Advanced Solvent-Free Synthesis of Alpha, Alpha-Dichloroethyl Cyclopropane for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for critical intermediates that balance high purity with operational safety. Patent CN109678651B introduces a groundbreaking preparation method for high-purity alpha, alpha-dichloroethyl cyclopropane, a vital building block for synthesizing cyclopropyl acetylene and subsequently anti-AIDS medications. This technology represents a significant leap forward by abandoning traditional solvent-heavy protocols in favor of a streamlined, solvent-free system that leverages in-situ by-product conversion. The core innovation lies in the strategic use of N,N-disubstituted amides, which react with phosphorus oxychloride to form a separable Vilsmeier reagent, thereby bypassing the hazardous distillation steps that typically degrade product quality. For R&D directors and procurement specialists, this patent offers a compelling pathway to achieve content levels exceeding 99% and total reaction yields surpassing 97%, setting a new benchmark for efficiency in fine chemical manufacturing.
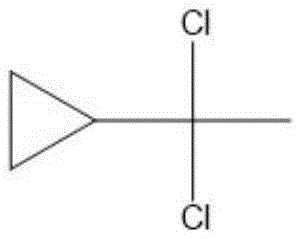
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha, alpha-dichloroethyl cyclopropane has been plagued by significant downstream processing challenges that inflate costs and compromise yield. Conventional methodologies typically involve the addition of phosphorus pentachloride to cyclopropyl methyl ketone within a large volume of inert organic solvent. While this approach facilitates mixing, it creates a complex reaction matrix where the by-product, phosphorus oxychloride, remains dissolved alongside the target molecule. This solubility issue necessitates rigorous water washing to remove the phosphorus species, followed by high-temperature distillation to isolate the final product. However, alpha, alpha-dichloroethyl cyclopropane is inherently unstable under acidic conditions and sensitive to thermal stress, meaning that the very purification steps required often lead to decomposition and the formation of difficult-to-remove impurities. Consequently, traditional routes struggle to consistently deliver the high purity demanded by modern pharmaceutical standards, while simultaneously generating substantial wastewater and hazardous waste streams.
The Novel Approach
In stark contrast, the methodology disclosed in CN109678651B fundamentally reengineers the reaction environment to turn a liability into an asset. By introducing N,N-disubstituted amides such as N,N-dimethylformamide or N-methyl-N-phenylformamide directly into the reaction vessel, the process eliminates the need for external inert solvents entirely. As the reaction proceeds between cyclopropyl methyl ketone and phosphorus pentachloride, the generated phosphorus oxychloride immediately reacts with the amide to form a Vilsmeier reagent. This transformation is critical because the resulting Vilsmeier complex is immiscible with the organic product layer, leading to spontaneous phase separation upon standing. This elegant solution allows for the direct removal of the lower by-product layer without any aqueous workup or thermal distillation, preserving the structural integrity of the heat-sensitive product and ensuring exceptional purity profiles right out of the reactor.
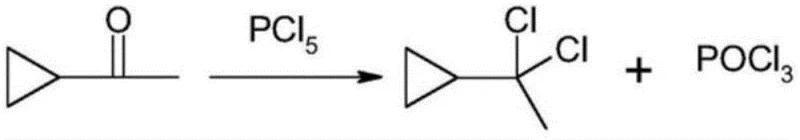
Mechanistic Insights into Solvent-Free Chlorination and Phase Separation
The mechanistic elegance of this process relies on the dual functionality of the N,N-disubstituted amide, which acts not merely as a medium but as a reactive scavenger for the chlorination by-products. Upon the slow addition of phosphorus pentachloride at controlled temperatures ranging from -25°C to 40°C, the primary chlorination of the ketone occurs rapidly to yield the desired alpha, alpha-dichloroethyl cyclopropane. Simultaneously, the phosphorus oxychloride generated as a stoichiometric by-product engages in a rapid condensation reaction with the amide present in the system. This secondary reaction converts the corrosive and soluble POCl3 into a stable, ionic Vilsmeier-Haack type complex. The formation of this complex drastically alters the polarity and density of the by-product phase, rendering it chemically distinct from the non-polar organic product layer. This differential solubility is the key driver for the high purity achieved, as it physically isolates the product from acidic contaminants that would otherwise catalyze degradation pathways during storage or further processing.
Furthermore, the control of impurities is intrinsically built into the thermodynamics of the phase separation. In traditional solvent-based systems, trace amounts of phosphorus species often remain entrained in the organic phase, requiring multiple wash cycles that increase water usage and risk hydrolysis of the product. In this novel solvent-free system, the complete ionization of the Vilsmeier reagent within the amide matrix ensures that virtually all phosphorus-containing species are sequestered in the lower layer. The upper layer, consisting almost exclusively of the target alpha, alpha-dichloroethyl cyclopropane, can be decanted or separated with minimal mechanical intervention. This mechanism not only simplifies the unit operations but also ensures that the final product is free from the acidic residues that typically plague batch chlorination reactions, thereby enhancing the stability and shelf-life of the intermediate for downstream coupling reactions.
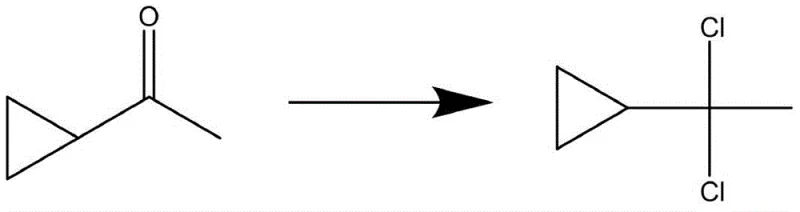
How to Synthesize Alpha, Alpha-Dichloroethyl Cyclopropane Efficiently
Implementing this advanced synthesis route requires precise control over stoichiometry and environmental conditions to maximize the formation of the separable Vilsmeier phase. The process begins with the charging of cyclopropyl methyl ketone and the selected N,N-disubstituted amide into a dry reactor, followed by a rigorous cycle of vacuum and nitrogen replacement to ensure an anhydrous and anaerobic environment. Phosphorus pentachloride is then introduced slowly to manage the exotherm, maintaining the reaction temperature within the optimal window of -25°C to 40°C depending on the specific amide used. Once the addition is complete, a holding period allows the reaction to reach completion and the phases to fully differentiate based on density and polarity. For detailed operational parameters, stoichiometry ratios, and specific workup instructions, please refer to the standardized synthesis guide below.
- Mix cyclopropyl methyl ketone with N,N-disubstituted amide in a reactor under anhydrous and anaerobic conditions.
- Slowly add phosphorus pentachloride at temperatures between -25°C and 40°C, reacting for 1-3 hours.
- Allow the mixture to stand for 2 hours to separate layers, then collect the upper organic layer containing the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this solvent-free methodology offers profound economic and logistical benefits that extend far beyond simple yield improvements. The elimination of inert organic solvents represents a direct reduction in raw material procurement costs and removes the associated expenses of solvent recovery or disposal. Furthermore, the ability to isolate the product through simple phase separation rather than energy-intensive fractional distillation significantly lowers utility consumption and reduces the thermal load on production facilities. This simplification of the workflow translates into shorter batch cycle times, allowing manufacturing plants to increase throughput without requiring additional capital investment in reactor capacity. Additionally, the generation of a saleable Vilsmeier reagent by-product creates a potential secondary revenue stream, effectively offsetting the cost of the amide reagent and improving the overall gross margin of the production campaign.
- Cost Reduction in Manufacturing: The economic impact of removing solvent usage and distillation steps cannot be overstated in large-scale chemical production. By operating without inert solvents, the facility eliminates the need for vast quantities of volatile organic compounds, reducing both purchase costs and the regulatory burden associated with VOC emissions. The avoidance of high-temperature distillation is particularly valuable given the thermal instability of the product; traditional methods often suffer from yield losses due to decomposition during purification, whereas this method preserves the product integrity through mild physical separation. Moreover, the conversion of a waste product (POCl3) into a marketable commodity (Vilsmeier reagent) transforms a disposal cost into a value-added output, driving substantial cost savings across the entire value chain.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on process robustness and the minimization of failure points. Traditional chlorination processes involving aqueous workups are prone to variability due to emulsion formation or incomplete phase separation, which can delay batch release and disrupt delivery schedules. The deterministic nature of the phase separation in this novel process ensures consistent batch-to-batch quality and predictable cycle times. Because the process avoids water washing, there is no risk of product hydrolysis or the generation of large volumes of acidic wastewater that require specialized treatment before discharge. This reliability allows supply chain planners to forecast production outputs with greater accuracy, ensuring continuous availability of this critical intermediate for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this technology inherently mitigates those concerns through design. The absence of flammable solvents reduces the fire hazard profile of the reactor, while the low-temperature operation minimizes the risk of thermal runaway. From an environmental perspective, the complete avoidance of the "three wastes" (wastewater, waste gas, and solid residue) associated with traditional workups aligns perfectly with increasingly stringent global environmental regulations. The process generates no acidic wastewater requiring neutralization, and the containment of phosphorus species within the reusable or sellable by-product layer ensures a cleaner production footprint. This green chemistry approach facilitates easier permitting for capacity expansion and positions the manufacturer as a sustainable partner for multinational corporations with strict ESG mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on how this technology resolves historical pain points in chlorination chemistry. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial manufacturing.
Q: How does the novel method improve purity compared to conventional solvent-based routes?
A: The novel method utilizes N,N-disubstituted amides to react with the by-product phosphorus oxychloride, forming a Vilsmeier reagent that separates into a distinct lower layer. This eliminates the need for water washing and high-temperature distillation, preventing product decomposition and achieving purity greater than 99%.
Q: What are the safety advantages of this solvent-free chlorination process?
A: By avoiding inert organic solvents and eliminating the high-temperature distillation step required in traditional methods, the process significantly reduces thermal risks. The product, which is unstable under acidic and heated conditions, is isolated via simple phase separation at mild temperatures (-25°C to 40°C).
Q: Can the by-products generated in this reaction be utilized commercially?
A: Yes, the lower layer formed after standing contains the Vilsmeier reagent, which is a valuable chemical intermediate itself. This by-product can be directly separated and sold, adding an additional revenue stream and improving the overall atom economy of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha, Alpha-Dichloroethyl Cyclopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex intermediates like alpha, alpha-dichloroethyl cyclopropane requires more than just a patent; it demands deep process engineering expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of solvent-free synthesis are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the absence of residual phosphorus species and solvent traces, guaranteeing that every batch meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to technical excellence ensures that the transition from laboratory scale to industrial production is seamless, safe, and economically viable.
We invite global partners to leverage our technical capabilities to optimize their supply chains for this critical intermediate. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this advanced manufacturing route for your specific volume requirements. We encourage interested parties to contact our technical procurement team to request specific COA data from our pilot runs and to discuss detailed route feasibility assessments tailored to your project timelines. Let us help you secure a stable, high-quality supply of alpha, alpha-dichloroethyl cyclopropane that drives efficiency and innovation in your final products.
