Advanced Synthesis of Benzoxazole Diamines for High-Performance Optical Polymers and Laser Dyes
The landscape of advanced electronic materials and high-performance polymers is constantly evolving, driven by the demand for materials with superior thermal stability and optical properties. Patent CN1583752A introduces a groundbreaking methodology for the synthesis of benzoxazole diamines, specifically targeting the production of p-phenylene-2,2'-bis(5-aminobenzoxazole) and 2,6-bis(p-aminophenyl)benzo[1,2-d;5,4-d']bisoxazole. These compounds are not merely academic curiosities; they are foundational building blocks for next-generation optical polymers and near-ultraviolet laser dyes that require exceptional stability under irradiation. The core innovation lies in the strategic modification of the classic polyphosphoric acid (PPA) condensation route. By integrating a reductive protection strategy using stannous chloride (SnCl2), the process effectively mitigates the notorious oxidation susceptibility of aminophenol precursors. This technical breakthrough addresses a long-standing bottleneck in the fine chemical industry, where the degradation of sensitive starting materials often leads to inconsistent batch quality and prohibitive costs. For R&D directors and procurement specialists alike, understanding this shift from traditional oxidative-prone methods to a protected reductive cyclization is essential for securing a reliable supply chain of high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzoxazole derivatives via condensation in polyphosphoric acid has been plagued by significant technical hurdles related to the stability of the reactants. Traditional protocols, such as those referenced in earlier literature like US4087409, typically involve the direct reaction of diaminophenol hydrochlorides with dicarboxylic acids without adequate protection against atmospheric oxygen. In these conventional setups, the amino and phenolic groups on the starting materials are highly susceptible to oxidation, especially under the elevated temperatures required for cyclodehydration. This oxidative degradation results in the formation of complex tarry by-products and quinoid impurities that are notoriously difficult to separate from the desired product. Furthermore, the purification processes associated with these older methods are labor-intensive and inefficient, often requiring hot dimethylformamide (DMF) dissolution followed by water precipitation and final sublimation. The reliance on sublimation is particularly problematic for commercial scale-up, as it inherently limits throughput and often fails to remove trace solvent residues completely, compromising the purity specifications required for electronic grade materials.
The Novel Approach
The methodology disclosed in CN1583752A represents a paradigm shift by introducing a reductive environment directly into the reaction matrix. Instead of accepting oxidation as an inevitable loss factor, this novel approach proactively adds stannous chloride (SnCl2) as a sacrificial reducing agent within the polyphosphoric acid medium. This simple yet profound modification creates a protective shield around the sensitive 2,4-diaminophenol hydrochloride or 4,6-diaminoresorcinol hydrochloride molecules. By scavenging dissolved oxygen and preventing the formation of oxidative by-products, the reaction pathway is steered cleanly towards the desired benzoxazole ring closure. Consequently, the crude product obtained is of significantly higher quality, allowing for a drastic simplification of the downstream processing. The need for energy-intensive sublimation is eliminated, replaced by straightforward solvent extraction and recrystallization techniques. This transition not only enhances the overall yield—reported to exceed 90% in optimized examples compared to roughly 75% in older methods—but also ensures a more robust and reproducible manufacturing process suitable for industrial demands.
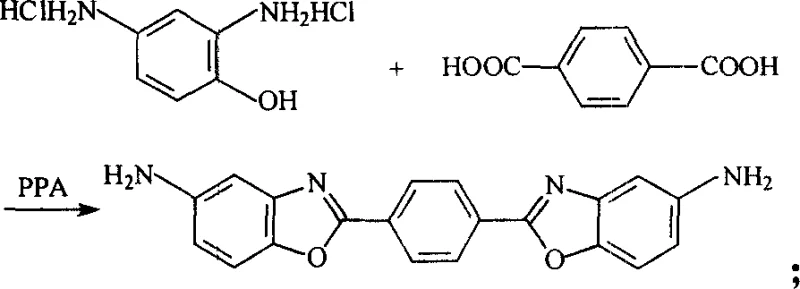
Mechanistic Insights into SnCl2-Assisted Cyclodehydration
To fully appreciate the technical superiority of this synthesis, one must delve into the mechanistic role of the tin species within the highly acidic polyphosphoric environment. The cyclization reaction fundamentally involves the nucleophilic attack of the phenolic hydroxyl group onto the carboxylic acid carbonyl, followed by dehydration to form the oxazole ring. However, the concurrent presence of free amino groups makes the aromatic ring electron-rich and vulnerable to electrophilic attack by oxygen. In the absence of a reducing agent, radical mechanisms initiate the degradation of the starting material before cyclization can complete. The introduction of SnCl2 alters this dynamic by establishing a redox buffer; the Sn(II) species is readily oxidized to Sn(IV) by any trace oxygen present in the system, effectively consuming the oxidant before it can attack the organic substrate. This preservation of the amine functionality ensures that the stoichiometry of the reaction remains intact, allowing for the full conversion of the limiting reagent. Moreover, the resulting tin by-products are inorganic salts that are highly soluble in the aqueous workup phase, facilitating their complete removal during the neutralization and washing steps, thereby contributing to the high purity of the final isolated solid.
Beyond the redox protection, the choice of polyphosphoric acid (PPA) as the solvent-catalyst system is critical for driving the equilibrium towards product formation. PPA acts as both a powerful dehydrating agent and a proton source, activating the carboxylic acid towards nucleophilic attack while simultaneously sequestering the water molecule produced during ring closure. The patent specifies precise control over the P2O5 concentration (80%-85%) to optimize viscosity and acidity, ensuring efficient mass transfer even at high solids loading. The reaction temperature profile, ramping from an initial mixing phase at 60-120°C to a final cyclization temperature of 140-210°C, is designed to manage the exotherm and prevent localized overheating which could trigger side reactions. This controlled thermal history, combined with the inert gas blanket (nitrogen or argon), creates an ideal kinetic environment for the formation of the rigid benzoxazole backbone. The result is a crystalline product with minimal structural defects, a prerequisite for its subsequent application in high-performance optical films where light scattering from impurities must be minimized.
![Synthesis reaction scheme of 2,6-bis(p-aminophenyl)benzo[1,2-d;5,4-d']bisoxazole](/insights/img/benzoxazole-diamine-synthesis-pharma-supplier-20260306024724-02.webp)
How to Synthesize Benzoxazole Diamine Efficiently
The practical execution of this synthesis requires careful attention to the preparation of the polyphosphoric acid medium and the sequential addition of reagents to maintain the integrity of the reducing environment. The process begins with the generation of the PPA solvent under strict inert atmosphere conditions, followed by the controlled addition of the amine and acid components. Once the homogeneous mixture is achieved, the critical addition of the stannous chloride reducing agent triggers the protective mechanism before the temperature is raised for cyclization. The detailed standardized operating procedures, including specific molar ratios, heating ramps, and workup protocols necessary to replicate the high yields described in the patent, are outlined in the technical guide below.
- Prepare polyphosphoric acid solution by mixing phosphoric acid and P2O5 under inert gas protection.
- Add diaminophenol hydrochloride and dicarboxylic acid reactants at controlled temperatures (60-120°C).
- Introduce SnCl2 reducing agent, heat to 140-210°C for cyclization, then neutralize and purify the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this SnCl2-assisted synthesis route offers tangible benefits that extend far beyond the laboratory bench, directly impacting the bottom line through operational efficiency and risk mitigation. The primary economic driver is the substantial improvement in material utilization; by preventing the oxidative loss of expensive aminophenol starting materials, the process maximizes the conversion of raw materials into saleable product. This reduction in waste translates directly into a lower cost of goods sold (COGS), as fewer kilograms of feedstock are required to produce a kilogram of the final intermediate. Furthermore, the elimination of the sublimation step represents a significant capital and operational expenditure saving. Sublimation is a batch process with low throughput and high energy consumption; replacing it with continuous or semi-continuous extraction and crystallization allows for much larger batch sizes and faster cycle times. This scalability is crucial for meeting the growing demand from the display and optical coating industries without requiring proportional increases in manufacturing footprint.
- Cost Reduction in Manufacturing: The integration of stannous chloride as a low-cost additive effectively replaces the need for complex purification trains. In traditional methods, the removal of oxidative tars and solvent residues often requires multiple recrystallizations or chromatographic steps, each adding labor, solvent, and time costs. By maintaining a clean reaction profile, the new method simplifies the isolation to a basic neutralization and filtration followed by a single recrystallization. This streamlining reduces solvent consumption drastically, particularly avoiding the use of large volumes of high-boiling solvents like DMF which are costly to recover and dispose of. Additionally, the higher yield means that the fixed costs of reactor time and utilities are amortized over a greater mass of product, further driving down the unit price for high-purity benzoxazole diamines.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the variability inherent in processes sensitive to air exposure. The conventional synthesis of these diamines is prone to batch-to-batch fluctuations depending on the efficiency of the inert gas purge and the quality of the starting amines. The robustness of the reductive protocol described in CN1583752A provides a safety margin against minor variations in operating conditions, ensuring consistent quality output. This reliability allows supply chain planners to forecast inventory levels with greater confidence and reduces the need for safety stock buffers. Moreover, the starting materials, such as terephthalic acid and diaminophenols, are commodity chemicals available from multiple global sources, reducing the risk of single-source bottlenecks. The ability to source raw materials broadly while applying a proprietary stabilization technique creates a resilient supply chain structure.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns well with modern green chemistry principles. The avoidance of sublimation reduces energy intensity, while the simplified workup minimizes the generation of hazardous organic waste streams. The tin by-products generated are inorganic and can be managed through standard wastewater treatment protocols or recovered, unlike complex organic sludge which requires incineration. The process is inherently scalable; the use of polyphosphoric acid is common in industrial settings, and the reaction conditions (temperatures up to 210°C) are well within the range of standard glass-lined or stainless steel reactors. This ease of scale-up from pilot plant to commercial tonnage ensures that suppliers can rapidly respond to market surges in demand for optical polymers without lengthy process re-validation periods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized benzoxazole intermediates. The answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit for your specific application needs. Understanding these nuances is vital for R&D teams assessing route feasibility and procurement teams negotiating supply agreements.
Q: Why is Stannous Chloride (SnCl2) critical in this synthesis?
A: SnCl2 acts as an oxygen scavenger in the acidic medium, preventing the oxidation of sensitive amino-phenol starting materials, which significantly boosts yield and purity.
Q: What are the primary applications of these benzoxazole diamines?
A: They serve as key monomers for optical polymers, nonlinear optical materials, and near-UV laser dyes due to their excellent stability and fluorescence properties.
Q: How does this method improve upon traditional sublimation purification?
A: The new method eliminates the need for difficult sublimation and hot DMF dissolution, replacing them with simpler extraction and recrystallization steps that ensure higher recovery rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazole Diamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team has extensively analyzed the reductive cyclization pathways described in CN1583752A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that achieving the theoretical yields of 98% requires precise control over moisture, oxygen levels, and temperature gradients, capabilities that are embedded in our state-of-the-art facilities. Our rigorous QC labs ensure that every batch of benzoxazole diamine meets stringent purity specifications, free from the oxidative impurities that plague inferior grades. Whether you are developing next-generation OLED materials or high-strength fibers, our commitment to quality assurance guarantees that your supply chain remains uninterrupted and your product performance remains uncompromised.
We invite you to engage with our technical sales team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your specific project. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us collaborate to bring high-performance optical materials to market faster and more efficiently, leveraging the power of innovative chemistry to drive your business forward.
