Advanced One-Step Cyclization Strategy for High-Purity Fluorobenzimidazole Intermediates
Advanced One-Step Cyclization Strategy for High-Purity Fluorobenzimidazole Intermediates
In the rapidly evolving landscape of pharmaceutical intermediate manufacturing, process intensification stands as a critical driver for both cost efficiency and supply chain reliability. A recent technological breakthrough, documented in patent CN112094237B, introduces a transformative synthesis method for fluorobenzimidazole derivatives that fundamentally alters the production paradigm. This innovation replaces the traditionally cumbersome two-step condensation and cyclization sequence with a streamlined, one-pot protocol utilizing excess formic acid. For global procurement leaders and R&D directors seeking a reliable pharmaceutical intermediate supplier, this development represents a significant leap forward in process robustness. By consolidating reaction steps, the technology not only enhances overall yield but also drastically simplifies the operational workflow, addressing key pain points related to solvent management and energy consumption in fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of fluorobenzimidazole scaffolds has been plagued by inefficiencies inherent in multi-step batch processing. The conventional route, as detailed in the background art of the patent, necessitates a distinct condensation phase in acetic acid to form a semi-open cyclic intermediate, followed by a separate ring-closing reaction mediated by formic acid. This bifurcated approach imposes severe logistical burdens on the manufacturing facility, requiring multiple heating and cooling cycles, intermediate isolation or solvent swapping, and extensive quality control checkpoints between stages. Furthermore, the reliance on acetic acid in the initial step introduces complications regarding solvent recovery and waste stream management, often leading to lower overall atom economy. The cumulative effect of these operational complexities is a capped yield potential, typically hovering between 80% and 85%, alongside increased risks of impurity accumulation during the transfer between reaction vessels.
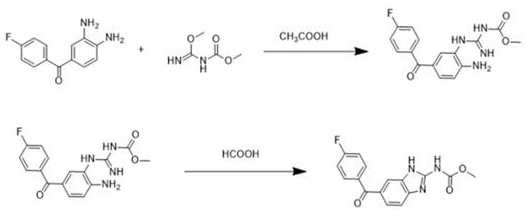
The Novel Approach
The methodology disclosed in CN112094237B elegantly circumvents these bottlenecks by leveraging the dual functionality of formic acid as both a reactant and a reaction medium. By introducing a substantial molar excess of formic acid (16 to 20 equivalents) directly to the mixture of the diamine substrate and methyl ester reactant, the process drives the cyclization to completion in a single thermal cycle. This "telescoped" strategy eliminates the need for the preliminary acetic acid condensation step entirely, thereby removing the associated unit operations of solvent evaporation and neutralization. The result is a remarkably clean reaction profile that achieves superior yields ranging from 86.9% to 87.6%. This direct cyclization not only accelerates the production timeline but also ensures a more consistent impurity profile, which is paramount for downstream API synthesis where strict regulatory specifications must be met.
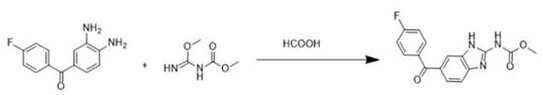
Mechanistic Insights into Formic Acid-Mediated Cyclization
The success of this novel synthetic route lies in the unique physicochemical properties of formic acid when employed in large excess. In this system, formic acid acts as a potent dehydrating agent, facilitating the removal of water molecules generated during the condensation of the amine and carbamate functionalities. The elevated concentration of formic acid shifts the chemical equilibrium decisively towards the formation of the benzimidazole ring, effectively suppressing the reverse hydrolysis reaction that often plagues equilibrium-limited cyclizations. Moreover, the reaction temperature of 80°C provides sufficient thermal energy to overcome the activation barrier for ring closure without inducing thermal degradation of the sensitive fluorobenzophenone moiety. This precise balance of acidity and thermal input ensures that the cyclization proceeds rapidly and selectively, minimizing the formation of polymeric byproducts or over-acylated species that are common in less optimized acidic environments.
From an impurity control perspective, the elimination of the acetic acid step is particularly advantageous. In the traditional two-step process, the semi-cyclic intermediate formed in acetic acid can sometimes undergo side reactions or incomplete conversion before being subjected to the second cyclization step. By bypassing this intermediate isolation, the new process reduces the residence time of reactive species in potentially degrading conditions. Additionally, the direct precipitation of the product into methanol at the end of the reaction serves as an effective purification step, as the high polarity of the reaction mixture combined with the anti-solvent effect of methanol promotes the crystallization of the target fluorobenzimidazole while leaving soluble impurities in the mother liquor. This intrinsic purification capability significantly reduces the burden on downstream recrystallization processes, further enhancing the overall process mass intensity (PMI).
How to Synthesize Fluorobenzimidazole Efficiently
Implementing this advanced synthesis protocol requires careful attention to reagent stoichiometry and thermal control to maximize the benefits of the one-pot design. The process begins with the precise charging of the diamine substrate and the methyl ester reactant into a standard glass-lined or stainless steel reactor, followed by the addition of the formic acid charge. It is critical to maintain the molar ratio of formic acid between 16 and 20 equivalents relative to the substrate to ensure the reaction medium remains sufficiently acidic throughout the conversion. The detailed standardized synthesis steps, including specific agitation rates and cooling profiles for the precipitation phase, are outlined in the technical guide below for process engineers.
- Charge the reaction vessel with the diamine substrate and methyl ester reactant, then add a significant excess of formic acid (16-20 molar equivalents) to serve as both reagent and solvent.
- Heat the reaction mixture to 80°C and maintain this temperature for 2.5 to 3 hours to ensure complete cyclization and conversion to the benzimidazole core.
- Upon completion, inject the hot reaction solution into methanol to induce precipitation, cool to room temperature, and filter to isolate the high-purity fluorobenzimidazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from a two-step to a one-step synthesis model offers profound strategic advantages beyond mere yield improvements. The consolidation of reaction steps translates directly into a reduction of the manufacturing footprint required per kilogram of output, allowing existing facilities to increase throughput without significant capital investment in new reactors. This process intensification aligns perfectly with the industry's drive towards cost reduction in pharmaceutical intermediate manufacturing, as it lowers the variable costs associated with utilities, labor, and solvent procurement. Furthermore, the simplified workflow enhances supply chain resilience by reducing the number of potential failure points; with fewer unit operations and intermediate handlings, the risk of batch failures due to operational error or equipment malfunction is significantly mitigated, ensuring more reliable delivery schedules for downstream customers.
- Cost Reduction in Manufacturing: The elimination of the acetic acid condensation step removes the necessity for a dedicated solvent recovery distillation column for acetic acid, thereby lowering the capital expenditure requirements for the production line. Additionally, the reduction in total processing time leads to substantial savings in energy consumption, as the reactor heating mantle is engaged for a shorter duration per batch. The use of formic acid, which is generally less expensive and easier to handle than specialized condensing agents, further contributes to a lower bill of materials, driving down the overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: By simplifying the synthesis to a single reaction vessel operation, the lead time for producing each batch is drastically shortened, allowing manufacturers to respond more agilely to fluctuations in market demand. The robustness of the one-pot method means that production schedules are less susceptible to delays caused by intermediate quality testing or equipment turnaround times between steps. This increased operational velocity ensures a steadier flow of high-purity intermediates to API manufacturers, reducing the risk of stockouts and enabling just-in-time inventory strategies that optimize working capital for all parties in the supply chain.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, as the exothermic nature of the reaction is easily managed at 80°C even in large-scale reactors, posing minimal safety risks during scale-up. From an environmental standpoint, the reduction in solvent usage—specifically the removal of acetic acid—significantly decreases the volume of hazardous waste generated per kilogram of product. This aligns with increasingly stringent global environmental regulations and corporate sustainability goals, making the manufacturing process more attractive to eco-conscious partners and reducing the costs associated with waste disposal and environmental compliance reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel fluorobenzimidazole synthesis method. These insights are derived directly from the experimental data and process descriptions found in patent CN112094237B, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning technology transfers and for procurement specialists negotiating long-term supply agreements.
Q: What is the primary yield improvement of the new one-step method compared to conventional synthesis?
A: According to patent CN112094237B, the novel one-step formic acid method achieves yields between 86.9% and 87.6%, whereas the traditional two-step acetic acid condensation followed by formic acid cyclization typically yields between 80% and 85%.
Q: How does eliminating the acetic acid step impact impurity profiles?
A: By bypassing the isolation of the semi-open cyclic intermediate, the new process reduces the risk of carrying over acetic acid-related impurities and minimizes the thermal degradation that can occur during multiple heating and solvent exchange cycles.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the simplification of the process into a single heating cycle at 80°C significantly enhances operational efficiency and safety, making it highly scalable for commercial manufacturing of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorobenzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of innovative synthetic routes like the one described in CN112094237B requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-pot cyclization are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of fluorobenzimidazole intermediate delivered meets the exacting standards required for modern drug development and commercial manufacturing.
We invite you to engage with our technical team to explore how this optimized synthesis can enhance your supply chain efficiency and reduce your overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this streamlined process. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your specific project needs, ensuring a seamless integration of this high-performance intermediate into your pipeline.
