Advanced Mefenacet Production: Green Chemistry & Commercial Scalability for Global Markets
Advanced Mefenacet Production: Green Chemistry & Commercial Scalability for Global Markets
The global agrochemical industry is undergoing a significant transformation driven by the urgent need for sustainable manufacturing processes and stricter environmental regulations. In this context, the technological breakthrough detailed in patent CN111303073B represents a pivotal advancement for the production of mefenacet, a high-efficiency, low-toxicity herbicide widely used for controlling gramineous weeds in rice cultivation. This patent introduces a novel synthetic pathway that leverages carbon dioxide (CO2) as a carbonylation reagent to synthesize benzothiazolone, which is subsequently reacted with 2-halogenated-N-methyl-N-phenyl acetamide. By shifting away from traditional, pollution-heavy routes, this method not only shortens the synthetic sequence but also embodies the core principles of green chemistry, offering a compelling value proposition for manufacturers seeking to reduce their carbon footprint while maintaining high production efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of mefenacet has relied on two primary methodologies, both of which present significant logistical and environmental challenges for large-scale producers. The first conventional route involves the condensation of N-methyl-N-phenyl-2-hydroxyacetamide with 2-chlorobenzothiazole, or alternatively, the hydrolysis and condensation of N-methyl-N-phenyl-2-acetoxy acetanilide with 2-chlorobenzothiazole. The second method utilizes benzothiazole ethyl sulfonate reacting with N-methyl-N-phenyl-2-hydroxyacetanilide followed by desulfonylation. A critical drawback shared by these traditional pathways is the reliance on 2-chlorobenzothiazole intermediates, the synthesis of which invariably generates toxic and harmful byproducts such as sulfur dioxide (SO2). These emissions pose severe environmental pollution risks and necessitate complex, costly waste treatment infrastructure. Furthermore, these multi-step routes involve numerous intermediates, leading to cumulative yield losses and increased operational complexity that hampers cost-effective mass production.
The Novel Approach
In stark contrast to the legacy methods, the innovative process disclosed in patent CN111303073B fundamentally reengineers the synthetic logic by utilizing benzothiazolone as a key starting material derived directly from CO2 fixation. This approach effectively bypasses the generation of sulfur dioxide, thereby eliminating a major source of hazardous waste and aligning the production process with modern environmental compliance standards. The new route is characterized by its remarkable brevity and mild reaction conditions, typically operating between 40°C and 100°C, which significantly reduces energy consumption compared to high-temperature alternatives. By streamlining the synthesis into fewer steps—specifically the carbonylation of o-aminothiophenol followed by a nucleophilic substitution—the method enhances overall atom economy. For procurement managers and supply chain directors, this translates to a more robust and resilient manufacturing protocol that minimizes the risk of regulatory shutdowns due to emissions violations while simplifying the supply chain for raw materials.
Mechanistic Insights into CO2-Mediated Carbonylation and Substitution
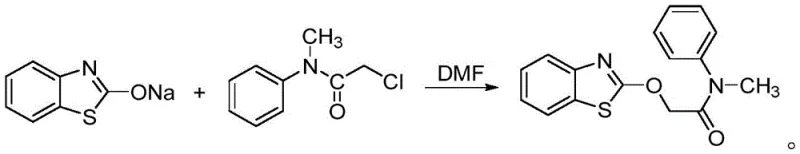
The core of this technological advancement lies in the efficient utilization of carbon dioxide as a C1 building block, a strategy that addresses both economic and ecological concerns. The mechanism initiates with the reaction of o-aminothiophenol and sodium sulfide in a polar aprotic solvent such as N-methylpyrrolidone (NMP) under a CO2 atmosphere. This carbonylation step cyclizes the precursor to form benzothiazolone, effectively sequestering CO2 into a valuable chemical scaffold. Following this, the benzothiazolone is treated with an alkali solution, such as sodium hydroxide or potassium carbonate, to generate the corresponding nucleophilic salt. This activation step is crucial as it increases the electron density on the nitrogen or oxygen atoms, priming the molecule for the subsequent coupling reaction. The final stage involves the reaction of this activated salt with 2-halogenated-N-methyl-N-phenyl acetamide (where the halogen can be Cl, Br, F, or I). This nucleophilic substitution displaces the halide, forging the critical ether or amine linkage that defines the mefenacet structure.
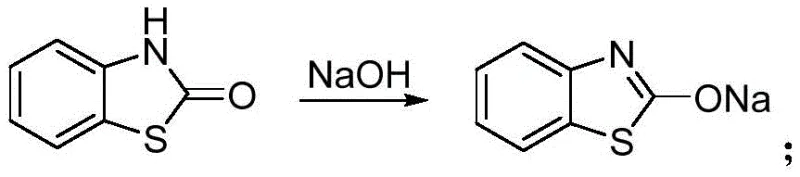
Understanding the nuances of this mechanism is vital for R&D directors focused on impurity control and process optimization. The choice of base and solvent plays a pivotal role in dictating the reaction kinetics and selectivity. Experimental data within the patent indicates that while various inorganic and organic bases (including triethylamine and sodium methoxide) are viable, inorganic bases like sodium hydroxide and potassium carbonate offer superior performance in terms of yield and ease of handling. Furthermore, the selection of the organic solvent for the acetamide component significantly influences crystal morphology and recovery rates during the final recrystallization. For instance, using DMF as a benign solvent paired with water as a poor solvent yields larger, more uniform crystals compared to systems using petroleum ether, facilitating easier filtration and drying. This level of mechanistic control ensures that the final product meets stringent purity specifications required for agrochemical registration.
How to Synthesize Mefenacet Efficiently
Implementing this synthesis route requires precise adherence to the optimized parameters identified in the patent to ensure maximum yield and product quality. The process is designed to be operationally simple, avoiding the need for exotic catalysts or extreme pressure conditions that often complicate scale-up. The following guide outlines the critical stages of the synthesis, from the initial CO2 fixation to the final purification. It is essential for process engineers to note that the molar ratios of reagents, particularly the balance between the benzothiazolone salt and the acetamide derivative, must be carefully controlled to prevent side reactions. Detailed standard operating procedures for each unit operation are provided below to assist technical teams in replicating the high yields observed in the laboratory examples.
- Synthesize benzothiazolone by reacting o-aminothiophenol with sodium sulfide and CO2 in NMP solvent at 60°C.
- Dissolve the resulting benzothiazolone in an alkali solution (e.g., NaOH) to form the benzothiazolone salt.
- React the salt solution with 2-halo-N-methyl-N-phenylacetamide in an organic solvent like DMF at 50°C to yield mefenacet.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and operational continuity, the adoption of this CO2-based synthesis route offers profound strategic benefits that extend beyond mere technical novelty. The elimination of sulfur dioxide generation removes the need for expensive scrubbing systems and hazardous waste disposal protocols, which are significant cost centers in traditional chemical manufacturing. Additionally, the use of CO2 as a feedstock leverages an abundant and inexpensive resource, potentially insulating the production cost from the volatility associated with specialized petrochemical intermediates. The simplified workflow, characterized by fewer reaction steps and milder temperatures, reduces the overall cycle time per batch, thereby increasing asset utilization rates and throughput capacity without requiring capital-intensive plant modifications.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the synthetic route and the mitigation of environmental compliance costs. By removing the step that generates toxic sulfur byproducts, manufacturers can significantly reduce expenditures related to waste treatment, emission monitoring, and regulatory reporting. Furthermore, the high yields achieved (up to 98% in optimized examples) minimize raw material waste, ensuring that a greater proportion of input costs are converted into saleable product. The ability to use common solvents like DMF and standard inorganic bases further contributes to a leaner cost structure, making the final herbicide more competitive in price-sensitive agricultural markets.
- Enhanced Supply Chain Reliability: From a sourcing perspective, this method relies on widely available commodity chemicals such as o-aminothiophenol, carbon dioxide, and basic alkalis, rather than complex, custom-synthesized intermediates like 2-chlorobenzothiazole which may have limited supplier bases. This diversification of raw material sources reduces the risk of supply disruptions caused by upstream bottlenecks or geopolitical factors. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, adds another layer of resilience, allowing production to continue even if specific grades of solvents are temporarily unavailable. This reliability is critical for maintaining consistent inventory levels to meet seasonal demand spikes in the agrochemical sector.
- Scalability and Environmental Compliance: The inherent safety and mildness of the reaction conditions make this process exceptionally well-suited for commercial scale-up. Operating at temperatures around 50°C reduces the thermal load on reactors and minimizes the risk of runaway exotherms, a common concern in large-scale organic synthesis. Moreover, the alignment with green chemistry principles positions manufacturers favorably with regulators and end-customers who are increasingly prioritizing sustainability. The reduced environmental footprint facilitates smoother permitting processes for new production lines and enhances the brand reputation of the supplier as a responsible partner in the global food security chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this mefenacet synthesis technology. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a reliable foundation for decision-making. Understanding these specifics helps bridge the gap between laboratory innovation and industrial application, ensuring that all stakeholders have a clear picture of the process capabilities and limitations.
Q: How does this new method improve environmental safety compared to traditional routes?
A: Traditional methods often involve 2-chlorobenzothiazole intermediates which generate toxic sulfur dioxide byproducts. This patented route utilizes CO2 carbonylation, eliminating hazardous sulfur emissions and aligning with green chemistry principles.
Q: What are the optimal reaction conditions for maximizing yield?
A: Experimental data indicates that a reaction temperature of 50°C provides the highest yield (up to 98%) while minimizing byproduct formation. Using DMF as the solvent and a molar ratio of 1:1.2:1.0 (benzothiazolone:alkali:acetamide) is preferred.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process features mild reaction conditions (40-100°C range) and uses readily available raw materials. The simplified synthetic route reduces operational complexity, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mefenacet Supplier
As the agrochemical landscape evolves towards greener and more efficient production methods, partnering with a technically adept manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging advanced synthetic methodologies like the CO2-mediated route to deliver high-purity mefenacet and related intermediates. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the demanding standards required for global herbicide registration and formulation.
We invite you to explore how our optimized manufacturing processes can drive value for your organization. Whether you are looking to secure a long-term supply of high-quality intermediates or need assistance in adapting this green synthesis route for your own operations, our team is ready to support your goals. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing objectives.
