Advanced Manufacturing of High-Purity Mannityl Nicotinate for Global Pharmaceutical Supply Chains
Advanced Manufacturing of High-Purity Mannityl Nicotinate for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously demands higher purity standards for active ingredients, particularly for cardiovascular agents like Mannityl Nicotinate, also known as Mannitol Hexanicotinate. Patent CN103819399A introduces a groundbreaking methodology that addresses critical purity and yield challenges inherent in traditional synthesis routes. This innovative approach leverages a specific chlorination-esterification sequence followed by a sophisticated multi-stage refining process to achieve product purity exceeding 99%, with single impurity levels strictly controlled below 0.5%. For R&D directors and procurement specialists, this represents a significant leap forward in ensuring patient safety and therapeutic efficacy, as high-purity raw materials are essential for avoiding adverse reactions in treatments for hyperlipidemia and hypertension.
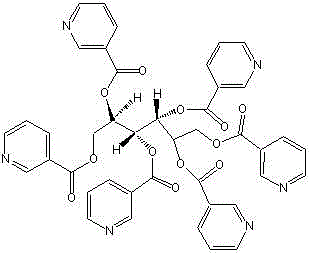
Beyond mere purity metrics, the patent outlines a robust framework for commercial viability by optimizing reagent usage and solvent selection. The transition from conventional thionyl chloride to phosphorus oxychloride as the activating agent marks a pivotal shift in process chemistry, offering better atom economy and reduced environmental impact. Furthermore, the elimination of toxic amide solvents like N,N-dimethylformamide (DMF) in favor of safer alcoholic solvents aligns with modern green chemistry principles and regulatory expectations for bulk drug manufacturing. This report delves deep into the mechanistic advantages, operational protocols, and supply chain benefits of this novel production method, providing a comprehensive roadmap for stakeholders looking to secure a reliable source of high-quality Mannityl Nicotinate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for Mannityl Nicotinate have long been plagued by inefficiencies and safety concerns that hinder optimal commercial production. Historically, manufacturers relied heavily on thionyl chloride as the chlorinating agent, which often required catalytic amounts of pyridine and a significant excess of the reagent to drive the reaction to completion. This stoichiometric imbalance not only inflated raw material costs but also generated substantial quantities of hazardous waste that required complex disposal procedures. Moreover, the subsequent purification steps frequently necessitated the use of large volumes of N,N-dimethylformamide (DMF), a solvent known for its reproductive toxicity and difficult removal profiles, posing serious risks to operator health and complicating solvent recovery systems.
The reliance on these harsh conditions often resulted in inconsistent product quality, where controlling trace impurities became a persistent challenge for quality assurance teams. The distillation processes required to remove excess chlorinating agents were energy-intensive and introduced thermal stress that could degrade the sensitive ester bonds in the molecule. Consequently, the final active pharmaceutical ingredient often struggled to meet the stringent purity specifications required for modern cardiovascular therapies, leading to batch rejections and supply chain disruptions. These legacy methods simply cannot support the rigorous demands of contemporary Good Manufacturing Practice (GMP) standards without incurring prohibitive operational costs and environmental liabilities.
The Novel Approach
In stark contrast, the methodology described in patent CN103819399A revolutionizes the production landscape by introducing a streamlined and safer chemical pathway. The core innovation lies in the substitution of thionyl chloride with phosphorus oxychloride, which acts as a highly efficient chlorinating and esterifying agent under milder conditions. This switch drastically reduces the consumption of chlorinating agents while simultaneously improving their utilization ratio, thereby minimizing waste generation at the source. The process operates within a solvent system that avoids the use of toxic DMF entirely, opting instead for alcoholic solvents such as ethanol or isopropanol, which are significantly safer for large-scale bulk drug production and easier to recover.
Furthermore, the novel approach incorporates a unique refining strategy based on acidolysis and low-temperature organic solvent processing, which allows for precise control over crystal growth and impurity exclusion. By carefully managing pH levels and temperature during the crystallization phases, the method effectively separates the target molecule from byproducts and unreacted starting materials. This results in a final product with purity levels consistently greater than 99%, ensuring that the risk of low-purity medicinal raw materials causing patient harm is virtually eliminated. The simplicity of the process flow, combined with relatively small cost losses during purification, makes this technique an ideal candidate for cost-effective and sustainable commercial manufacturing.
Mechanistic Insights into Phosphorus Oxychloride-Mediated Esterification
The chemical foundation of this superior synthesis route rests on the efficient activation of nicotinic acid followed by a controlled nucleophilic attack by mannitol. In the initial step, nicotinic acid reacts with phosphorus oxychloride in a solvent environment, typically pyridine or similar bases, to generate the reactive chlorinated intermediate, nicotinoyl chloride. This activation step is critical as it converts the carboxylic acid group into a highly electrophilic acyl chloride, priming it for the subsequent esterification reaction. The use of phosphorus oxychloride ensures a clean conversion with minimal side reactions, setting the stage for high-yield formation of the hexa-ester structure.
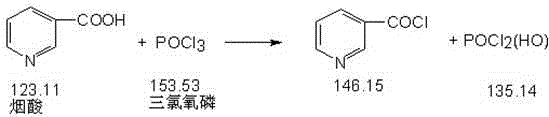
Following the activation, the process moves to the esterification phase where the chlorinated nicotinic acid intermediate reacts with mannitol. The stoichiometry is carefully controlled to ensure that all six hydroxyl groups of the mannitol molecule are successfully esterified, forming the symmetrical Mannityl Nicotinate structure. The reaction conditions are optimized to prevent partial esterification or degradation of the sugar alcohol backbone, which are common sources of difficult-to-remove impurities. The resulting crude product is then subjected to a rigorous purification regimen that leverages the differential solubility of the ester in acidic and alcoholic media.
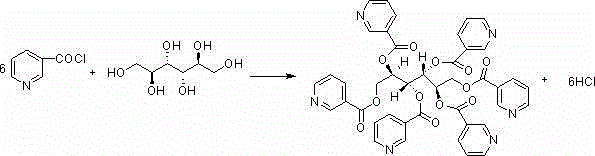
Impurity control is achieved through a multi-stage refining process that exploits the physicochemical properties of the target molecule. The crude product is first dissolved in a diluted acid solution, such as sulfuric acid, and treated with activated carbon to remove colored impurities and heavy metal traces. Subsequent crystallization steps involve the careful addition of basic solutions to adjust the pH, inducing precipitation of the pure ester while leaving soluble impurities in the mother liquor. This acid-base manipulation, combined with low-temperature washing using alcohols, ensures that the final crystal lattice is free from occluded solvents and byproducts, delivering the exceptional purity profile required for pharmaceutical applications.
How to Synthesize Mannityl Nicotinate Efficiently
The synthesis of Mannityl Nicotinate via this patented route involves a sequence of precise chemical transformations and purification steps designed to maximize yield and purity. The process begins with the activation of nicotinic acid using phosphorus oxychloride, followed by the esterification with mannitol under controlled thermal conditions. Once the crude ester is formed, it undergoes a specialized decolorization and multi-step recrystallization protocol involving acid dissolution and pH-adjusted precipitation. For R&D teams looking to implement this technology, understanding the specific parameters for temperature control, solvent ratios, and pH adjustment is critical to replicating the high success rates reported in the patent data.
- Activate nicotinic acid using phosphorus oxychloride in a solvent environment to form the chlorinated intermediate.
- React the activated intermediate with mannitol under controlled temperature to generate the crude ester product.
- Purify the crude product through acidolysis, decolorization with activated carbon, and multi-stage low-temperature recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel manufacturing process offers tangible strategic benefits that extend beyond simple unit cost metrics. The shift away from hazardous reagents and solvents fundamentally alters the risk profile of the supply chain, reducing the likelihood of regulatory shutdowns or environmental compliance issues that can disrupt availability. By utilizing more common and safer chemicals like phosphorus oxychloride and ethanol, manufacturers can secure raw material sourcing more easily and maintain consistent production schedules even during market fluctuations. This resilience is crucial for maintaining the continuity of supply for essential cardiovascular medications in a volatile global market.
- Cost Reduction in Manufacturing: The replacement of thionyl chloride with phosphorus oxychloride leads to a significant optimization in reagent consumption, directly lowering the variable costs associated with raw materials. Additionally, the elimination of DMF removes the need for expensive solvent recovery systems and hazardous waste disposal fees, further driving down the overall cost of goods sold. The simplified process flow also reduces energy consumption by avoiding high-vacuum distillation steps, contributing to a leaner and more economically efficient production model that allows for competitive pricing strategies.
- Enhanced Supply Chain Reliability: By relying on widely available and stable chemical inputs, the manufacturing process becomes less susceptible to supply shocks that often affect specialized or highly regulated reagents. The robustness of the synthesis route means that production can be scaled up or adjusted rapidly to meet demand surges without compromising on quality or lead times. This flexibility ensures that downstream pharmaceutical partners can rely on a steady stream of high-purity intermediates, minimizing the risk of stockouts and enabling better inventory planning for finished drug products.
- Scalability and Environmental Compliance: The use of alcoholic solvents instead of toxic amides greatly simplifies the scale-up process, as safety protocols for handling flammable alcohols are well-established and less burdensome than those for reproductive toxins. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the environmental footprint of the manufacturing facility. Consequently, producers can expand capacity with greater confidence, knowing that the process meets stringent international standards for safety and sustainability, which is increasingly a prerequisite for partnerships with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Mannityl Nicotinate using this advanced methodology. These insights are derived directly from the detailed experimental data and process descriptions found in the underlying patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this supply source into their existing manufacturing networks.
Q: What is the primary advantage of using phosphorus oxychloride over thionyl chloride in this synthesis?
A: Using phosphorus oxychloride significantly reduces the consumption of the chlorinating agent and improves its utilization ratio compared to traditional thionyl chloride methods. This substitution also enhances the on-site production environment by minimizing hazardous byproducts and simplifying the reaction workflow.
Q: How does this process achieve purity levels greater than 99%?
A: The process employs a rigorous multi-step purification strategy involving acidolysis followed by low-temperature organic solvent processing. By utilizing alcoholic solvents instead of toxic amide solvents like DMF and implementing precise pH control during crystallization, single impurity rates are maintained below 0.5%.
Q: Is this manufacturing method suitable for large-scale commercial production?
A: Yes, the method is designed for scalability by replacing hazardous solvents like DMF with safer alcoholic solvents such as ethanol or isopropanol. This shift not only improves operator safety but also simplifies solvent recovery and waste treatment, making it highly viable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mannityl Nicotinate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering active pharmaceutical ingredients that meet the highest standards of purity and consistency. Our expertise as a CDMO partner allows us to translate complex laboratory innovations, such as the phosphorus oxychloride-mediated synthesis route, into robust commercial realities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Mannityl Nicotinate delivers the therapeutic performance your patients depend on.
We invite you to collaborate with us to leverage these technological advancements for your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can strengthen your supply chain and drive your business forward.
