Advanced Synthesis of AZD3759 Intermediate via Green Catalytic Reduction and Mitsunobu Coupling
Advanced Synthesis of AZD3759 Intermediate via Green Catalytic Reduction and Mitsunobu Coupling
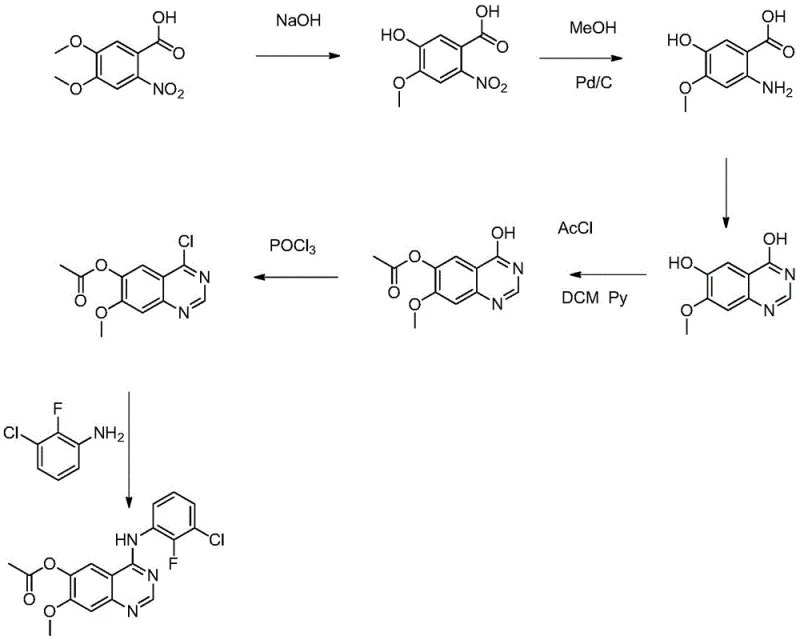
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of critical oncology intermediates. A significant breakthrough in this domain is detailed in patent CN110590683B, which discloses a novel preparation method for the key intermediate of the targeted lung cancer drug AZD3759. This intermediate, chemically known as 4-[(3-chloro-2-fluorophenyl)amino]-6-acetoxy-7-methoxyquinazoline, plays a pivotal role in the synthesis of EGFR inhibitors designed to penetrate the blood-brain barrier. The disclosed technology represents a paradigm shift from traditional hazardous methodologies to a greener, more cost-effective approach that leverages iron-catalyzed reduction and direct Mitsunobu coupling. For R&D directors and procurement specialists alike, understanding this route is essential for securing a reliable supply chain for next-generation CNS-active therapeutics.
![Chemical structure of the target AZD3759 intermediate 4-[(3-chloro-2-fluorophenyl)amino]-6-acetoxy-7-methoxyquinazoline](/insights/img/azd3759-intermediate-mitsunobu-synthesis-pharma-supplier-20260306023312-01.png)
The structural complexity of this quinazoline derivative requires precise functional group manipulation, particularly regarding the installation of the aniline moiety and the management of phenolic hydroxyl groups. The innovation lies not just in the final assembly but in the strategic reordering of synthetic steps to minimize waste and maximize safety. By replacing expensive noble metal catalysts with abundant iron salts and eliminating toxic chlorinating agents, this process addresses two of the most significant pain points in fine chemical manufacturing: operational safety and environmental compliance. As we delve deeper into the technical specifics, it becomes clear why this methodology offers a superior alternative for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of similar quinazoline derivatives has relied heavily on routes that involve harsh chlorination and high-pressure hydrogenation steps. As illustrated in the prior art referenced within the patent documentation, the conventional pathway typically utilizes phosphorus oxychloride (POCl3) to activate the quinazoline ring for nucleophilic substitution. This reagent is notoriously hazardous, generating substantial quantities of acidic wastewater that require complex and costly neutralization processes before disposal. Furthermore, the reduction of the nitro precursor in traditional routes often employs palladium on carbon (Pd/C) under hydrogen pressure. This necessitates the use of specialized high-pressure reactors, which introduces significant capital expenditure and safety risks related to hydrogen gas handling. The combination of toxic reagents and high-pressure operations creates a bottleneck for manufacturers aiming to scale production efficiently while maintaining strict EHS (Environment, Health, and Safety) standards.
The Novel Approach
In stark contrast, the methodology outlined in CN110590683B ingeniously circumvents these obstacles by reimagining the bond formation strategy. Instead of activating the quinazoline with chlorine, the new route employs a Mitsunobu reaction to directly couple the hydroxy-quinazoline with 3-chloro-2-fluoroaniline. This elegant transformation occurs under mild conditions using triphenylphosphine and an azo reagent, completely bypassing the need for POCl3. Additionally, the nitro reduction step is revolutionized by substituting the expensive Pd/C catalyst with a mixture of ferric chloride hexahydrate and activated carbon. This heterogeneous catalytic system operates effectively with hydrazine hydrate at atmospheric pressure, eliminating the need for high-pressure equipment. The result is a streamlined process that not only reduces the number of synthetic steps but also fundamentally alters the safety profile of the manufacturing operation, making it far more attractive for large-scale industrial application.
Mechanistic Insights into FeCl3-Catalyzed Reduction and Mitsunobu Coupling
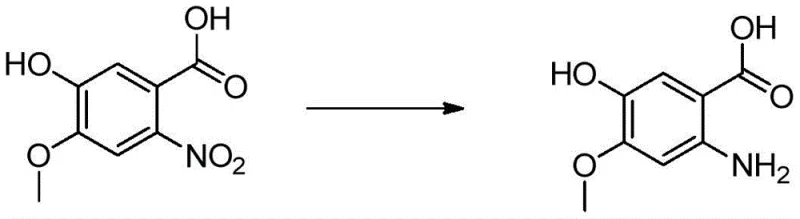
The core of this technological advancement rests on two distinct mechanistic pillars: the iron-catalyzed transfer hydrogenation and the stereospecific Mitsunobu etherification. In the reduction phase, the ferric chloride/activated carbon mixture acts as a potent catalyst for the decomposition of hydrazine hydrate, generating active hydrogen species in situ. These species effectively reduce the nitro group of 2-nitro-4-methoxy-5-hydroxybenzoic acid to the corresponding amine without affecting other sensitive functional groups. The presence of activated carbon likely enhances the dispersion of the iron species and facilitates electron transfer, mimicking the activity of noble metals but at a fraction of the cost. This mechanism allows the reaction to proceed smoothly at temperatures between 60-100°C, providing a robust and scalable alternative to catalytic hydrogenation that avoids the logistical challenges of storing and transporting hydrogen gas.
Following the formation of the quinazoline core and selective acetylation, the final C-N bond formation is achieved through the Mitsunobu reaction mechanism. This reaction involves the activation of the phenolic hydroxyl group at the 4-position of the quinazoline ring by triphenylphosphine and diisopropyl azodicarboxylate (DIAD). The resulting activated intermediate is highly susceptible to nucleophilic attack by the 3-chloro-2-fluoroaniline. Unlike traditional nucleophilic aromatic substitution which often requires elevated temperatures and activated leaving groups (like chlorine), the Mitsunobu protocol proceeds efficiently at mild temperatures ranging from 0-30°C. This mildness is crucial for preserving the integrity of the acetoxy group and preventing side reactions such as hydrolysis or polymerization. The high regioselectivity and yield observed in this step underscore the precision of this modern organic transformation in complex molecule synthesis.
How to Synthesize 4-[(3-chloro-2-fluorophenyl)amino]-6-acetoxy-7-methoxyquinazoline Efficiently
Implementing this novel synthetic route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the alkaline hydrolysis of 6-nitroveratric acid, followed by the critical iron-catalyzed reduction. Subsequent cyclization with formamidine acetate builds the quinazoline scaffold, which is then selectively acetylated. The final coupling step demands precise stoichiometry of the phosphine and azo reagents to ensure complete conversion. For process chemists looking to replicate this success, the following standardized procedure outlines the critical operational windows and reagent ratios derived directly from the patent examples. Adhering to these guidelines ensures the production of high-purity material suitable for downstream pharmaceutical applications.
- Hydrolyze 6-nitroveratric acid with NaOH at 80-100°C to obtain 2-nitro-4-methoxy-5-hydroxybenzoic acid.
- Reduce the nitro group using hydrazine hydrate with a FeCl3/Activated Carbon catalyst mixture at 60-100°C.
- Cyclize the amino acid with formamidine acetate in ethylene glycol monomethyl ether at 80-130°C to form the quinazoline core.
- Acetylate the 6-position hydroxyl group using acetyl chloride and triethylamine in DCM at low temperature.
- Perform Mitsunobu reaction with 3-chloro-2-fluoroaniline using PPh3 and DIAD in THF to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic advantages beyond mere technical elegance. The elimination of phosphorus oxychloride removes a major regulatory burden associated with the handling and disposal of corrosive and toxic chemicals. This simplification of the waste stream significantly reduces the operational overhead related to environmental compliance and wastewater treatment facilities. Furthermore, the shift away from high-pressure hydrogenation means that production can be conducted in standard glass-lined or stainless steel reactors without the need for expensive autoclaves. This flexibility allows for broader manufacturing capacity utilization and reduces the lead time for setting up new production lines, thereby enhancing the overall agility of the supply chain in responding to market demands for AZD3759.
- Cost Reduction in Manufacturing: The replacement of palladium on carbon with an iron-based catalyst system represents a drastic reduction in raw material costs. Palladium is a precious metal with volatile pricing and high procurement barriers, whereas ferric chloride is an abundant and inexpensive commodity chemical. Additionally, the removal of the chlorination step eliminates the cost of purchasing POCl3 and the subsequent neutralization bases required to treat the acidic byproducts. By streamlining the synthesis into fewer steps with cheaper reagents, the overall cost of goods sold (COGS) for this intermediate is significantly lowered, allowing for more competitive pricing in the final API market.
- Enhanced Supply Chain Reliability: Relying on noble metal catalysts often introduces supply chain vulnerabilities due to geopolitical factors affecting mining and refining. By transitioning to an iron-catalyzed process, manufacturers mitigate the risk of catalyst shortages. Moreover, the reagents used in the Mitsunobu reaction, such as triphenylphosphine and DIAD, are widely available from multiple global suppliers, ensuring a stable and continuous flow of materials. This diversification of the supply base reduces the risk of production stoppages and guarantees consistent delivery schedules for downstream pharmaceutical clients who depend on timely intermediate availability for their own clinical or commercial timelines.
- Scalability and Environmental Compliance: The inherent safety of operating at atmospheric pressure and moderate temperatures makes this process highly scalable. There is no need for complex safety interlocks associated with high-pressure hydrogen gas, simplifying the engineering requirements for scale-up from pilot plant to multi-ton commercial production. From an environmental perspective, the absence of heavy metal residues and phosphorous waste aligns perfectly with modern green chemistry principles. This facilitates easier regulatory approval in jurisdictions with stringent environmental laws and supports the sustainability goals of major pharmaceutical companies who are increasingly prioritizing green suppliers in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived from the specific experimental data and claims found in the patent literature, providing a clear picture of what potential partners can expect regarding yield, purity, and operational feasibility. Understanding these details is crucial for making informed decisions about technology transfer and long-term supply agreements.
Q: How does the new FeCl3-catalyzed reduction improve safety compared to traditional methods?
A: The novel process replaces high-pressure palladium-carbon hydrogenation with a hydrazine hydrate reduction catalyzed by ferric chloride and activated carbon. This eliminates the need for specialized high-pressure reactors and significantly reduces the risk associated with hydrogen gas handling, making the operation safer and more accessible for standard chemical plants.
Q: What are the environmental benefits of using the Mitsunobu reaction in this route?
A: By utilizing a direct Mitsunobu coupling between the hydroxy-quinazoline and the aniline derivative, the process completely avoids the use of phosphorus oxychloride (POCl3). This removal prevents the generation of large volumes of highly acidic and toxic phosphorous-containing wastewater, drastically simplifying waste treatment and reducing environmental compliance costs.
Q: What purity levels can be achieved with this optimized synthetic route?
A: According to the patent data, the final product 4-[(3-chloro-2-fluorophenyl)amino]-6-acetoxy-7-methoxyquinazoline can be achieved with an HPLC purity of over 99%. The stepwise yields are also robust, with the initial hydrolysis step reaching up to 97.6% and the reduction step achieving 93.1%, ensuring high overall material throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-[(3-chloro-2-fluorophenyl)amino]-6-acetoxy-7-methoxyquinazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes for oncology intermediates like the AZD3759 precursor. Our technical team has extensively analyzed the innovations presented in CN110590683B and possesses the expertise to implement this green chemistry approach effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing floor is seamless. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required for GMP API synthesis.
We invite pharmaceutical partners to leverage our capabilities for cost-effective and sustainable manufacturing solutions. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a reliable supply of high-quality intermediates while optimizing your production costs and minimizing environmental impact through advanced catalytic technologies.
