Scalable Production of 2,5,6-Triamino-4-Pyrimidinone via Mild Acylation Cyclization
Scalable Production of 2,5,6-Triamino-4-Pyrimidinone via Mild Acylation Cyclization
The pharmaceutical industry continuously seeks robust and scalable pathways for critical heterocyclic intermediates, particularly those serving as precursors for life-saving therapies. Patent CN107311939B discloses a groundbreaking preparation method for substituted pyrimidinone derivatives, specifically targeting the synthesis of 2,5,6-triamino-4-pyrimidinone. This compound serves as a pivotal building block for Sapropterin Dihydrochloride, an essential treatment for tetrahydrobiopterin (BH4) deficiency. The disclosed technology represents a significant leap forward in process chemistry, transitioning away from the hazardous and low-yielding protocols of the past toward a streamlined, three-step sequence involving acylation, cyclization, and hydrolysis. For R&D directors and procurement specialists alike, this innovation promises not only enhanced chemical efficiency but also a more reliable supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,5,6-triamino-4-pyrimidinone has been plagued by inefficient multi-step sequences that struggle to meet the demands of modern Good Manufacturing Practice (GMP) standards. Prior art, such as the methodology described by Waer et al. (US 20040077859), relies on starting materials like 2,6-diamino-4-chloropyrimidine, necessitating harsh substitution reactions that often result in overall yields hovering around a mere 30%. Similarly, routes disclosed by Yoshida et al. (JP61009587) utilize 2,4-diamino-6-hydroxy-pyrimidine, involving nitrosation steps that introduce significant safety hazards and complicate waste management. These conventional pathways are characterized by rigorous reaction conditions, difficult post-treatment procedures, and the generation of complex impurity profiles that require extensive purification resources.
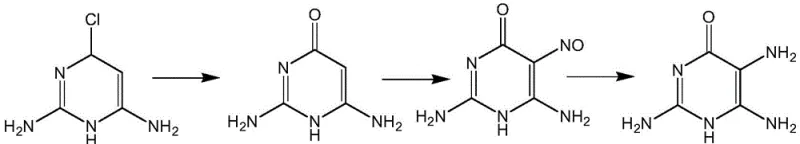
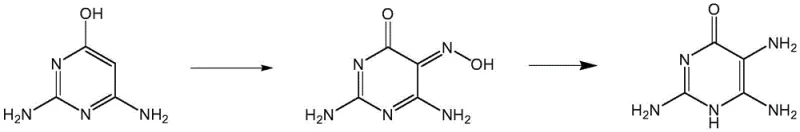
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes ethyl 2-amino-cyanoacetate as a cost-effective and readily available starting material. This strategy employs a clever protecting group tactic where an acyl group is first introduced to the amino functionality, followed by a highly selective cyclization with a guanidine salt. This sequence effectively constructs the pyrimidinone core under remarkably mild alkaline conditions, avoiding the extreme temperatures and corrosive reagents typical of older chlorination or nitrosation routes. The final hydrolysis step cleanly removes the protecting group to reveal the target triamino structure, delivering a process that is not only chemically elegant but also operationally simpler for plant engineers to manage.
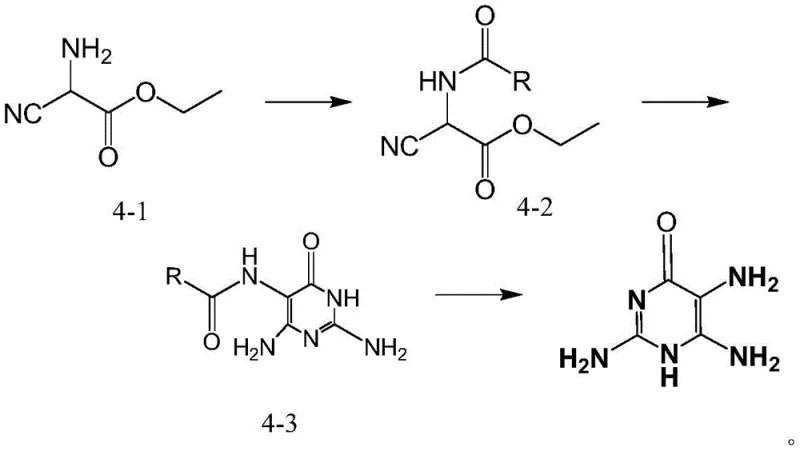
Mechanistic Insights into Acylation-Cyclization-Hydrolysis Cascade
The core of this technological advancement lies in the precise control of reactivity during the cyclization phase. By pre-acylating the ethyl 2-amino-cyanoacetate, the synthesis creates a differentiated electronic environment that guides the nucleophilic attack of the guanidine species specifically towards the nitrile and ester functionalities. This selectivity is crucial for preventing polymerization or the formation of regio-isomers, which are common pitfalls in pyrimidine synthesis. The reaction proceeds smoothly in solvents like ethanol at temperatures between 35-55°C, facilitated by sodium hydroxide, ensuring that the energy barrier for ring closure is overcome without degrading the sensitive amino groups. This mechanistic precision translates directly into a cleaner reaction mass, reducing the burden on downstream purification units.
Furthermore, the hydrolysis mechanism is optimized to preserve the integrity of the newly formed heterocyclic ring while efficiently cleaving the amide bond at the 5-position. Conducted under acidic conditions using hydrochloric acid at moderate temperatures, this step ensures that the final product, 2,5,6-triamino-4-pyrimidinone, is obtained with high structural fidelity. The avoidance of strong oxidizing agents or heavy metal catalysts means that the impurity profile is significantly simplified, primarily consisting of hydrolyzable byproducts rather than persistent metal residues. For quality control teams, this implies a more straightforward analytical validation process and a reduced risk of failing stringent heavy metal specifications required for pharmaceutical intermediates.
How to Synthesize 2,5,6-Triamino-4-Pyrimidinone Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the acylating agent and the pH control during the cyclization step to maximize the formation of the protected intermediate. The process is designed to be telescoped or run in discrete batches depending on the facility's capabilities, with the acylation occurring at near-ambient temperatures to minimize energy consumption. Operators should note that the choice of acylating agent, whether acetic anhydride or benzoyl chloride, can influence the crystallization properties of the intermediate, offering flexibility in process design. For a comprehensive breakdown of the standardized operating procedures and specific molar ratios, please refer to the detailed guide below.
- Perform acylation of ethyl 2-amino-cyanoacetate with acetic anhydride or benzoyl chloride at 0-40°C to form the protected intermediate.
- Execute cyclization by reacting the acylated intermediate with guanidine hydrochloride under alkaline conditions at 35-55°C.
- Complete the synthesis via acidic hydrolysis using hydrochloric acid at 35-55°C to remove the protecting group and obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology addresses several critical pain points that typically inflate the cost of goods sold (COGS) for complex heterocyclic intermediates. By eliminating the need for expensive chlorinated starting materials and hazardous nitrosation reagents, the raw material costs are substantially reduced, creating a more favorable margin structure for bulk purchasers. The mild reaction conditions also imply lower energy requirements for heating and cooling, contributing to a greener manufacturing footprint that aligns with increasingly strict environmental regulations. For supply chain heads, the reliance on commodity chemicals like ethyl 2-amino-cyanoacetate ensures a stable and resilient supply base, mitigating the risks associated with sourcing specialized or controlled precursors.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and hazardous reagents removes the necessity for expensive scavenging steps and specialized waste disposal protocols. This simplification of the downstream processing workflow leads to significant operational savings, as the plant can operate with standard stainless steel equipment rather than requiring exotic alloys resistant to extreme corrosion. Furthermore, the higher overall yield means less raw material is wasted per kilogram of finished product, directly enhancing the economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Utilizing ethyl 2-amino-cyanoacetate as the primary feedstock leverages a widely available global supply chain, reducing the lead time risks associated with custom-synthesized starting materials. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up, ensuring that production targets can be met consistently even during periods of high market demand. This reliability is paramount for pharmaceutical customers who require uninterrupted supply to maintain their own drug formulation schedules.
- Scalability and Environmental Compliance: The aqueous workup and use of common organic solvents like ethanol facilitate easier solvent recovery and recycling, minimizing the environmental impact of the production process. The absence of heavy metals and toxic byproducts simplifies the regulatory compliance landscape, allowing for faster approval of manufacturing sites and smoother audits. This environmental friendliness not only reduces disposal costs but also enhances the corporate social responsibility profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis pathway. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this technology. Understanding these nuances is essential for making informed decisions about process integration and long-term sourcing strategies.
Q: What are the key advantages of this new synthesis route over conventional methods?
A: The novel route utilizes mild reaction conditions (0-55°C) compared to the harsh environments of prior art, resulting in significantly higher overall yields (up to 47% total vs 30-40%) and simplified post-treatment processes suitable for industrial scale-up.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method is specifically designed for large-scale industrial production, utilizing readily available raw materials like ethyl 2-amino-cyanoacetate and avoiding expensive or hazardous transition metal catalysts that complicate supply chains.
Q: How does the protecting group strategy improve product purity?
A: By introducing a protected amino group at the 5-position prior to cyclization, the reaction achieves superior selectivity, minimizing side reactions and facilitating easier purification of the final 2,5,6-triamino-4-pyrimidinone intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5,6-Triamino-4-Pyrimidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2,5,6-triamino-4-pyrimidinone meets the exacting standards required for API synthesis. Our infrastructure is designed to handle the specific nuances of acylation and cyclization chemistries, providing a secure and compliant manufacturing environment.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in your pharmaceutical development journey.
