Advanced Catalytic Synthesis of 3,3',4,4'-Biphenyltetracarboxylic Acid for High-Performance Polyimides
The global demand for high-performance polyimide materials continues to surge, driven by critical applications in flexible displays, aerospace composites, and next-generation lithium battery systems. At the heart of this advanced material supply chain lies 3,3',4,4'-biphenyltetracarboxylic acid (BPTA), a pivotal monomer that dictates the thermal stability and mechanical integrity of the final polymer. A groundbreaking technical disclosure found in patent CN101607894B outlines a novel preparation method that addresses longstanding inefficiencies in BPTA synthesis. This proprietary approach leverages a unique palladium carbon-potassium iodide binary composite catalyst to facilitate the self-dehalogenation coupling of 4-halophthalic acid derivatives. By shifting away from traditional stoichiometric oxidants or expensive homogeneous catalysts, this technology offers a robust pathway for producing electronic chemical intermediates with exceptional purity and yield. For R&D directors and procurement strategists, understanding this mechanistic shift is essential for securing a competitive edge in the high-purity OLED material and polymer additive markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of biphenyltetracarboxylic acid has been plagued by significant economic and technical bottlenecks that hinder efficient commercial scale-up of complex polymer additives. Prior art methods, such as those disclosed in US5095144, often rely on palladium-iron catalysts with glycerin as a reducing agent, yet these processes frequently struggle to achieve yields exceeding 74.2% despite complete conversion of the starting sodium salt. Alternative routes involving oxidative coupling of dimethyl phthalate or electrolytic coupling methods introduce further complications, including harsh reaction conditions, complex downstream purification requirements, and the generation of substantial hazardous waste streams. Furthermore, nickel-based catalytic systems, while sometimes effective for coupling, often suffer from low selectivity and the difficult removal of trace metal impurities, which is unacceptable for high-purity electronic chemical manufacturing. These legacy technologies result in elevated production costs, inconsistent batch quality, and extended lead times for high-purity intermediates, creating vulnerability in the supply chain for downstream polyimide manufacturers who require stringent material specifications.
The Novel Approach
The innovative methodology described in the patent data introduces a paradigm shift by utilizing a palladium carbon-potassium iodide binary composite catalyst in conjunction with simple alcohol reducing agents like methanol or glycerin. This system operates under relatively mild alkaline conditions at temperatures ranging from 80°C to 100°C, significantly reducing energy consumption compared to high-temperature fusion or aggressive oxidative processes. The introduction of potassium iodide as a promoter enhances the catalytic activity of the palladium carbon, facilitating a more efficient dehalogenation coupling mechanism that drives yields substantially higher, with experimental data indicating results reaching up to 91.5%. Crucially, the heterogeneous nature of the catalyst allows for straightforward filtration and potential regeneration, directly addressing the cost reduction in electronic chemical manufacturing by minimizing precious metal loss. This streamlined process not only simplifies the operational workflow but also ensures a more consistent impurity profile, making it an ideal candidate for reliable agrochemical intermediate supplier networks transitioning into high-value electronic materials.
Mechanistic Insights into Pd/C-KI Catalyzed Dehalogenation Coupling
The core of this technological advancement lies in the synergistic interaction between the palladium active sites and the iodide promoter within the binary composite catalyst structure. During the reaction, the 4-halophthalic acid substrate, dissolved in an alkaline medium, undergoes oxidative addition to the palladium center, a step that is kinetically accelerated by the presence of iodide ions which modify the electronic environment of the metal. The alcohol reducing agent, added dropwise over a controlled period of 3 to 8 hours, serves as the hydrogen source necessary for the reductive elimination step that forms the critical carbon-carbon bond between the two phthalic acid moieties. This controlled addition prevents local exotherms and minimizes side reactions such as over-reduction or esterification, ensuring that the reaction pathway remains selective for the desired biphenyl tetracarboxylate structure. The molar ratio of the effective hydroxyl groups in the alcohol to the halophthalic acid is meticulously maintained between 1:1 and 1:3, optimizing the stoichiometry to maximize conversion while minimizing excess reagent waste.
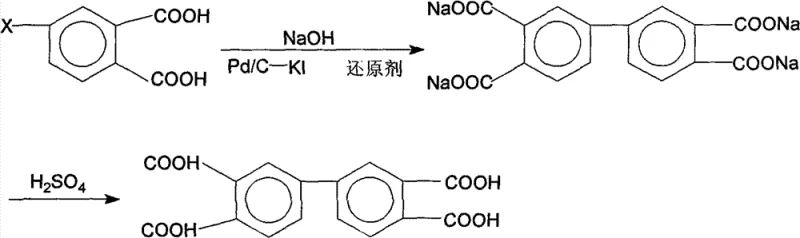
Following the coupling reaction, the resulting product exists primarily as a water-soluble alkali metal salt, which allows for the easy physical separation of the solid catalyst via filtration. This phase separation is a critical control point for maintaining product purity, as it prevents catalyst leaching into the final product stream. The subsequent acidification step using dilute sulfuric acid precipitates the free acid form of the biphenyltetracarboxylic acid, which is then subjected to a rigorous refining process involving hot water washing. This purification stage is instrumental in removing residual inorganic salts and organic byproducts, yielding a final product with purity levels consistently above 99.5% and melting points in the range of 297-300°C. Such precise control over the impurity spectrum is vital for R&D teams focusing on the defect density and dielectric properties of the resulting polyimide films used in semiconductor and display applications.
How to Synthesize 3,3',4,4'-Biphenyltetracarboxylic Acid Efficiently
Implementing this synthesis route requires careful attention to the preparation of the binary catalyst and the precise control of reaction parameters to ensure reproducibility and safety. The process begins with the dissolution of solid potassium iodide in an aqueous methanol solution, followed by the incorporation of the palladium carbon catalyst and activated carbon to form the active composite material. Once the catalyst is prepared, the main reaction involves dissolving the 4-halophthalic acid in a 3-10% alkali solution, ensuring the molar ratio of carboxyl groups to hydroxide ions is maintained between 1:2 and 1:3 for optimal solubility and reactivity. The detailed standardized synthesis steps see the guide below, which outlines the specific temperature ramps, addition rates, and workup procedures necessary to achieve the high yields reported in the patent examples. Adhering to these protocols allows manufacturers to replicate the success of the laboratory examples on a larger scale while maintaining the stringent quality controls required for commercial supply.
- Preparation of alkali solution and dissolution of 4-halophthalic acid with specific molar ratios.
- Addition of Pd/C-KI binary catalyst and controlled dropwise addition of alcohol reducing agent at 80-100°C.
- Acidification of the filtrate with sulfuric acid to precipitate and refine the crude BPTA product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology translates into tangible strategic benefits that extend beyond simple unit cost metrics. The ability to utilize a recoverable heterogeneous catalyst system drastically simplifies the downstream processing requirements, eliminating the need for complex chromatographic purification or extensive solvent exchanges that typically drive up operational expenditures. By removing the dependency on stoichiometric amounts of expensive transition metal complexes or hazardous oxidizing agents, the process inherently reduces the raw material cost burden and mitigates the environmental compliance risks associated with heavy metal waste disposal. This efficiency gain supports a more resilient supply chain, as the raw materials involved—such as halophthalic acids, common alcohols, and alkali hydroxides—are widely available commodity chemicals with stable market pricing. Consequently, this reduces lead time for high-purity intermediates and ensures a more predictable production schedule, which is critical for meeting the just-in-time delivery demands of multinational electronics and aerospace clients.
- Cost Reduction in Manufacturing: The implementation of the Pd/C-KI binary catalyst system offers a profound opportunity for cost optimization by significantly extending the lifecycle of the precious metal catalyst through effective recovery and reuse protocols. Unlike homogeneous catalytic systems where metal removal is costly and often incomplete, this heterogeneous approach allows for simple filtration, thereby reducing the consumption of palladium per kilogram of product produced. Furthermore, the use of inexpensive alcohol reducing agents instead of specialized hydride donors or high-pressure hydrogen gas lowers the utility and safety infrastructure costs associated with the reactor setup. These cumulative efficiencies result in substantial cost savings without compromising the quality of the final electronic grade material, making it a financially viable option for large-scale production.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes directly to supply chain stability by minimizing the risk of batch failures due to sensitive reaction conditions. The tolerance of the catalyst system to variations in raw material quality and the moderate operating temperatures reduce the likelihood of thermal runaways or equipment corrosion, ensuring consistent uptime for manufacturing facilities. Additionally, the simplified workup procedure accelerates the overall cycle time from raw material intake to finished goods packaging, allowing suppliers to respond more agilely to fluctuations in market demand. This reliability is paramount for partners seeking a reliable electronic chemical supplier who can guarantee continuity of supply for critical polyimide precursors used in mission-critical applications.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed to minimize the generation of hazardous byproducts, aligning with increasingly stringent global regulations on industrial emissions and waste management. The aqueous nature of the reaction medium and the use of sulfuric acid for precipitation generate waste streams that are easier to treat and neutralize compared to organic solvent-heavy processes. This green chemistry advantage facilitates easier permitting for capacity expansion and reduces the long-term liability associated with environmental remediation. The straightforward scale-up potential means that production volumes can be increased from pilot scale to multi-ton annual capacity with minimal re-engineering of the core process, supporting the growing demand for high-performance polymers in emerging markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific BPTA synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, catalyst handling, and product specifications. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios or for procurement specialists negotiating supply agreements. The answers reflect the practical realities of scaling this chemistry while maintaining the high standards required for electronic and aerospace material applications.
Q: What is the primary advantage of the Pd/C-KI binary catalyst over traditional methods?
A: The Pd/C-KI binary catalyst system significantly enhances the yield of 3,3',4,4'-biphenyltetracarboxylic acid compared to conventional palladium-only or nickel-based catalysts, achieving yields over 90% while allowing for catalyst recovery and reuse.
Q: Can this synthesis method be scaled for industrial polyimide production?
A: Yes, the process utilizes common reagents like alcohols and aqueous alkali solutions under moderate temperatures (80-100°C), making it highly suitable for commercial scale-up with reduced safety risks and operational complexity.
Q: How does this method impact the purity of the final electronic grade material?
A: By optimizing the molar ratios of reactants and employing a specific refining step involving hot water washing, the method consistently produces BPTA with purity levels exceeding 99.5%, which is critical for high-performance polyimide applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3',4,4'-Biphenyltetracarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent literature can be reliably replicated at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze trace metal content and organic impurities, guaranteeing that every batch of BPTA meets the exacting standards required for polyimide synthesis. Our commitment to quality assurance means that clients can trust our materials to perform consistently in their downstream polymerization processes, reducing the risk of film defects or performance variability in the final electronic components.
We invite forward-thinking organizations to collaborate with us to leverage this advanced catalytic technology for their supply chain needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than theoretical projections. Together, we can secure a sustainable and cost-effective supply of high-performance chemical intermediates that drive innovation in the global electronics and advanced materials sectors.
