Advanced Synthesis of Deuterated Diazepam Intermediates for Forensic and Pharmaceutical Applications
Advanced Synthesis of Deuterated Diazepam Intermediates for Forensic and Pharmaceutical Applications
The global demand for high-purity deuterated standards in forensic toxicology and pharmaceutical analysis has created a critical niche for advanced chemical synthesis capabilities. Patent CN107501196B, published in early 2020, introduces a robust and innovative methodology for preparing key intermediates used in the synthesis of Diazepam-D5 and Diazepam-D8. This technology addresses a significant gap in the domestic production of deuterated substances, which have historically been reliant on expensive imports often supplied only as unstable methanol solutions rather than stable solid powders. By utilizing 6-chloro-2-methyl-4H-3,1-benzoxazin-4-one as a strategic starting material, the disclosed process enables the efficient construction of the benzodiazepine core with precise deuteration patterns. This breakthrough not only enhances the availability of essential reference materials for law enforcement and clinical laboratories but also establishes a new benchmark for quality in the production of isotopically labeled pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the procurement of deuterated benzodiazepine standards has been fraught with logistical and technical challenges that hinder widespread analytical application. Most commercially available options were imported as methanol solutions, which suffer from inherent stability issues over long storage periods, leading to concentration drifts that compromise the accuracy of quantitative analysis. Furthermore, the reliance on foreign suppliers created a bottleneck in supply chain continuity, particularly for sensitive forensic applications where immediate access to certified reference materials is mandatory. The high cost associated with these imported standards, driven by complex logistics and limited competition, severely restricted their adoption in routine screening protocols. Additionally, conventional synthetic routes often involved harsh reaction conditions or multi-step sequences with poor atom economy, resulting in lower overall yields and difficult purification processes that introduced isotopic dilution or impurities detrimental to mass spectrometric performance.
The Novel Approach
The methodology outlined in the patent represents a paradigm shift towards a more sustainable and efficient production model for deuterated intermediates. By employing a convergent synthesis strategy centered on a Grignard addition followed by a streamlined cyclization sequence, the process achieves high chemical purity with significantly simplified operational steps. The use of readily available reagents such as deuterated bromobenzene to generate the Grignard species allows for precise control over the isotopic labeling position, ensuring the final product meets the rigorous specifications required for internal standards. This approach eliminates the need for unstable solution formulations by yielding a stable solid powder that can be easily weighed and dissolved as needed, thereby extending shelf life and reducing waste. The mild reaction conditions, particularly the ambient temperature cyclization using ammonia gas, further underscore the practicality of this method for large-scale manufacturing while maintaining the structural integrity of the sensitive diazepine ring system.
Mechanistic Insights into Grignard Addition and Cyclization
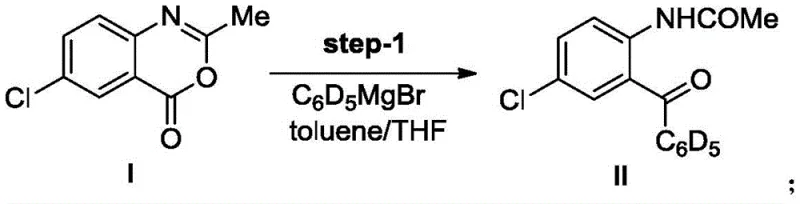
The cornerstone of this synthesis lies in the initial nucleophilic attack of the deuterated phenyl Grignard reagent on the benzoxazinone ring, a transformation that sets the stereochemical and isotopic foundation for the entire molecule. In Step 1, the reaction between 6-chloro-2-methyl-4H-3,1-benzoxazin-4-one and C6D5MgBr proceeds via a highly selective addition to the carbonyl carbon, followed by ring opening and subsequent rearrangement to form the acetamide intermediate. This step is critical as it introduces the deuterated phenyl ring which serves as the isotopic label for the final diazepam analog. The choice of toluene and THF as co-solvents optimizes the solubility of both the organometallic reagent and the organic substrate, facilitating a smooth reaction profile at low temperatures (0°C) which minimizes side reactions such as over-addition or proton exchange that could dilute the deuterium content.
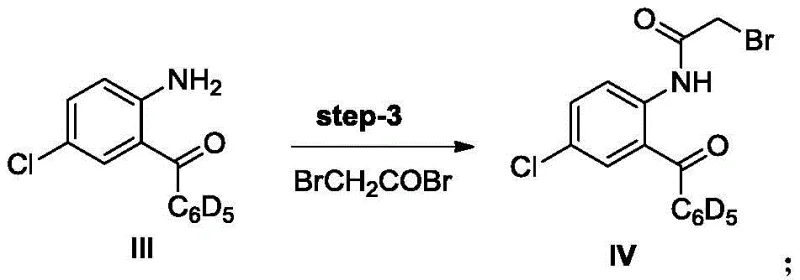
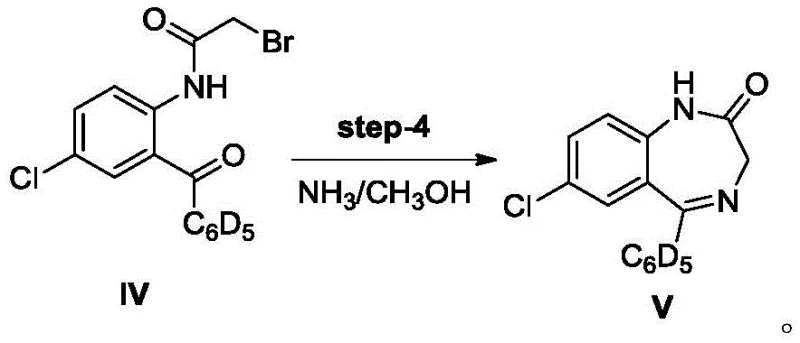
Following the formation of the ketone backbone, the subsequent acylation and cyclization steps demonstrate elegant control over regioselectivity and ring closure. In Step 3, the amino group of the benzophenone derivative is selectively acylated with bromoacetyl bromide in the presence of a tertiary amine base, creating the necessary leaving group for the final ring closure. The mechanism involves the nucleophilic attack of the amine nitrogen on the acid bromide, forming an amide bond while the base scavenges the generated HBr to drive the equilibrium forward. Finally, Step 4 utilizes ammonia gas in methanol to induce an intramolecular nucleophilic substitution where the amide nitrogen attacks the alpha-carbon of the bromoacetyl group. This cyclization closes the seven-membered diazepine ring, releasing bromide and establishing the characteristic 1,4-benzodiazepin-2-one scaffold with high fidelity.
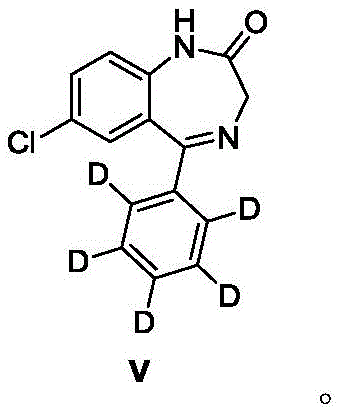
How to Synthesize 7-Chloro-1,3-dihydro-5-(phenyl-d5)-2H-1,4-benzodiazepin-2-one Efficiently
Executing this synthesis requires careful attention to moisture exclusion and temperature control, particularly during the Grignard formation and addition phases. The process begins with the in-situ generation of the deuterated Grignard reagent, followed by its controlled addition to the benzoxazinone substrate to ensure complete conversion without exothermic runaway. Subsequent hydrolysis and acylation steps utilize standard workup procedures involving aqueous washes and organic extractions, which are familiar to process chemists and easily adaptable to pilot plant scales. The final cyclization step is notably user-friendly, requiring only a sealed vessel and ammonia gas pressure, avoiding the need for hazardous high-pressure autoclaves or exotic catalysts. For a comprehensive, step-by-step guide including exact molar ratios, solvent volumes, and purification parameters, please refer to the standardized protocol below.
- React 6-chloro-2-methyl-4H-3,1-benzoxazin-4-one with deuterated phenylmagnesium bromide (C6D5MgBr) in toluene/THF at 0°C to form the acetamide intermediate.
- Hydrolyze the acetamide intermediate using NaOH in ethanol under reflux to remove the protecting group and yield the amino-ketone.
- Acylate the amino group with bromoacetyl bromide in the presence of a base like triethylamine to form the bromoacetamide precursor.
- Perform cyclization by reacting the bromoacetamide with ammonia gas in methanol at 50°C to close the diazepine ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented technology offers substantial benefits that directly address the pain points of supply chain volatility and cost inefficiency in the specialty chemicals sector. By enabling the domestic production of high-value deuterated intermediates, organizations can mitigate the risks associated with international shipping delays, customs clearance hurdles, and currency fluctuations that often plague the import of forensic standards. The ability to source these materials as stable solid powders rather than solutions significantly reduces transportation costs and hazards, as solids generally have longer shelf lives and do not require special containment for volatile solvents. Furthermore, the simplified synthetic route reduces the number of unit operations and purification stages, which translates to lower manufacturing overheads and a smaller environmental footprint through reduced solvent consumption and waste generation.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the use of commodity chemicals like toluene, ethanol, and ammonia drastically lowers the raw material costs associated with producing deuterated standards. By avoiding the need for expensive chromatographic separations typically required to remove isotopic impurities, the process achieves a more favorable cost structure that can be passed down to the end-user. The high yield reported in the patent examples suggests that material throughput is maximized, reducing the cost per gram of the final active pharmaceutical ingredient (API) intermediate. This economic efficiency makes it feasible to produce larger batches, realizing economies of scale that were previously unattainable with legacy import-dependent models.
- Enhanced Supply Chain Reliability: Establishing a local or regional supply base for these critical intermediates ensures business continuity for forensic laboratories and pharmaceutical manufacturers who cannot afford stockouts. The robustness of the synthesis, characterized by mild conditions and tolerance to minor variations in reagent quality, means that production schedules are less likely to be disrupted by batch failures. This reliability is paramount for maintaining the accreditation and operational readiness of testing facilities that rely on consistent availability of certified reference materials. Moreover, the flexibility of the process allows for rapid scale-up from laboratory to commercial quantities, ensuring that sudden spikes in demand can be met without lengthy lead times.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing the use of hazardous reagents and optimizing solvent recovery opportunities. The final cyclization step, which generates ammonium bromide as a byproduct, produces waste streams that are easier to treat and dispose of compared to heavy metal catalyst residues found in alternative cross-coupling methods. This alignment with environmental regulations reduces the compliance burden on manufacturing sites and lowers the costs associated with waste disposal. The scalability is further evidenced by the use of standard reactor types and agitation methods, making the technology transfer to multi-ton production facilities straightforward and low-risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these deuterated intermediates. Understanding the nuances of isotopic purity and stability is essential for stakeholders involved in quality control and regulatory affairs. The answers provided are derived directly from the technical specifications and experimental data disclosed in the patent literature, ensuring accuracy and relevance for professional users.
Q: What are the key advantages of this deuterated diazepam intermediate synthesis?
A: The process described in patent CN107501196B offers mild reaction conditions, simple operation, and produces intermediates with high chemical purity and excellent stability, suitable for preparing forensic standards.
Q: Why is high-purity deuterated internal standard critical for forensic analysis?
A: Deuterated internal standards compensate for matrix effects and instrument variability in mass spectrometry, ensuring accurate quantification of diazepam in complex biological samples.
Q: Can this synthesis route be scaled for commercial production?
A: Yes, the use of common solvents like toluene, ethanol, and chloroform, along with standard reagents like Grignard reagents and ammonia, indicates strong potential for scalable commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Chloro-1,3-dihydro-5-(phenyl-d5)-2H-1,4-benzodiazepin-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of accurate diagnostic tools and therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify isotopic enrichment and chemical identity. Our commitment to quality assurance means that every batch of deuterated intermediate we supply is accompanied by comprehensive documentation, supporting your regulatory filings and quality audits with confidence.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our optimized process can reduce your total cost of ownership. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to secure a reliable supply chain for your most critical chemical building blocks.
