incidents that could halt production, thereby guaranteeing a more predictable and reliable delivery timeline for customers relying on this intermediate for their own API production schedules.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory benchtop to multi-ton commercial production because it avoids the physical limitations of microwave penetration depth. The reaction generates minimal waste, and the absence of heavy metal catalysts simplifies wastewater treatment and disposal, aligning with increasingly strict environmental regulations. This green chemistry profile not only reduces the environmental footprint but also lowers the costs associated with waste management and regulatory compliance, making it an attractive option for sustainable chemical manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this reduction technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits and chemical nuances for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing production lines or for initiating new development projects focused on benzimidazole derivatives.
Q: What are the primary advantages of this new reduction method over conventional microwave synthesis?
A: The novel method eliminates the need for harsh microwave conditions at 120°C and expensive fluorinated starting materials. Instead, it utilizes mild temperatures (20-40°C) and readily available 3-nitro-4-acetamidobenzoic acid, significantly lowering raw material costs and equipment requirements.
Q: Which reducing agents are compatible with this synthesis route?
A: The patent discloses versatility in reducing agents, including borane (BH3), diisobutyl aluminum hydride (DIBAL-H), red aluminum (Red-Al), or combinations of alkali metal hydrides (LiAlH4, NaBH4) with boron trifluoride diethyl etherate.
Q: How does this process impact impurity control in the final API intermediate?
A: By avoiding high-temperature microwave irradiation which can promote side reactions, this mild reduction pathway minimizes thermal degradation products. The specific workup procedures involving alkaline dispersion and crystallization further ensure high purity suitable for downstream benzimidazole condensation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Nitro-4-Ethylaminobenzyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the broader pharmaceutical value chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless and robust. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of 3-nitro-4-ethylaminobenzyl alcohol meets the highest standards required for drug substance manufacturing. Our facility is equipped to handle the specific solvent systems and reducing agents described in this patent, guaranteeing consistent quality and supply continuity for our global clientele.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and drive down your overall production costs effectively.
