Advanced Green Synthesis Of Isoargentine: A Technical Breakthrough For Commercial Scale-Up
The landscape of agrochemical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways. A pivotal development in this sector is detailed in patent CN111233815B, which discloses a novel preparation method for isoargentine (isoiridomyrmecin), a critical iridoid compound utilized extensively as an insect semiochemical and in biological assays. This patent represents a substantial leap forward from legacy methodologies, addressing long-standing issues regarding toxicity and yield that have historically plagued the production of this valuable pheromone. By shifting away from hazardous heavy metal reagents toward a catalytic hydrogenation and mild oxidation strategy, this technology offers a robust framework for industrial application. For R&D directors and procurement specialists alike, understanding the nuances of this pathway is essential for securing a reliable agrochemical intermediate supplier capable of meeting stringent purity and sustainability standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in the patent data, the synthesis of isoargentine was fraught with significant chemical and operational challenges that hindered efficient cost reduction in agrochemical intermediate manufacturing. Historical approaches, such as those documented by the Hofferberth group, relied heavily on the use of mercury acetate [Hg(OAc)2] for hydroxymercuration-reduction reactions. This reliance on mercury introduced severe environmental liabilities, requiring complex waste treatment protocols to handle toxic heavy metal byproducts. Furthermore, these legacy routes were characterized by cumbersome multi-step sequences involving disulfide protection and harsh oxidative conditions, which collectively resulted in suboptimal overall yields, often hovering around merely 30%. The low selectivity and conversion rates associated with these mercury-mediated processes not only inflated raw material costs but also complicated the purification of the final product, making large-scale production economically unviable and environmentally unsustainable.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a streamlined, three-step synthetic route that elegantly bypasses the need for toxic mercury reagents. The process initiates with a nucleophilic addition reaction where the starting material, 4,7-dimethyl-1,4a,5,6,7,7a-hexahydrocyclopentapyran, is treated with benzyl alcohol, trichloroacetonitrile, and a silicon reagent to form a protected intermediate. This is followed by a catalytic hydrogenation step using a palladium reagent to remove the benzyl group, and finally, a mild oxidation using TPAP and NMO to yield the target lactone. This strategic redesign not only simplifies the operational workflow but also dramatically enhances the environmental profile of the synthesis. The elimination of mercury aligns perfectly with modern green chemistry principles, while the improved yield profile (increasing to 60-71%) ensures better resource utilization. This approach effectively solves the scalability bottleneck, enabling the commercial scale-up of complex pheromones with greater economic efficiency.
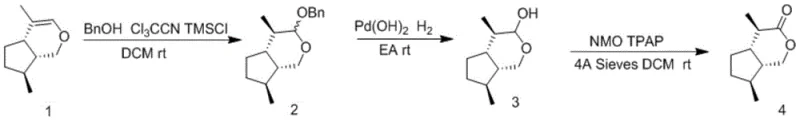
Mechanistic Insights into Catalytic Hydrogenation and TPAP Oxidation
The core of this technological advancement lies in its sophisticated yet practical mechanistic design, particularly in the final oxidation stage. The use of Tetrapropylammonium Perruthenate (TPAP) in conjunction with N-Methylmorpholine N-oxide (NMO) serves as a highly selective catalytic system for oxidizing the primary alcohol intermediate to the corresponding lactone. Unlike stoichiometric oxidants that generate vast amounts of inorganic waste, the TPAP/NMO system operates catalytically, where NMO acts as the co-oxidant to regenerate the active ruthenium species. This mechanism ensures that the oxidation proceeds under exceptionally mild conditions, typically at room temperature, preserving the delicate stereochemical integrity of the iridoid skeleton. For R&D teams, this implies a significant reduction in side reactions and degradation products, directly translating to a cleaner crude reaction profile and simplified downstream processing.
Furthermore, the stereochemical outcome of this synthesis is a critical differentiator for high-value applications. The patent data highlights that the rotation of the methyl group at the C9 position allows for the formation of diastereomers, with the R-isomer being the biologically active target. While conventional methods struggle to achieve high diastereomeric excess, often yielding only 30% of the desired R-type, this novel pathway leverages the specific steric environment created during the benzyl protection and subsequent hydrogenation steps to favor the formation of the R-isomer, achieving levels between 70-80%. This inherent selectivity reduces the need for expensive and yield-loss-inducing chiral separation techniques later in the pipeline. By controlling impurities at the source through precise mechanistic tuning, the process delivers high-purity isoargentine that meets the rigorous specifications required for bioassay and pest control applications.
How to Synthesize Isoargentine Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and safety. The process is designed to be operationally simple, utilizing common laboratory and industrial solvents such as dichloromethane, ethyl acetate, and alcohols, which facilitates easy technology transfer from lab to plant. The following guide outlines the standardized procedural flow derived from the patent examples, emphasizing the critical control points for temperature and reagent stoichiometry. For detailed standard operating procedures and specific batch records, please refer to the technical documentation provided below.
- Perform nucleophilic addition on 4,7-dimethyl-1,4a,5,6,7,7a-hexahydrocyclopentapyran using benzyl alcohol and trichloroacetonitrile with a silicon reagent to form the protected intermediate.
- Execute catalytic hydrogenation using a palladium reagent under hydrogen atmosphere to remove the benzyl protecting group and generate the hemiacetal precursor.
- Conduct final oxidation using N-methylmorpholine oxide (NMO) and tetrapropylammonium perruthenate (TPAP) with molecular sieves to yield the target isoargentine lactone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the supply chain risk profile. By eliminating the dependency on mercury-based reagents, which are subject to increasingly stringent global regulations and handling restrictions, manufacturers can avoid potential supply disruptions and regulatory compliance penalties. Additionally, the shift to room temperature reactions across all three steps eliminates the need for energy-intensive heating or cryogenic cooling infrastructure. This thermal efficiency translates directly into lower utility costs and reduced carbon footprint, aligning with corporate sustainability goals without compromising output quality.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substantial improvement in overall yield, which more than doubles compared to prior art methods. Higher yields mean less raw material is required per kilogram of finished product, directly lowering the Cost of Goods Sold (COGS). Furthermore, the removal of toxic mercury reagents eliminates the significant costs associated with hazardous waste disposal and specialized containment equipment. The use of catalytic amounts of TPAP rather than stoichiometric oxidants further optimizes reagent costs, ensuring a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes significantly to reducing lead time for high-purity agrochemical intermediates. Because the reaction conditions are mild and tolerant of various solvent systems (including ethers, esters, and halogenated hydrocarbons), the process is less susceptible to batch failures caused by minor fluctuations in temperature or mixing. The availability of starting materials and reagents like benzyl alcohol and palladium on carbon is high, ensuring a stable supply base. This reliability allows for more accurate production planning and inventory management, minimizing the risk of stockouts for critical downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new engineering challenges, but this methodology is inherently scalable due to its simplicity and safety profile. The absence of highly exothermic steps or toxic gas evolution simplifies reactor design and safety protocols. From an environmental perspective, the process generates significantly less hazardous waste, easing the burden on effluent treatment plants and ensuring compliance with international environmental standards such as REACH. This 'green' credential is increasingly becoming a prerequisite for doing business with major multinational agrochemical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of isoargentine using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on performance metrics and operational feasibility. Understanding these details is crucial for stakeholders evaluating the viability of integrating this technology into their existing production portfolios.
Q: How does the new synthesis method improve upon previous mercury-based routes?
A: The new method eliminates the use of toxic mercury acetate reagents required in prior art, replacing them with a greener benzyl protection and palladium-catalyzed hydrogenation strategy. This significantly reduces environmental hazards and waste disposal costs while improving overall yield from approximately 30% to over 60%.
Q: What is the stereoselectivity achieved in this patented process?
A: The process demonstrates superior stereocontrol compared to conventional methods, achieving an R-isomer content of 70-80%, whereas traditional techniques typically yield only around 30% of the desired R-type isomer, reducing the burden on downstream purification.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, all reaction steps proceed under mild conditions, primarily at room temperature (15-35°C), without the need for extreme heating or cryogenic cooling. This thermal efficiency, combined with the use of common solvents like dichloromethane and ethyl acetate, facilitates straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoargentine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic technologies to maintain a competitive edge in the global market. Our technical team has thoroughly analyzed the pathway described in CN111233815B and possesses the expertise to implement this green, mercury-free synthesis at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of isoargentine delivered meets the highest standards of quality and consistency required for sensitive bioassay and pheromone applications.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and reliability for your organization.
