Optimizing Ticagrelor Intermediate Production: A Technical Analysis of Novel Iodination Strategies
Optimizing Ticagrelor Intermediate Production: A Technical Analysis of Novel Iodination Strategies
The pharmaceutical landscape for anticoagulants has been significantly reshaped by the introduction of Ticagrelor, a potent P2Y12 receptor inhibitor. As demand for this critical active pharmaceutical ingredient (API) continues to grow, the efficiency of its supply chain relies heavily on the robustness of its key intermediates. Patent CN110981883B introduces a groundbreaking preparation method for the Ticagrelor key intermediate iodide, specifically (3aS,4S,6R,6aR)-4-iodomethyl-6-methoxy-2,2-dimethyltetrahydrofuran[3,4-d][1,3]dioxolane. This technical insight report analyzes the proprietary two-step synthesis route disclosed in the patent, highlighting how replacing traditional hydrochloric acid catalysis with thionyl chloride and utilizing a trimethylchlorosilane-mediated iodination strategy can drastically enhance process economics. For R&D directors and procurement managers alike, understanding these mechanistic shifts is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical furanose derivative has been plagued by inefficiencies that hinder large-scale commercial viability. One prevalent prior art method, illustrated in earlier patent literature, involves a three-step sequence starting from D-ribose that generates equimolar amounts of sodium p-toluenesulfonate by-products. 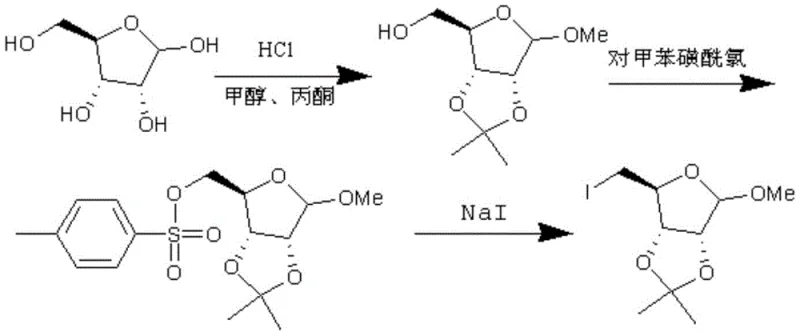 This accumulation of stoichiometric waste not only complicates the purification process but also imposes a heavy burden on waste treatment facilities, driving up the operational expenditure for manufacturers. Furthermore, alternative routes reported in academic journals often rely on iodine and triphenylphosphine systems. While chemically feasible, these methods suffer from low reaction yields and the generation of substantial quantities of triphenylphosphine oxide. The removal of this oxide byproduct is notoriously difficult and energy-intensive, often requiring extensive chromatography or recrystallization steps that erode the overall mass balance and delay production timelines.
This accumulation of stoichiometric waste not only complicates the purification process but also imposes a heavy burden on waste treatment facilities, driving up the operational expenditure for manufacturers. Furthermore, alternative routes reported in academic journals often rely on iodine and triphenylphosphine systems. While chemically feasible, these methods suffer from low reaction yields and the generation of substantial quantities of triphenylphosphine oxide. The removal of this oxide byproduct is notoriously difficult and energy-intensive, often requiring extensive chromatography or recrystallization steps that erode the overall mass balance and delay production timelines.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology outlined in CN110981883B streamlines the synthesis into a highly efficient two-step protocol. The innovation lies in the strategic selection of reagents that maximize atom economy and minimize downstream processing. By employing thionyl chloride as a catalyst in the initial protection and methylation step, the reaction conversion rate is greatly improved, bypassing the equilibrium limitations often seen with aqueous hydrochloric acid. Subsequently, the iodination step utilizes an in-situ generated iodinating agent from trimethylchlorosilane and sodium iodide. This combination allows for mild reaction conditions that preserve the stereochemical integrity of the sugar backbone while ensuring high conversion. The result is a process that is not only shorter but also inherently cleaner, eliminating the need for complex byproduct removal associated with phosphine oxides or sulfonate salts.
Mechanistic Insights into Thionyl Chloride Catalysis and In-Situ Iodination
The first stage of this synthesis involves the transformation of D-ribose into a protected intermediate (Intermediate 1). The use of thionyl chloride (SOCl2) in a methanol and acetone solvent system serves a dual purpose: it acts as a dehydrating agent to drive the formation of the acetonide protecting group and facilitates the methylation of the anomeric position. Operating at a controlled temperature of 10-15°C during addition prevents exothermic runaway, while maintaining the reaction at 25 ± 3°C for 24 hours ensures complete conversion. This specific catalytic environment promotes the formation of the desired stereoisomer with a yield reaching over 90%, a significant improvement over acid-catalyzed equilibria that often leave unreacted starting material. The subsequent removal of solvents under negative pressure yields a crude product ready for the next step without extensive purification, demonstrating the robustness of the catalytic system.
The second stage involves a nucleophilic substitution where the primary hydroxyl group is converted to an iodide. The mechanism relies on the activation of the hydroxyl group by trimethylchlorosilane (TMSCl), forming a reactive silyl ether intermediate which is then displaced by iodide ions from sodium iodide. 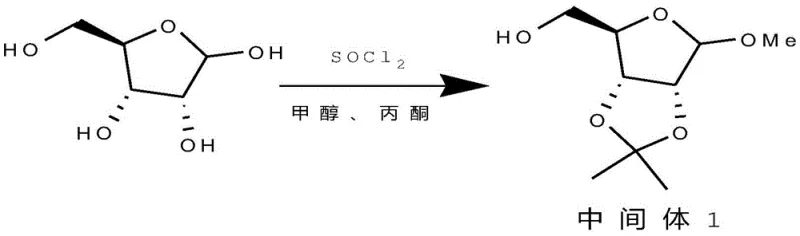 This reaction is conducted in acetonitrile under nitrogen protection to prevent oxidation of the iodide species. The dropwise addition of TMSCl at 20 ± 2°C controls the rate of silylation, preventing side reactions. Following a one-hour stir at room temperature, the reaction is quenched with water. The workup procedure is remarkably simple: distilling off the acetonitrile and extracting with toluene effectively separates the organic product from inorganic salts. This mechanistic pathway avoids the harsh conditions of Appel-type reactions using PPh3/I2, thereby reducing the impurity profile and simplifying the isolation of the final key intermediate iodide with 85% yield and 95% purity.
This reaction is conducted in acetonitrile under nitrogen protection to prevent oxidation of the iodide species. The dropwise addition of TMSCl at 20 ± 2°C controls the rate of silylation, preventing side reactions. Following a one-hour stir at room temperature, the reaction is quenched with water. The workup procedure is remarkably simple: distilling off the acetonitrile and extracting with toluene effectively separates the organic product from inorganic salts. This mechanistic pathway avoids the harsh conditions of Appel-type reactions using PPh3/I2, thereby reducing the impurity profile and simplifying the isolation of the final key intermediate iodide with 85% yield and 95% purity.
How to Synthesize Ticagrelor Key Intermediate Iodide Efficiently
Implementing this novel synthesis route requires precise control over reaction parameters to replicate the high yields reported in the patent data. The process begins with the careful preparation of Intermediate 1, where temperature control during the thionyl chloride addition is critical to safety and selectivity. Once the intermediate is secured, the iodination step must be performed under anhydrous conditions initially, followed by a controlled quench to manage the exotherm and facilitate phase separation. The detailed standardized synthetic steps, including specific molar ratios and workup protocols necessary for GMP-compliant manufacturing, are outlined in the guide below.
- Synthesize Intermediate 1 by reacting D-ribose with methanol and acetone using thionyl chloride as a catalyst at 10-15°C, then warming to 25°C.
- Prepare the iodination mixture by dissolving Intermediate 1 and sodium iodide in acetonitrile under nitrogen protection.
- Add trimethylchlorosilane dropwise at 20°C, stir for 1 hour, quench with water, and extract with toluene to isolate the key iodide intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this optimized synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational costs and enhanced supply reliability. By condensing the synthesis into fewer steps and eliminating the need for expensive and difficult-to-remove reagents like triphenylphosphine, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing. The absence of stubborn byproducts means less solvent consumption for purification and shorter batch cycle times, allowing facilities to increase throughput without capital expansion. Furthermore, the use of commodity chemicals like thionyl chloride and sodium iodide ensures that raw material sourcing remains stable and unaffected by the volatility often seen with specialized organophosphorus reagents.
- Cost Reduction in Manufacturing: The elimination of triphenylphosphine and the reduction of reaction steps fundamentally alters the cost structure of production. Traditional methods incur hidden costs associated with the disposal of phosphine oxide waste and the extended labor hours required for complex purifications. By switching to the TMSCl/NaI system, these variable costs are substantially lowered. Additionally, the high conversion rate of over 90% in the first step minimizes the loss of expensive D-ribose starting material, ensuring that the input costs are maximized in the final output. This efficiency translates into a more competitive pricing model for the final API, providing a strategic advantage in tender negotiations.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex syntheses that have multiple failure points. This two-step method reduces the number of unit operations, thereby statistically lowering the risk of batch failures due to human error or equipment malfunction. The robustness of the thionyl chloride catalysis means that the process is less sensitive to minor fluctuations in reaction conditions compared to enzymatic or highly sensitive organometallic alternatives. Consequently, suppliers adopting this technology can offer more consistent lead times and maintain higher safety stock levels, mitigating the risk of shortages for downstream API manufacturers who depend on just-in-time delivery schedules.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals bottlenecks related to heat transfer and waste management. This novel route is inherently scalable because it avoids the generation of solid sludge (like phosphine oxides) that can clog filters and reactors at large volumes. The liquid-liquid extraction workup using toluene and water is a standard unit operation easily managed in multi-purpose reactors ranging from pilot to commercial scale. Moreover, the reduced waste profile aligns with increasingly stringent environmental regulations, lowering the compliance burden and potential fines associated with hazardous waste disposal. This makes the process not only economically viable but also sustainable for long-term commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits for potential partners.
Q: Why is thionyl chloride preferred over hydrochloric acid in the first step?
A: According to patent CN110981883B, using thionyl chloride significantly improves the reaction conversion rate compared to traditional hydrochloric acid catalysis, achieving yields over 90% for Intermediate 1.
Q: What are the advantages of the TMSCl/NaI system over traditional iodination methods?
A: The trimethylchlorosilane/sodium iodide system offers milder reaction conditions and simpler post-treatment compared to methods using triphenylphosphine and iodine, which generate difficult-to-remove oxide byproducts.
Q: What is the overall purity profile of the synthesized intermediate?
A: The optimized process described in the patent yields the key intermediate iodide with a purity of approximately 95% and an isolated yield of 85%, suitable for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ticagrelor Intermediate Supplier
As the global demand for cardiovascular therapeutics evolves, partnering with a manufacturer that possesses deep technical expertise in process optimization is essential. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN110981883B to deliver superior quality intermediates. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric requirements of major pharmaceutical contracts. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Ticagrelor intermediate meets the exacting standards required for FDA and EMA filings.
We invite procurement leaders and R&D teams to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain needs. By collaborating with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our optimized processes can reduce your total cost of ownership. Contact us today to discuss how we can support your API production goals with reliable, high-quality chemical solutions.
