Advanced Synthetic Route for 2-Methyl-3-Trifluoromethyl Phenylamine: Scalable Production for Veterinary APIs
The pharmaceutical landscape for veterinary medicines continues to evolve, demanding more efficient and cost-effective pathways for critical active pharmaceutical ingredient (API) intermediates. Patent CN102491906A introduces a robust synthetic methodology for 2-methyl-3-trifluoromethyl phenylamine, a pivotal precursor in the manufacturing of Flunixin Meglumine, a widely utilized non-steroidal anti-inflammatory drug (NSAID) in animal health. This innovation addresses long-standing challenges in regioselectivity and yield that have historically plagued the production of this complex fluorinated aniline derivative. By shifting the synthetic starting point to p-toluenesulfonic acid, the disclosed process eliminates the formation of difficult-to-separate isomeric mixtures, offering a streamlined approach that enhances both chemical efficiency and economic viability for large-scale manufacturers. The strategic integration of sulfonic acid blocking groups ensures precise positioning of substituents on the aromatic ring, a critical factor for downstream biological activity.
Furthermore, the technical implications of this patent extend beyond mere yield improvements; they represent a fundamental shift in how fluorinated veterinary intermediates can be sourced reliably. For R&D directors and process chemists, the ability to bypass hazardous high-pressure fluorination steps while maintaining high purity standards is a significant operational advantage. The method leverages common industrial reagents such as nitric acid, bromine, and potassium monofluoride, reducing dependency on exotic or prohibitively expensive catalysts. This accessibility translates directly into supply chain resilience, ensuring that production schedules for essential animal health medications remain uninterrupted. As global demand for high-quality veterinary therapeutics grows, adopting such optimized synthetic routes becomes not just a technical preference but a strategic necessity for maintaining competitive margins and product availability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methyl-3-trifluoromethyl phenylamine has been fraught with significant chemical and economic inefficiencies that hindered widespread commercial adoption. Prior art methods, such as those disclosed in U.S. Patents 4172095 and 4831193, often relied on rearrangement reactions that suffered from poor regioselectivity, inevitably generating complex mixtures of isomers that required energy-intensive rectification and purification steps. These traditional pathways frequently resulted in overall yields ranging merely from 28% to 50%, representing a substantial loss of raw materials and increased waste generation. Moreover, certain legacy processes depended on expensive reagents like trimethylammonium thionyl bromide and consumed large quantities of n-Butyl Lithium, driving up the cost of goods sold (COGS) to unsustainable levels for commodity veterinary drugs. The lack of business-like starting materials in some foreign methods further complicated logistics, creating bottlenecks that made consistent supply difficult to guarantee for large pharmaceutical integrators.
The Novel Approach
In stark contrast, the novel approach detailed in CN102491906A utilizes p-toluenesulfonic acid as a readily available and inexpensive starting material to fundamentally restructure the synthetic logic. By employing the sulfonic acid group as a temporary blocking and directing group, the new method achieves superior control over the nitration and bromination steps, effectively preventing the formation of unwanted by-products that plague earlier techniques. This strategic modification allows for a cleaner reaction profile where the target 3-nitro-4-methyl-5-bromobenzenesulfonic acid intermediate is formed with high specificity. Subsequent desulfonation reveals the desired substitution pattern without the need for complex separation technologies. The transition from a trichloromethyl intermediate to the final trifluoromethyl group via nucleophilic substitution with potassium monofluoride further simplifies the operation, removing the need for specialized high-pressure reactors. This holistic redesign of the synthesis pathway results in a dramatic improvement in process mass intensity and overall economic feasibility.
Mechanistic Insights into the Multi-Step Substitution and Fluorination Strategy
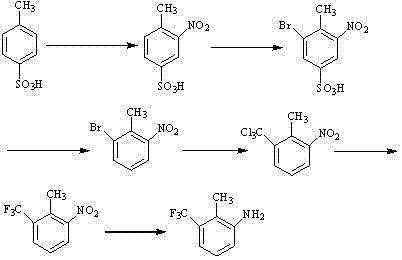
The core of this synthetic breakthrough lies in the meticulous orchestration of electrophilic aromatic substitutions followed by a halogen exchange sequence. The process initiates with the nitration of p-toluenesulfonic acid in concentrated sulfuric acid, where the sulfonic acid group directs the incoming nitro group to the meta position relative to itself, while the methyl group directs ortho/para, resulting in the specific 3-nitro-4-methyl configuration. Following this, bromination occurs selectively at the remaining open position activated by the methyl group, yielding 3-nitro-4-methyl-5-bromobenzenesulfonic acid. The subsequent desulfonation step is critical; heating this intermediate in dilute sulfuric acid removes the sulfonic acid group, leaving behind the precisely oriented 2-nitro-6-bromotoluene scaffold. This sequence effectively uses the sulfonic acid moiety as a 'chemical mask' to ensure the nitro and bromo groups end up in the correct 1,2,3-relationship on the final ring, a feat difficult to achieve through direct substitution on toluene due to competing directing effects.
Following the establishment of the aromatic core, the transformation of the bromine atom into a trifluoromethyl group represents the most chemically sophisticated phase of the route. The protocol employs n-Butyl Lithium to effect a lithium-halogen exchange at low temperatures, generating a reactive aryl lithium species which is then quenched with carbon tetrachloride to install a trichloromethyl group. This trichloromethyl intermediate serves as a precursor for the final fluorination step, where potassium monofluoride (KF) in dimethylformamide (DMF) facilitates a nucleophilic substitution. This halogen exchange (Halex-type) reaction replaces the three chlorine atoms with fluorine, leveraging the high lattice energy of the resulting potassium chloride salt to drive the equilibrium forward. Finally, the nitro group is reduced to an amine using catalytic hydrogenation with palladium on carbon, completing the synthesis of 2-methyl-3-trifluoromethyl phenylamine under mild conditions that preserve the integrity of the sensitive trifluoromethyl group.
How to Synthesize 2-Methyl-3-Trifluoromethyl Phenylamine Efficiently
The execution of this synthesis requires careful attention to temperature control and stoichiometry, particularly during the lithiation and fluorination stages to maximize yield and safety. The patent outlines a standardized six-step protocol that begins with the preparation of 3-nitro-4-methyl benzenesulfonic acid and concludes with the catalytic reduction of the nitro group. Each step has been optimized to minimize side reactions, such as over-nitration or incomplete halogen exchange, ensuring a high-purity final product suitable for pharmaceutical applications. Operators must adhere to strict anhydrous conditions during the organolithium step to prevent quenching of the reactive intermediate, while the fluorination step requires sustained heating to ensure complete conversion of the trichloromethyl precursor. Detailed standard operating procedures (SOPs) regarding reagent addition rates and workup protocols are essential for replicating the high yields reported in the patent examples.
- Nitration of p-toluenesulfonic acid with nitric and sulfuric acid to form 3-nitro-4-methyl benzenesulfonic acid.
- Bromination followed by acidic desulfonation to yield 2-nitro-6-bromotoluene with high regioselectivity.
- Lithiation and trichloromethylation using n-Butyl Lithium and carbon tetrachloride, followed by fluorination with KF and catalytic hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend well beyond the laboratory bench, directly impacting the bottom line and operational stability. The primary advantage lies in the drastic simplification of the raw material portfolio; by utilizing p-toluenesulfonic acid, a commodity chemical produced in massive volumes globally, manufacturers can decouple their supply chains from the volatility associated with specialized, low-volume intermediates. This shift ensures a more reliable sourcing strategy, reducing the risk of production stoppages due to raw material shortages. Furthermore, the elimination of expensive reagents like trimethylammonium thionyl bromide and the significant reduction in n-Butyl Lithium consumption translate directly into lower variable costs per kilogram of finished product. The process avoids the need for custom-synthesized starting materials, allowing procurement teams to leverage existing vendor relationships and spot market pricing for bulk chemicals.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the removal of costly purification steps associated with isomer separation in older methods. By achieving high regioselectivity early in the synthesis, the need for resource-intensive distillation or chromatography is minimized, leading to substantial savings in energy and solvent consumption. Additionally, the replacement of high-pressure fluorination equipment with standard atmospheric or low-pressure reactors reduces capital expenditure (CAPEX) requirements for facility upgrades. The overall yield improvement means that less raw material is wasted per unit of output, effectively lowering the cost of goods sold and improving margin potential for the final veterinary drug.
- Enhanced Supply Chain Reliability: Reliance on widely available industrial chemicals such as sulfuric acid, nitric acid, and bromine ensures that the supply chain remains robust against geopolitical or logistical disruptions. Unlike processes dependent on niche fluorinating agents that may have limited suppliers, the reagents used in this pathway are produced by multiple chemical manufacturers worldwide. This diversity in sourcing options provides procurement teams with greater negotiating power and flexibility. The simplified logistics of handling stable solid intermediates, rather than unstable or hazardous liquids required in alternative routes, also reduces transportation costs and regulatory compliance burdens associated with hazardous material shipping.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed for seamless transition from pilot plant to multi-ton commercial production. The avoidance of high-pressure fluorination not only enhances safety but also simplifies the engineering controls required for scale-up, allowing for faster capacity expansion. Waste streams are more manageable due to the absence of heavy metal catalysts in the fluorination step and the use of aqueous workups for desulfonation. This aligns with increasingly stringent environmental regulations, reducing the cost of waste treatment and disposal. The ability to run the final hydrogenation step at moderate temperatures and pressures further underscores the process's suitability for green chemistry initiatives and sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology for 2-methyl-3-trifluoromethyl phenylamine. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety profiles, and scalability factors. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer or process optimization projects. The answers reflect the consensus of the patent's findings regarding the superiority of the sulfonic acid-directed route over traditional rearrangement or direct substitution methods.
Q: What are the key advantages of using p-toluenesulfonic acid as a starting material?
A: Using p-toluenesulfonic acid allows for precise regiocontrol during nitration and bromination, overcoming the mixture formation issues seen in prior art rearrangement methods, thereby significantly improving overall yield and purity.
Q: How does this process improve safety compared to traditional fluorination methods?
A: This method utilizes a nucleophilic substitution fluorination with Potassium Monofluoride (KF) in DMF, which avoids the hazardous high-pressure fluorination operations and specialized equipment required by older technologies.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process utilizes commercially available raw materials like tosics acid and avoids expensive catalysts or extreme conditions, making it highly suitable for industrial scale-up with consistent quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-3-Trifluoromethyl Phenylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of next-generation veterinary therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex chemistry involved in fluorinated aniline synthesis is managed with precision and consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Whether your requirement is for clinical trial materials or full-scale commercial supply, our infrastructure is designed to provide the reliability and quality assurance that global pharmaceutical partners demand.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall production costs without compromising on quality. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your supply chain requirements. Let us collaborate to bring efficient, cost-effective, and high-purity veterinary drug intermediates to the market, ensuring the health and well-being of animals worldwide through superior chemical manufacturing.
