Advanced Chiral Phosphine Amide Ester Catalysts for Efficient Asymmetric Henry Reactions
Advanced Chiral Phosphine Amide Ester Catalysts for Efficient Asymmetric Henry Reactions
The landscape of asymmetric synthesis is constantly evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is documented in patent CN102382140A, which introduces a novel class of chiral phosphine amide ester compounds designed to serve as highly efficient organocatalysts. These compounds, specifically characterized as 4H-1,3,2-benzoxazaphosphorin-4-one derivatives, represent a sophisticated fusion of phosphorus chemistry and chiral oxazoline motifs. For R&D directors and process chemists, the introduction of such robust chiral ligands offers a promising avenue to overcome the limitations of traditional transition metal catalysis, particularly in carbon-carbon bond-forming reactions. The strategic design of these molecules allows for precise stereochemical control, which is paramount when synthesizing complex active pharmaceutical ingredients (APIs) where even minor impurities can compromise safety and efficacy. By leveraging the unique electronic and steric properties of the phosphorus-oxazoline framework, manufacturers can achieve superior reaction outcomes that were previously difficult or costly to attain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the execution of asymmetric Henry reactions and similar nucleophilic additions has relied heavily on transition metal complexes involving precious metals such as palladium, iridium, or copper. While effective in many scenarios, these conventional methods often suffer from significant drawbacks that impact both the economic and environmental viability of large-scale manufacturing processes. One primary concern is the residual metal contamination, which necessitates rigorous and expensive purification steps to meet the stringent regulatory limits imposed by health authorities for pharmaceutical products. Furthermore, the ligands associated with these metal catalysts can be prohibitively expensive, sensitive to air and moisture, and sometimes difficult to synthesize in multi-kilogram quantities with consistent quality. The reliance on scarce resources also introduces supply chain volatility, making long-term production planning challenging for procurement managers who must secure reliable sources of critical reagents. Additionally, the waste streams generated from heavy metal catalysis pose substantial environmental disposal challenges, increasing the overall cost of compliance and sustainability efforts for chemical manufacturers.
The Novel Approach
In contrast to these traditional hurdles, the novel approach outlined in the patent data presents a metal-free or low-metal alternative that utilizes organophosphorus chemistry to drive asymmetric induction. The core innovation lies in the synthesis of a chiral phosphine amide ester scaffold, which integrates a chiral amino alcohol-derived oxazoline ring directly onto a phosphorus-containing heterocyclic system. 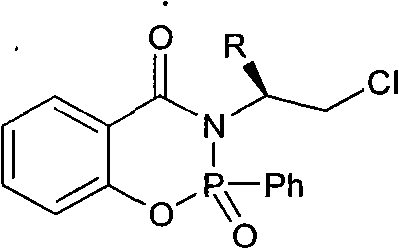 This structural architecture creates a rigid chiral pocket that effectively directs the approach of substrates during the catalytic cycle, ensuring high levels of stereoselectivity without the need for expensive transition metals. The synthesis itself is remarkably straightforward, involving the condensation of 2-cyanophenol with various D-amino alcohols followed by phosphorylation, a route that utilizes abundant and cost-effective starting materials. This shift towards organocatalysis or main-group element catalysis not only simplifies the downstream processing by eliminating heavy metal removal steps but also aligns with the growing industry trend towards greener and more sustainable chemical manufacturing practices. For supply chain leaders, this translates to a more resilient production model that is less susceptible to the geopolitical and market fluctuations associated with precious metal commodities.
This structural architecture creates a rigid chiral pocket that effectively directs the approach of substrates during the catalytic cycle, ensuring high levels of stereoselectivity without the need for expensive transition metals. The synthesis itself is remarkably straightforward, involving the condensation of 2-cyanophenol with various D-amino alcohols followed by phosphorylation, a route that utilizes abundant and cost-effective starting materials. This shift towards organocatalysis or main-group element catalysis not only simplifies the downstream processing by eliminating heavy metal removal steps but also aligns with the growing industry trend towards greener and more sustainable chemical manufacturing practices. For supply chain leaders, this translates to a more resilient production model that is less susceptible to the geopolitical and market fluctuations associated with precious metal commodities.
Mechanistic Insights into Chiral Phosphine Amide Ester Catalysis
The exceptional performance of these chiral phosphine amide esters in asymmetric transformations can be attributed to the synergistic interaction between the Lewis acidic phosphorus center and the chiral oxazoline moiety. In the context of the Henry reaction, which involves the nitroaldol addition of nitromethane to aldehydes, the catalyst likely operates by activating the nitroalkane through hydrogen bonding or Lewis acid-base interactions while simultaneously organizing the aldehyde substrate within its chiral cleft. The rigidity of the benzoxazaphosphorin ring system prevents conformational flexibility that could lead to non-selective background reactions, thereby enforcing a specific trajectory for the nucleophilic attack. This precise spatial arrangement is critical for differentiating between the enantiotopic faces of the prochiral aldehyde, resulting in the formation of one enantiomer over the other with high fidelity. The presence of the phenyl group on the phosphorus atom further enhances the steric bulk, contributing to the discrimination of transition states and ultimately dictating the stereochemical outcome of the reaction. Understanding these mechanistic nuances allows process chemists to fine-tune reaction conditions, such as solvent choice and temperature, to maximize both yield and enantiomeric excess.
Experimental data from the patent highlights the practical efficacy of this mechanism, particularly when applied to the synthesis of beta-nitro alcohols, which are valuable precursors for beta-amino acids and other bioactive molecules.  As demonstrated in the provided examples, specific derivatives of the catalyst, such as the one derived from D-phenylglycinol, exhibited outstanding enantioselectivity, achieving values greater than 99% ee in the reaction of benzaldehyde with nitromethane. This level of optical purity is often difficult to achieve with first-generation catalysts and underscores the potency of the new structural motif. The mechanism also appears to be robust across different substrates, as variations in the R-group of the amino alcohol component allow for the modulation of steric and electronic properties to suit specific reaction partners. Such tunability is a hallmark of advanced catalyst design, providing R&D teams with a versatile toolkit to address diverse synthetic challenges. The ability to operate under relatively mild conditions, such as room temperature in methanol, further attests to the efficiency of the catalytic cycle, minimizing energy consumption and reducing the risk of thermal degradation for sensitive substrates.
As demonstrated in the provided examples, specific derivatives of the catalyst, such as the one derived from D-phenylglycinol, exhibited outstanding enantioselectivity, achieving values greater than 99% ee in the reaction of benzaldehyde with nitromethane. This level of optical purity is often difficult to achieve with first-generation catalysts and underscores the potency of the new structural motif. The mechanism also appears to be robust across different substrates, as variations in the R-group of the amino alcohol component allow for the modulation of steric and electronic properties to suit specific reaction partners. Such tunability is a hallmark of advanced catalyst design, providing R&D teams with a versatile toolkit to address diverse synthetic challenges. The ability to operate under relatively mild conditions, such as room temperature in methanol, further attests to the efficiency of the catalytic cycle, minimizing energy consumption and reducing the risk of thermal degradation for sensitive substrates.
How to Synthesize Chiral Phosphine Amide Ester Efficiently
The synthesis of these high-value chiral catalysts follows a logical and scalable two-step sequence that is well-suited for adaptation in a GMP-compliant manufacturing environment. The process begins with the formation of a chiral oxazoline intermediate, which serves as the foundational chiral building block for the final catalyst structure. This initial step requires careful control of anhydrous conditions to prevent hydrolysis of the nitrile group and ensure high conversion rates. Following the isolation and purification of the intermediate, the second step involves the introduction of the phosphorus functionality through a reaction with phenylphosphonic dichloride.
- Reflux 2-cyanophenol and D-amino alcohol in anhydrous chlorobenzene with ZnCl2 catalyst for 24 hours to form the oxazoline intermediate.
- React the purified intermediate with phenylphosphonic dichloride in a benzene and pyridine mixture under reflux for 15 hours.
- Purify the final crude product using column chromatography to obtain the target chiral phosphine amide ester with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this chiral phosphine amide ester technology offers compelling advantages that directly address the pain points of modern chemical procurement and supply chain management. The shift away from precious metal catalysts fundamentally alters the cost structure of the synthesis, removing the dependency on volatile commodity markets for metals like palladium or iridium. This transition not only stabilizes raw material costs but also eliminates the need for specialized scavenging resins and extensive analytical testing for heavy metal residues, leading to significant operational savings. For procurement managers, this means a more predictable budget and reduced exposure to supply disruptions caused by mining strikes or geopolitical tensions affecting metal exports. Furthermore, the simplified purification workflow reduces the overall processing time and solvent consumption, contributing to a leaner and more efficient manufacturing operation that can respond more agilely to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated removal processes results in a drastically simplified production workflow that lowers the overall cost of goods sold. By utilizing abundant organic starting materials such as 2-cyanophenol and D-amino alcohols, manufacturers can avoid the premium pricing associated with specialized organometallic reagents. The reduction in downstream processing steps, specifically the removal of metal scavengers and the associated filtration units, decreases capital expenditure on equipment and maintenance. Additionally, the potential for solvent recovery and recycling is enhanced due to the absence of metal-contaminated waste streams, further driving down operational expenses. These cumulative efficiencies translate into a more competitive pricing structure for the final chiral intermediates, allowing companies to offer better value to their downstream pharmaceutical clients.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and readily available chiral pool materials ensures a robust and continuous supply chain that is less prone to interruptions. Unlike rare earth metals or complex proprietary ligands that may have single-source suppliers, the precursors for this catalyst are produced by multiple global vendors, fostering a competitive sourcing environment. This diversity in the supply base mitigates the risk of shortages and provides procurement teams with greater leverage in negotiations. Moreover, the stability of the catalyst structure suggests a longer shelf life and easier storage requirements compared to air-sensitive metal complexes, reducing inventory losses and logistics complexities. A stable supply of high-quality catalyst is essential for maintaining uninterrupted production schedules for critical API intermediates, ensuring that downstream drug manufacturing timelines are met without delay.
- Scalability and Environmental Compliance: The synthetic route described is inherently scalable, utilizing standard unit operations such as reflux and column chromatography that can be easily translated from benchtop to pilot plant and full commercial scale. The absence of toxic heavy metals simplifies waste treatment protocols, making it easier for facilities to comply with increasingly stringent environmental regulations regarding effluent discharge. This green chemistry profile enhances the corporate sustainability image and reduces the liability associated with hazardous waste disposal. The ability to scale up without encountering the mass transfer or heat dissipation issues often seen with heterogeneous metal catalysts ensures that production capacity can be expanded rapidly to meet surging demand. Consequently, supply chain heads can plan for long-term growth with confidence, knowing that the manufacturing process is both environmentally sound and technically feasible at large volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral catalyst technology in industrial settings. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering a clear picture of what potential partners can expect.
Q: What represents the key structural advantage of this chiral phosphine amide ester?
A: The compound features a rigid 4H-1,3,2-benzoxazaphosphorin-4-one core fused with a chiral oxazoline ring, which provides a well-defined chiral environment essential for high enantioselectivity in asymmetric transformations.
Q: How does this catalyst perform in Henry reactions compared to traditional methods?
A: In the asymmetric Henry reaction of benzaldehyde, specific variants of this catalyst (such as derivative 1b) have demonstrated exceptional enantioselectivity exceeding 99% ee, significantly outperforming many conventional ligands that often struggle to reach such high stereochemical control.
Q: Is the synthesis of this catalyst scalable for industrial production?
A: Yes, the synthetic route utilizes readily available starting materials like 2-cyanophenol and commercially sourced D-amino alcohols, and the reaction conditions involve standard reflux techniques in common organic solvents, facilitating straightforward scale-up from laboratory to commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphine Amide Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral catalysts like the phosphine amide esters described in CN102382140A for the next generation of pharmaceutical synthesis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market-ready product is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements, to guarantee that every batch meets the highest industry standards. We understand that consistency is key in the supply of chiral intermediates, and our dedicated technical team works closely with clients to optimize process parameters for maximum yield and reproducibility.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific synthetic challenges. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your project, demonstrating how switching to this organophosphorus catalyst system can optimize your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. By partnering with us, you gain access to a reliable supply chain and deep technical expertise that will accelerate your development timelines and secure your position in the competitive global market.
