Revolutionizing 2-Chloro-3-methylbenzoic Acid Production via Catalytic Oxidation for Global Supply Chains
Introduction to Advanced Synthetic Pathways for Chlorobenzoic Acids
The global demand for high-purity chlorobenzoic acid derivatives, particularly 2-chloro-3-methylbenzoic acid, has surged due to their critical role as key intermediates in the synthesis of next-generation anthranilic diamide insecticides and various pharmaceutical agents. However, the historical reliance on inefficient and environmentally burdensome synthetic routes has created significant bottlenecks in the supply chain for reliable agrochemical intermediate suppliers. A groundbreaking technical disclosure found in patent CN108530297B introduces a transformative manufacturing protocol that fundamentally alters the economic and ecological landscape of producing this vital molecule. This patent details a robust, three-stage continuous process that leverages catalytic aerobic oxidation, replacing legacy methods plagued by low atom utilization and hazardous reagents. By shifting the paradigm from stoichiometric oxidants and cryogenic organometallics to a catalytic system driven by molecular oxygen, this technology offers a pathway to drastically simplified post-treatment and superior product purity. For procurement managers and R&D directors alike, understanding the mechanistic nuances of this innovation is essential for securing long-term supply continuity and achieving substantial cost reduction in agrochemical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-chloro-3-methylbenzoic acid has been dominated by two primary methodologies, both of which present severe drawbacks for modern large-scale production. The first, known as the diazo hydrogenation method, involves a tedious four-step sequence starting from 2-nitro-3-methyl benzoic acid. As illustrated in the reaction scheme below, this route necessitates esterification, palladium-catalyzed hydrogenation, diazotization with sodium nitrite, and final hydrolysis.
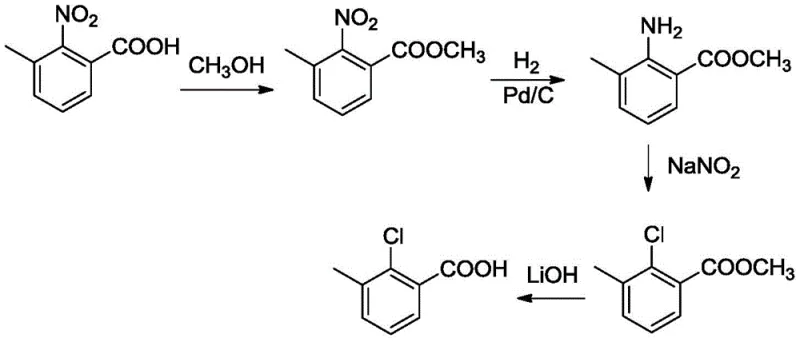
The cumulative yield of this legacy process is abysmal, totaling merely 47.2%, primarily due to the inherent losses in the diazotization step which typically achieves only 70% conversion. Furthermore, the generation of massive quantities of acidic wastewater containing nitrite salts creates an overwhelming environmental compliance burden. The second conventional approach utilizes a butyl lithium strategy, which, while avoiding nitrogenous waste, introduces extreme safety hazards. This method requires the use of sec-butyl lithium at ultra-low temperatures, demanding specialized cryogenic reactors and rigorous safety protocols to prevent thermal runaway or fire.
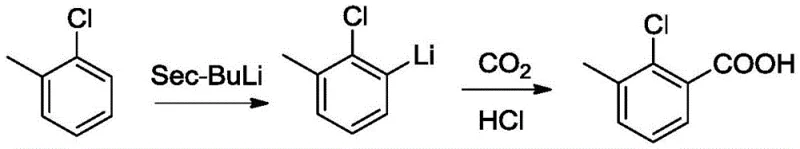
Despite these stringent requirements, the butyl lithium method suffers from poor regioselectivity, leading to a product yield of only 15% and significant formation of isomeric impurities that are difficult to separate. Both traditional routes fail to meet the modern standards for green chemistry, resulting in high production costs, complex waste streams, and unreliable supply chains for high-purity agrochemical intermediates.
The Novel Approach
In stark contrast to these antiquated techniques, the novel approach disclosed in the patent utilizes a direct catalytic oxidation of 2,6-dimethylchlorobenzene, a commercially abundant and inexpensive feedstock. This innovative route bypasses the need for nitrogen-containing precursors and cryogenic organometallic reagents entirely. The core of this breakthrough lies in a cobalt-catalyzed aerobic oxidation system that operates under moderate pressure and temperature conditions. By carefully controlling the oxygen partial pressure between 0.1 MPa and 0.8 MPa, the process achieves exceptional selectivity for the mono-carboxylic acid product, effectively suppressing the formation of the undesired di-acid byproduct (2,6-dicarboxychlorobenzene). This method not only simplifies the reaction engineering by eliminating the need for complex protection-deprotection sequences but also enables a solvent-free or low-solvent operation in subsequent steps. The result is a streamlined manufacturing process that significantly enhances atom economy and reduces the environmental footprint, making it an ideal solution for the commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into Cobalt-Catalyzed Aerobic Oxidation
The heart of this technological advancement is the cobalt-catalyzed oxidation mechanism, which facilitates the selective conversion of a methyl group to a carboxylic acid functionality in the presence of a chlorine substituent. The reaction proceeds via a free-radical chain mechanism initiated by the cobalt catalyst, typically cobalt acetylacetonate or cobalt acetate, in conjunction with a bromide promoter such as potassium bromide. The bromide ion plays a critical role in generating bromine radicals that abstract hydrogen atoms from the benzylic methyl group, forming a stable benzyl radical intermediate. This radical subsequently reacts with molecular oxygen to form a peroxy radical, which propagates the chain reaction and eventually leads to the formation of the aldehyde and finally the carboxylic acid. The precise control of oxygen pressure is paramount; maintaining the pressure within the optimal range of 0.3 MPa to 0.5 MPa ensures that the concentration of dissolved oxygen is sufficient to drive the oxidation forward without promoting over-oxidation to the di-acid species.
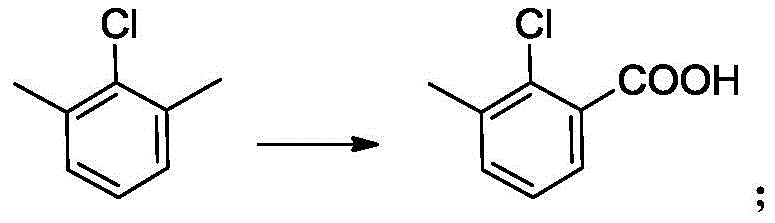
Furthermore, the presence of the chlorine atom on the aromatic ring exerts an electronic effect that influences the reactivity of the adjacent methyl groups, requiring fine-tuning of the catalyst loading and reaction temperature (70-75°C) to achieve high conversion. The patent highlights that the catalyst system remains dissolved in the mother liquor after product crystallization, allowing for nearly 90% of the catalyst to be recycled directly into the next batch. This homogeneous catalytic cycle not only minimizes metal waste but also ensures consistent reaction kinetics across multiple production runs. For R&D teams, this implies a highly robust process window where minor fluctuations in feedstock quality can be tolerated without compromising the final purity specifications, thereby ensuring the production of high-purity OLED material or pharmaceutical grade intermediates with minimal batch-to-batch variation.
How to Synthesize 2-Chloro-3-methylbenzoic Acid Efficiently
The implementation of this novel synthesis route involves a sequential three-step protocol that maximizes material throughput while minimizing waste generation. The process begins with the esterification of 2,6-dimethylphenol with phosgene to generate 2,6-dimethyl phenyl chloroformate, followed by a solvent-free decarboxylation to yield the key intermediate 2,6-dimethylchlorobenzene, and concludes with the aforementioned catalytic oxidation. Each step is optimized for catalyst recovery and solvent recycling, creating a closed-loop system that drastically lowers operating expenses. The detailed standardized synthetic steps, including specific molar ratios, temperature profiles, and work-up procedures, are outlined in the comprehensive guide below.
- Perform esterification of 2,6-dimethylphenol with phosgene using a recyclable amine catalyst to form 2,6-dimethyl phenyl chloroformate.
- Execute solvent-free decarboxylation of the chloroformate intermediate using zinc chloride at 100-110°C to yield 2,6-dimethylchlorobenzene.
- Conduct cobalt-catalyzed aerobic oxidation of 2,6-dimethylchlorobenzene at 0.3-0.5 MPa oxygen pressure to obtain the final high-purity acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel catalytic oxidation process represents a strategic opportunity to optimize total cost of ownership and mitigate supply risks. The elimination of expensive and scarce starting materials like 2-nitro-3-methyl benzoic acid, coupled with the removal of hazardous reagents like sec-butyl lithium, fundamentally reshapes the cost structure of production. By utilizing commodity chemicals such as 2,6-dimethylphenol and oxygen, the process insulates the supply chain from the volatility associated with specialty fine chemical markets. Moreover, the ability to recycle catalysts and solvents multiple times without significant loss of activity translates into substantial cost savings in raw material procurement and waste disposal fees. This efficiency gain allows for more competitive pricing models and improved margin stability for downstream customers seeking cost reduction in pharma intermediates manufacturing.
- Cost Reduction in Manufacturing: The novel process eliminates the need for costly noble metal catalysts like palladium on carbon, which are required in the traditional hydrogenation route and necessitate expensive recovery processes to meet residual metal specifications. Instead, the use of earth-abundant cobalt salts, which can be recycled along with the mother liquor, dramatically lowers the catalyst cost per kilogram of product. Additionally, the high selectivity of the oxidation step minimizes the formation of hard-to-remove impurities, reducing the need for energy-intensive recrystallization or chromatographic purification steps. This streamlined downstream processing significantly cuts utility costs and labor hours, driving down the overall manufacturing expense.
- Enhanced Supply Chain Reliability: By relying on widely available industrial feedstocks such as 2,6-dimethylphenol and phosgene, the supply chain becomes far more resilient to disruptions compared to routes dependent on niche nitro-compounds. The robustness of the catalytic system, which tolerates variations in oxygen pressure and maintains high yields over extended periods, ensures consistent production output. This reliability is crucial for reducing lead time for high-purity agrochemical intermediates, allowing manufacturers to respond swiftly to market demand fluctuations without the risk of batch failures or prolonged downtime associated with equipment cleaning and maintenance.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial continuous production in mind. The solvent-free nature of the decarboxylation step and the efficient recycling of solvents in the esterification step significantly reduce the volume of organic waste generated. Furthermore, the avoidance of nitrite salts and heavy metal waste streams simplifies wastewater treatment requirements, ensuring compliance with increasingly stringent environmental regulations. This green chemistry profile not only reduces the environmental impact but also future-proofs the manufacturing facility against potential regulatory changes, securing long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear perspective on the operational benefits and performance metrics of the new route versus legacy methods. Understanding these details is vital for technical teams evaluating the feasibility of adopting this process for commercial production.
Q: How does the new catalytic oxidation method compare to traditional diazotization in terms of yield?
A: The traditional diazotization route suffers from a cumulative yield of only 47.2% across four steps, whereas the novel catalytic oxidation pathway achieves significantly higher single-step yields (up to 95%) and eliminates multiple purification stages, drastically improving overall atom economy.
Q: What are the safety advantages of avoiding the butyl lithium method?
A: The conventional butyl lithium method requires hazardous cryogenic conditions and pyrophoric reagents, posing severe operational risks. The new process operates at moderate temperatures (70-75°C) and pressures (0.3-0.5 MPa), utilizing stable cobalt catalysts and eliminating the need for ultra-low temperature infrastructure.
Q: Can the catalysts and solvents be recycled in this new industrial process?
A: Yes, the process is designed for circular efficiency. The amine catalyst in the esterification step can be reused over 20 times, zinc chloride in decarboxylation over 10 times, and 90% of the oxidation catalyst is recovered directly in the mother liquor, substantially reducing raw material consumption and waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-3-methylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern chemical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the catalytic oxidation of 2,6-dimethylchlorobenzene are translated into reliable commercial reality. Our state-of-the-art facilities are equipped to handle the specific pressure and temperature requirements of this process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for agrochemical and pharmaceutical applications. We are committed to delivering high-purity 2-chloro-3-methylbenzoic acid that supports your downstream synthesis needs with unwavering consistency.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this novel route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective source of this vital intermediate.
