Revolutionizing Statin Intermediate Production with Safe Bismuth-Catalyzed Cyclization and Scalable Oxidation
The global demand for HMG-CoA reductase inhibitors, commonly known as statins, continues to drive the need for efficient and scalable synthetic routes for their key chiral intermediates. Patent CN103224484A introduces a groundbreaking preparation method for 2-((4R,6S)-6-chloromethyl-2-alkyl-1,3-dioxane-4-yl)acetic acid, a critical building block in the synthesis of these life-saving hypolipidemic drugs. This innovation addresses the longstanding challenges associated with traditional synthetic pathways by utilizing a mild bismuth nitrate-catalyzed cyclization followed by a robust haloform reaction. By shifting away from hazardous reagents and complex purification protocols, this technology offers a streamlined approach that aligns perfectly with modern green chemistry principles and industrial safety standards. For pharmaceutical manufacturers seeking to optimize their supply chains, this patent represents a pivotal shift towards more sustainable and cost-effective production methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this vital statin intermediate has been plagued by significant operational hazards and economic inefficiencies inherent in older methodologies. As illustrated in prior art such as Chinese patents CN101613341 and CN102180862, the conventional Blaise reaction pathway necessitates the use of equimolar amounts of methoxydiethylboron, a chiral control reagent that is notoriously pyrophoric and decomposes rapidly upon exposure to moisture. 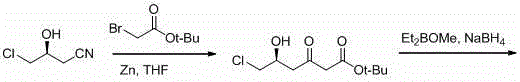 This requirement mandates strictly anhydrous and oxygen-free environments along with deep low-temperature operations, imposing severe constraints on reactor design and increasing capital expenditure for specialized equipment. Furthermore, alternative routes reported in literature, such as those utilizing mercuric acetate chloride and ytterbium trifluoromethanesulfonate, introduce toxic heavy metals into the process stream, creating substantial environmental liabilities and requiring expensive downstream removal processes to meet regulatory purity specifications. Other methods relying on strong organic bases like potassium tert-butoxide often suffer from poor selectivity, generating difficult-to-separate by-products such as conjugated dienes and disproportionation products that necessitate resource-intensive chromatographic purification.
This requirement mandates strictly anhydrous and oxygen-free environments along with deep low-temperature operations, imposing severe constraints on reactor design and increasing capital expenditure for specialized equipment. Furthermore, alternative routes reported in literature, such as those utilizing mercuric acetate chloride and ytterbium trifluoromethanesulfonate, introduce toxic heavy metals into the process stream, creating substantial environmental liabilities and requiring expensive downstream removal processes to meet regulatory purity specifications. Other methods relying on strong organic bases like potassium tert-butoxide often suffer from poor selectivity, generating difficult-to-separate by-products such as conjugated dienes and disproportionation products that necessitate resource-intensive chromatographic purification.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach detailed in CN103224484A leverages the unique Lewis acidity of bismuth nitrate to facilitate a highly efficient cyclization under remarkably mild conditions. This new strategy eliminates the need for pyrophoric reagents and toxic heavy metal catalysts, replacing them with stable, commercially available salts that can be handled with standard safety protocols. The reaction proceeds smoothly at temperatures ranging from 0°C to 50°C, significantly reducing energy consumption compared to cryogenic methods while maintaining high stereochemical control. By avoiding the formation of complex impurity profiles associated with strong base catalysis, this method simplifies the workup procedure to basic aqueous extraction and drying, thereby removing the bottleneck of chromatographic separation. This transition from high-risk, high-cost chemistry to a robust, benign catalytic system fundamentally transforms the economic and operational landscape for producing this high-value pharmaceutical intermediate.
Mechanistic Insights into Bi(NO3)3-Catalyzed Cyclization and Oxidative Cleavage
The core of this technological advancement lies in the dual-step mechanism that combines Lewis acid activation with classical oxidative cleavage to construct the chiral dioxane ring system with high fidelity. In the first step, bismuth nitrate acts as a potent yet mild Lewis acid catalyst, coordinating with the carbonyl oxygen of the aldehyde and the hydroxyl group of the starting enone to promote an intramolecular nucleophilic attack. This coordination lowers the activation energy for the formation of the 1,3-dioxane ring, ensuring that the reaction proceeds with excellent diastereoselectivity to favor the desired (4R,6S) configuration without the need for stoichiometric chiral auxiliaries. The stability of the bismuth catalyst allows the reaction to tolerate a wider range of solvents, including dichloromethane and chlorinated alkanes, providing flexibility in process optimization. 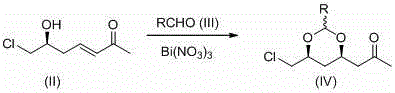 Following the successful construction of the cyclic intermediate, the process employs a haloform reaction using hypohalites such as sodium hypochlorite or sodium hypobromite to effect the oxidative cleavage of the methyl ketone moiety. This transformation converts the acetyl group directly into the carboxylic acid functionality required for the final statin side chain, utilizing a well-understood mechanism that involves tri-halogenation of the alpha-methyl group followed by nucleophilic acyl substitution. The use of aqueous hypohalite solutions ensures that the by-products are water-soluble halides and haloforms, which are easily separated from the organic product phase, resulting in a crude product of high purity that often requires no further purification beyond crystallization or simple distillation.
Following the successful construction of the cyclic intermediate, the process employs a haloform reaction using hypohalites such as sodium hypochlorite or sodium hypobromite to effect the oxidative cleavage of the methyl ketone moiety. This transformation converts the acetyl group directly into the carboxylic acid functionality required for the final statin side chain, utilizing a well-understood mechanism that involves tri-halogenation of the alpha-methyl group followed by nucleophilic acyl substitution. The use of aqueous hypohalite solutions ensures that the by-products are water-soluble halides and haloforms, which are easily separated from the organic product phase, resulting in a crude product of high purity that often requires no further purification beyond crystallization or simple distillation.
From an impurity control perspective, this mechanistic pathway offers distinct advantages by minimizing the generation of side products that typically plague statin intermediate synthesis. The mild nature of the bismuth catalyst prevents the dehydration of the hydroxyl group, a common side reaction in acid-catalyzed cyclizations that leads to conjugated diene impurities which are difficult to remove. Additionally, the specificity of the haloform reaction for methyl ketones ensures that other functional groups within the molecule, such as the chloromethyl group, remain intact, preserving the integrity of the chiral centers established in the first step. This high level of chemoselectivity reduces the burden on quality control laboratories, as the impurity profile is significantly cleaner compared to routes involving harsh oxidants or strong bases. Consequently, the overall yield of the process is maximized not only by high conversion rates but also by the minimization of material loss during purification, directly contributing to improved process mass intensity (PMI) metrics.
How to Synthesize 2-((4R,6S)-6-chloromethyl-2-alkyl-1,3-dioxane-4-yl)acetic acid Efficiently
The implementation of this synthesis route in a manufacturing setting follows a logical progression that prioritizes safety and reproducibility at every stage. The process begins with the dissolution of the chiral enone starting material in a suitable organic solvent, followed by the controlled addition of the aldehyde and the bismuth nitrate catalyst under ambient lighting conditions to prevent photodegradation. Once the cyclization is complete, the reaction mixture undergoes a straightforward aqueous workup to remove the catalyst and excess reagents, yielding the protected dioxanone intermediate in high yield.
- Perform a Lewis acid catalyzed addition reaction between (S)-7-chloro-6-hydroxyhept-3-en-2-one and an aldehyde using bismuth nitrate in an organic solvent.
- Isolate the intermediate 1-((4R,6S)-6-chloromethyl-2-alkyl-1,3-dioxane-4-yl)propan-2-one through standard aqueous workup and drying.
- Subject the intermediate to a haloform reaction using a hypohalite solution to oxidatively cleave the methyl ketone to the target acetic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple yield improvements. By fundamentally altering the reagent profile of the synthesis, this process removes several critical bottlenecks that have historically constrained the supply of this key statin intermediate. The shift away from specialized, hazardous reagents to commodity chemicals creates a more resilient supply chain that is less susceptible to market volatility and sourcing disruptions. Furthermore, the simplification of the purification process reduces the dependency on expensive chromatography resins and solvents, leading to a leaner and more cost-efficient manufacturing operation. These factors combined position this technology as a superior choice for long-term commercial partnerships focused on reliability and sustainability.
- Cost Reduction in Manufacturing: The elimination of pyrophoric chiral boron reagents and expensive rare earth catalysts like ytterbium trifluoromethanesulfonate results in a drastic reduction in raw material costs. Since the bismuth nitrate catalyst is used in catalytic amounts and the oxidants are inexpensive commodity chemicals, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the removal of chromatographic purification steps saves substantial amounts of solvent and silica gel, further driving down operational expenses. The mild reaction conditions also reduce energy costs associated with heating and cooling, contributing to a more economical production profile.
- Enhanced Supply Chain Reliability: Sourcing pyrophoric reagents often requires specialized vendors and strict transportation protocols, introducing potential points of failure in the supply chain. By switching to stable, shelf-stable reagents like bismuth nitrate and sodium hypochlorite, manufacturers can source materials from a broader range of suppliers, ensuring continuity of supply. The robustness of the reaction conditions means that production is less likely to be halted due to minor fluctuations in temperature or humidity, enhancing overall plant uptime. This reliability is crucial for meeting the rigorous delivery schedules demanded by major pharmaceutical companies.
- Scalability and Environmental Compliance: The absence of toxic mercury and the use of benign bismuth catalysts simplify waste treatment processes, ensuring compliance with increasingly stringent environmental regulations. The haloform reaction generates waste streams that are easier to treat compared to heavy metal-contaminated effluents, reducing the cost and complexity of environmental management. The process is inherently scalable, as demonstrated by the ability to run reactions at room temperature without the need for specialized cryogenic equipment. This scalability allows for seamless transition from pilot plant to commercial scale production, facilitating rapid market entry for new generic statin formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bismuth-catalyzed synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation to ensure accuracy and relevance. Understanding these details is essential for technical teams evaluating the feasibility of adopting this new methodology for their specific production needs.
Q: Why is the bismuth nitrate method superior to traditional Blaise reactions for this intermediate?
A: Traditional Blaise reactions require pyrophoric chiral boron reagents and strict anhydrous conditions, whereas the bismuth nitrate method uses stable, non-pyrophoric catalysts under mild conditions, significantly enhancing operational safety and reducing equipment costs.
Q: Does this process involve toxic heavy metals like mercury?
A: No, unlike previous methods reported in literature that utilized toxic mercuric acetate chloride, this patented process relies on bismuth, which is environmentally benign, and standard hypohalites, eliminating the need for complex heavy metal removal steps.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process features mild reaction temperatures ranging from 0°C to 50°C, uses commercially available raw materials, and avoids difficult chromatographic purifications, making it highly amenable to multi-kilogram and ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-((4R,6S)-6-chloromethyl-2-alkyl-1,3-dioxane-4-yl)acetic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-((4R,6S)-6-chloromethyl-2-alkyl-1,3-dioxane-4-yl)acetic acid meets the exacting standards required for API synthesis. Our facility is equipped to handle the specific safety and environmental requirements of this bismuth-catalyzed process, delivering a product that is both high-quality and sustainably produced.
We invite you to collaborate with us to leverage this advanced technology for your statin drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative synthesis method can enhance your supply chain efficiency and reduce your overall manufacturing costs.
