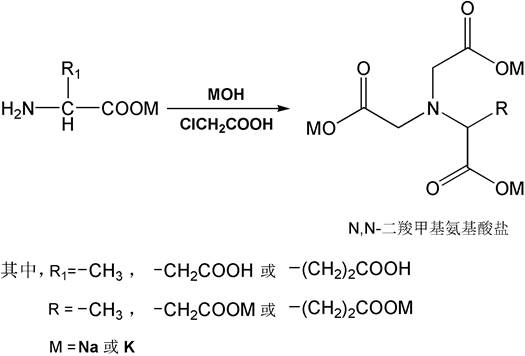
Scaling Green Chelating Agents: Continuous Production of N,N-Dicarboxymethyl Amino Acid Salts
The global demand for sustainable and biodegradable chemical solutions has intensified the search for alternatives to traditional chelating agents like EDTA and NTA. Patent CN110563596B introduces a groundbreaking continuous production method for synthesizing N,N-dicarboxymethyl amino acid salts, such as GLDA and MGDA, which serve as superior green chelating agents. This technology leverages a sophisticated two-stage liquid-liquid static mixing system followed by a continuous reactor heating process, fundamentally shifting the paradigm from inefficient batch processing to high-efficiency continuous manufacturing. By utilizing amino acids, liquid caustic soda, and chloroacetic acid as primary raw materials, this method not only stabilizes product quality but also drastically enhances equipment utilization rates. For R&D directors and procurement specialists seeking reliable green chelating agent supplier partnerships, understanding the mechanistic advantages of this continuous flow chemistry is critical for securing long-term supply chain resilience and cost competitiveness in the fine chemical intermediates market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis methods for N,N-dicarboxymethyl amino acid salts predominantly rely on intermittent kettle-type reactors, which inherently suffer from significant operational inefficiencies and quality inconsistencies. In these batch processes, the material transfer periods are excessively long, leading to thermal gradients that compromise reaction uniformity and result in unstable quality among different production batches. Furthermore, the low equipment utilization rate associated with stop-start batch operations drives up the overall production cost, making it difficult to achieve the economies of scale required for competitive cost reduction in surfactant manufacturing. The prior art also highlights issues with high impurity content and the use of toxic raw materials like hydrocyanic acid in older methods, which pose severe safety risks and generate substantial wastewater containing cyanogen residues. These legacy limitations create bottlenecks for supply chain heads who require consistent, high-volume output without the regulatory burdens associated with hazardous waste disposal and corrosion-prone equipment.
The Novel Approach
The innovative continuous production method disclosed in the patent overcomes these historical barriers by implementing a seamless flow of reactants through static mixers and a coil-type continuous reactor. This approach ensures that the amino acid salt aqueous solution, alkali solution, and chloroacetic acid are mixed uniformly and reacted under precisely controlled thermal conditions, typically between 70-110°C. By maintaining a constant flow rate and residence time, the system eliminates the thermal hotspots common in batch reactors, thereby stabilizing product quality and achieving yields as high as 96% in optimized examples. The process is designed to be safe and environmentally friendly, generating no three wastes throughout the entire operation, which aligns perfectly with modern environmental compliance standards. This shift to continuous processing facilitates the commercial scale-up of complex polymer additives and chelating agents, allowing manufacturers to respond rapidly to market demands while minimizing the carbon footprint associated with chemical production.
Mechanistic Insights into Continuous N-Alkylation Synthesis
The core chemical transformation involves the nucleophilic substitution of amino groups on the amino acid backbone with carboxymethyl groups derived from chloroacetic acid under alkaline conditions. In the first stage, the amino acid is converted into its salt form using sodium or potassium hydroxide, which activates the nitrogen atom for subsequent alkylation. The mixture then enters a secondary static mixer where it encounters the chloroacetic acid solution; the intense turbulence generated within the static mixer ensures molecular-level contact between the reactants before they enter the heated reaction zone. This pre-mixing is crucial for preventing localized over-concentration of chloroacetic acid, which could otherwise lead to excessive formation of byproducts like 2-hydroxyacetic acid. The continuous reactor, often a coil type, provides a plug-flow environment where every molecule experiences the same residence time and temperature profile, ensuring uniform conversion rates. This precise control over reaction kinetics is what enables the process to achieve high purity specifications and rigorous QC labs standards required by top-tier pharmaceutical and agrochemical clients.
Following the reaction phase, the effluent is transferred to a heat-preservation external steaming kettle where negative pressure evaporation is employed to concentrate the product and separate byproduct salts. This step is critical for impurity control mechanisms, as it allows for the crystallization and removal of sodium chloride, which is a major byproduct of the neutralization reaction. The filtrate, now enriched with the target N,N-dicarboxymethyl amino acid salt, is diluted to the desired concentration, typically around 38% mass fraction for commercial applications. The ability to recycle the distilled water from the evaporation step back into the raw material dissolution tank further enhances the sustainability profile of the process. For technical teams evaluating high-purity OLED material or specialty chemical precursors, this level of impurity management demonstrates a robust capability to deliver materials that meet stringent downstream processing requirements without extensive purification steps.
How to Synthesize N,N-Dicarboxymethyl Amino Acid Salt Efficiently
Implementing this continuous synthesis route requires careful calibration of flow rates and molar ratios to maximize yield and minimize waste. The process begins with the preparation of feed streams where the molar ratio of amino acid to alkali is strictly controlled between 1:(1.8-2.4) to ensure complete salt formation without excess base. Detailed standardized synthesis steps see the guide below for specific operational parameters regarding temperature and pressure settings.
- Prepare amino acid salt aqueous solution and alkali solution, pumping them continuously into a first-stage static mixer at a molar ratio of 1: (1.8-2.4).
- Feed the mixed materials into a secondary static mixer while introducing chloroacetic acid aqueous solution, ensuring a molar ratio of amino acid salt to chloroacetic acid of 1: (2.05-2.30).
- Inject the fully mixed stream into a continuous coil reactor maintained at 70-110°C for 1-6 hours, followed by negative pressure evaporation and solid-liquid separation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this continuous manufacturing technology offers profound strategic benefits beyond simple unit cost metrics. The elimination of batch-to-batch variability reduces the risk of production delays caused by off-spec material, thereby enhancing supply chain reliability and ensuring consistent availability of critical raw materials for formulation teams. The process design inherently supports scalability, allowing production volumes to be increased simply by extending run times or numbering up reactor units rather than building entirely new facilities. This flexibility is essential for reducing lead time for high-purity biodegradable intermediates during periods of sudden market demand spikes. Furthermore, the absence of toxic reagents like hydrocyanic acid simplifies regulatory compliance and reduces the overhead costs associated with safety monitoring and hazardous waste treatment.
- Cost Reduction in Manufacturing: The continuous nature of the reaction significantly lowers energy consumption per unit of product compared to heating and cooling large batch vessels repeatedly. By improving equipment utilization rates, the capital expenditure required per ton of output is drastically reduced, leading to substantial cost savings over the lifecycle of the plant. The high yield achieved minimizes raw material waste, ensuring that expensive amino acid inputs are converted efficiently into valuable chelating agents. Additionally, the recovery and reuse of byproduct sodium chloride and distilled water further contribute to a leaner cost structure, providing a competitive edge in price-sensitive markets without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the continuous flow system ensures a steady output of material, mitigating the risks associated with batch failures that can disrupt downstream production schedules. The use of readily available raw materials such as amino acids and chloroacetic acid secures the supply base against volatility often seen with specialized reagents. This stability allows supply chain heads to plan inventory levels more accurately and reduce the need for safety stock buffers. The simplified process flow also means fewer mechanical components subject to wear and tear, resulting in higher uptime and more predictable maintenance windows, which is crucial for maintaining uninterrupted service to global clients.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward due to the modular nature of continuous reactors, avoiding the non-linear challenges often encountered when scaling batch chemistry. The green chemistry principles embedded in the method, such as zero three-waste generation and the production of biodegradable end-products, align with increasingly strict global environmental regulations. This proactive compliance reduces the risk of future regulatory shutdowns and enhances the brand reputation of companies adopting this technology. The ability to produce eco-friendly alternatives to EDTA positions suppliers as leaders in sustainability, appealing to end-consumers and corporate buyers who prioritize green sourcing in their procurement policies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous production of N,N-dicarboxymethyl amino acid salts. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature to ensure accuracy and relevance for industry professionals.
Q: What are the primary advantages of the continuous synthesis method over batch processing?
A: The continuous method significantly improves equipment utilization rates and stabilizes product quality across batches. Unlike traditional intermittent kettle synthesis which suffers from long material transfer periods and inconsistent quality, this process ensures uniform reaction conditions, leading to higher yields and reduced production costs without generating three wastes.
Q: How does this process address environmental compliance concerns?
A: This synthesis route is designed to be safe and environmentally friendly by eliminating the generation of three wastes during the whole process. The method utilizes amino acids and chloroacetic acid under controlled conditions, producing biodegradable end-products that meet OECD standards, thereby replacing toxic traditional chelating agents like EDTA and NTA.
Q: What purity levels can be expected from the final GLDA or MGDA solution?
A: Experimental data indicates that the process yields high-purity solutions with minimal byproduct content. For instance, tetrasodium glutamate diacetate (GLDA) solutions can achieve mass fractions around 38% with byproduct 2-hydroxyacetic acid content kept as low as 1.34%, ensuring high performance in downstream applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Dicarboxymethyl Amino Acid Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous flow chemistry in delivering high-performance green chelating agents to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to industrial reality. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of GLDA or MGDA meets the exacting standards required by the pharmaceutical and agrochemical industries. We understand that consistency is key, and our advanced manufacturing capabilities allow us to maintain tight control over critical process parameters, delivering products with minimal impurity profiles and reliable performance characteristics.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this advanced synthesis technology. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our continuous production methods can enhance your product portfolio. Let us help you secure a sustainable and cost-effective supply of high-purity biodegradable intermediates for your next generation of eco-friendly formulations.
