Advanced Catalytic Asymmetric Synthesis of Non-neferitone Intermediates for Commercial Scale-up
Advanced Catalytic Asymmetric Synthesis of Non-neferitone Intermediates for Commercial Scale-up
The pharmaceutical industry is constantly seeking robust, scalable, and cost-effective pathways for synthesizing complex active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is detailed in patent CN114149427A, which discloses a novel synthetic method for non-neferitone and its key intermediates. Non-neferitone, a non-steroidal mineralocorticoid receptor antagonist, is critical for treating chronic kidney disease, yet its historical synthesis has been plagued by inefficiencies. This new methodology replaces traditional, labor-intensive chiral column resolution with a sophisticated chemical chiral resolution strategy. By leveraging specific chiral catalysts and optimized reaction conditions, the process achieves high yields and exceptional stereochemical control. For R&D directors and procurement managers alike, this represents a paradigm shift towards more sustainable and economically viable manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of non-neferitone and similar chiral molecules has relied heavily on chiral column resolution techniques. While effective on a small laboratory scale, these methods present severe bottlenecks for industrial application. The reliance on preparative chiral HPLC columns introduces exorbitant costs associated with the stationary phases and the massive volumes of solvents required for elution. Furthermore, the technical complexity of operating chiral columns often leads to significant batch-to-batch variability and extended processing times. From a supply chain perspective, the low throughput of chiral chromatography limits the ability to meet large-scale commercial demands, creating a fragile supply chain vulnerable to disruptions. Additionally, the recovery of the unwanted enantiomer is often difficult, leading to substantial raw material wastage and increased environmental burden due to solvent disposal.
The Novel Approach
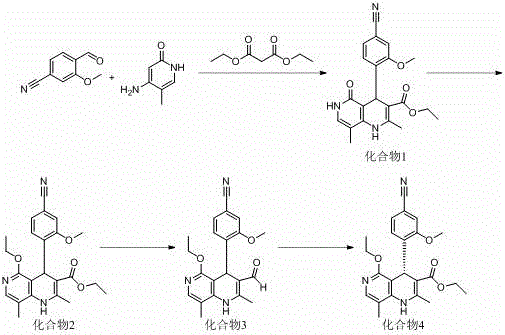
The innovative route presented in the patent data fundamentally alters the manufacturing landscape by introducing a chemical chiral resolution step mediated by specialized catalysts. Instead of physical separation on a column, the process utilizes a dynamic kinetic resolution or asymmetric induction strategy during the formation of Compound 4. As illustrated in the reaction scheme, the pathway flows logically from the condensation of simple starting materials to the final chiral intermediate. This approach allows for the use of standard reactor equipment rather than specialized chromatography systems. The elimination of the chiral column step not only simplifies the operational workflow but also significantly enhances the overall mass balance of the process. By converting the racemic or prochiral precursor directly into the desired enantiomer with high selectivity, the method ensures that the final product meets rigorous purity standards without the prohibitive costs associated with traditional resolution techniques.
Mechanistic Insights into Chiral Catalyst-Mediated Resolution
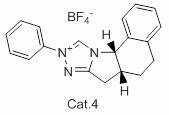
The core of this technological advancement lies in the selection and application of specific chiral catalysts, such as the series denoted as Cat. 1 through Cat. 17 in the patent documentation. In the preferred embodiment, Catalyst 7 (Cat. 7) is utilized to facilitate the stereoselective transformation of Compound 3 into the chiral Compound 4. The mechanism likely involves the coordination of the catalyst with the substrate and the oxidizing agent, creating a chiral environment that favors the formation of one enantiomer over the other. The presence of a base, such as N,N-diisopropylethylamine, and an oxidant like 3,5,3',5'-tetra-tert-butyldicyclohexyl-2,5,2',5'-tetraene-4,4'-dione, suggests an oxidative dynamic kinetic resolution process. This intricate interplay ensures that even if the starting material is racemic, the reaction drives towards the single desired stereoisomer, effectively doubling the theoretical yield compared to classical resolution methods where 50% of the material is discarded.
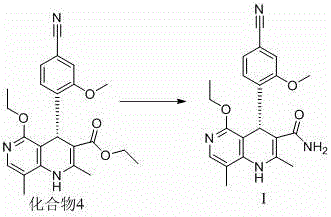
Beyond stereocontrol, the mechanistic design of this route also addresses critical impurity profiles that concern R&D teams. The stepwise progression from Compound 1 to Compound 4 allows for the removal of by-products at intermediate stages, preventing the carryover of impurities into the final API. For instance, the reduction step converting Compound 2 to Compound 3 uses sodium borohydride under controlled temperatures (0°C to 50°C), which minimizes side reactions such as over-reduction or decomposition of sensitive functional groups like the nitrile moiety. The subsequent chiral resolution step is performed in dichloromethane with precise temperature control (heating to 40°C then cooling to 10°C), which promotes the crystallization of the desired enantiomer while keeping impurities in the mother liquor. This level of control results in a final intermediate with an ee value of 99.5% and chemical purity of 98%, demonstrating the robustness of the catalytic system in suppressing the formation of undesired isomers and structural analogs.
How to Synthesize Non-neferitone Intermediate Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with a solvent-free or inert solvent condensation to form the scaffold, followed by alkylation and reduction. The critical final transformation involves the chiral catalyst. Operators must maintain an inert atmosphere, typically nitrogen, especially during the reduction and chiral resolution steps to prevent oxidation of sensitive intermediates. The use of specific molar ratios, such as a catalyst loading of 0.02-0.5:1 relative to the substrate, is vital for economic efficiency. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring that technical teams can replicate the high yields and purity reported.
- Condense diethyl malonate, 4-cyano-2-methoxybenzaldehyde, and 4-amino-5-methyl-2-hydroxypyridine using acetic acid at 80°C to form Compound 1.
- Perform alkylation on Compound 1 with triethyl orthoformate and sulfuric acid catalyst under solvent-free conditions to yield Compound 2.
- Reduce Compound 2 using sodium borohydride in tetrahydrofuran at controlled temperatures (0°C to 50°C) to obtain Compound 3.
- Execute chiral resolution of Compound 3 using a specialized chiral catalyst (e.g., Cat. 7) and ethanol to isolate the optically active Compound 4.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage is the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures (OPEX). By removing the need for expensive chiral chromatography columns and the associated solvent recovery infrastructure, the capital expenditure (CAPEX) required for setting up production lines is significantly lowered. Furthermore, the ability to perform key steps, such as the initial condensation and alkylation, under solvent-free conditions reduces the volume of hazardous waste generated, aligning with increasingly strict environmental regulations and lowering disposal costs. This streamlined approach ensures a more reliable supply of high-purity intermediates, mitigating the risk of production delays caused by equipment bottlenecks or raw material shortages.
- Cost Reduction in Manufacturing: The elimination of chiral column resolution is the most significant cost-saving driver in this new process. Traditional chiral separation consumes vast amounts of solvents and requires specialized, high-maintenance equipment, all of which contribute to a high cost per kilogram of the final product. By switching to a chemical chiral resolution method using recoverable or low-loading catalysts, the process inherently lowers the variable costs associated with production. Additionally, the high yields reported in the patent examples (e.g., 86.2% for Compound 1 and 91% for Compound 2) indicate efficient atom economy, meaning less raw material is wasted. This efficiency translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for non-neferitone and related therapeutic agents.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available and stable starting materials such as diethyl malonate and 4-cyano-2-methoxybenzaldehyde. Unlike proprietary chiral auxiliaries that may have limited suppliers, the reagents used in this pathway are commodity chemicals with robust global supply networks. The simplified post-treatment procedures, which involve standard extraction and crystallization techniques rather than complex chromatographic separations, reduce the dependency on highly specialized technical labor. This ease of operation means that production can be scaled across multiple manufacturing sites without significant retraining or technology transfer hurdles, ensuring continuity of supply even in the face of regional disruptions or logistical challenges.
- Scalability and Environmental Compliance: The process is explicitly designed with industrial scale-up in mind, addressing a common pain point in pharmaceutical manufacturing. The ability to run reactions like the initial condensation without solvent or with minimal solvent reduces the reactor volume required per unit of product, effectively increasing the throughput of existing facilities. Moreover, the reduction in solvent usage and the avoidance of heavy metal catalysts (using organic chiral catalysts instead) simplifies waste management and regulatory compliance. The process generates less hazardous waste, making it easier to meet environmental, health, and safety (EHS) standards. This 'green chemistry' aspect not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturer, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within patent CN114149427A, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for commercial production. The clarity provided here aims to bridge the gap between patent theory and practical industrial application.
Q: How does this new synthesis method improve upon traditional chiral column resolution?
A: Traditional methods rely on expensive chiral HPLC columns which are technically demanding and have low throughput. This patent utilizes a chemical chiral resolution method with specific catalysts (like Cat. 7), eliminating the need for preparative chiral chromatography, thereby drastically reducing raw material loss and simplifying post-treatment.
Q: What are the purity and enantiomeric excess (ee) specifications achievable with this route?
A: The process described in patent CN114149427A demonstrates high efficiency, achieving a chemical purity of 98% for the intermediate Compound 4 and an enantiomeric excess (ee) value of up to 99.5%, meeting stringent requirements for pharmaceutical registration.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is designed for industrial scalability. Key steps, such as the initial condensation and alkylation, can be performed under solvent-free conditions or with common inert solvents, and the avoidance of complex chiral separation equipment facilitates easier scale-up from kilogram to tonnage levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Non-neferitone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN114149427A for the production of non-neferitone intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative chemistry to life. Our state-of-the-art facilities are equipped to handle the specific requirements of this process, including inert atmosphere reactions and precise temperature control for chiral resolution. We are committed to delivering products with stringent purity specifications and rigorous QC labs to ensure that every batch meets the exacting standards of the global pharmaceutical industry. Our team of expert chemists is ready to optimize this route further, ensuring maximum efficiency and yield for your specific commercial needs.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective, and high-quality supply of non-neferitone intermediates, ensuring your drug development programs proceed without interruption.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
