Scalable Manufacturing of High-Purity Prasugrel Hydrobromide via Green Bromination
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for the production of critical antithrombotic agents. Patent CN103524530A introduces a significant technological advancement in the synthesis of Prasugrel hydrobromide, a key active pharmaceutical ingredient (API) used in the prevention of thrombotic cardiovascular events. This innovation addresses long-standing challenges in the manufacturing of thienopyridine derivatives by replacing hazardous halogenating agents with a greener, in-situ bromination system. By utilizing hydrobromic acid and hydrogen peroxide, the process eliminates the reliance on expensive and toxic N-bromosuccinimide (NBS) and benzoyl peroxide, marking a pivotal shift towards safer chemical manufacturing. For global procurement leaders and R&D directors, this patent represents a viable strategy for cost reduction in API manufacturing while simultaneously enhancing the safety profile of the production facility. The method ensures high purity through optimized recrystallization protocols, directly impacting the quality of the final drug product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Prasugrel and its salts has relied on legacy routes that pose significant operational and economic burdens. As illustrated in prior art such as CN1214031C, the conventional pathway involves the generation of a Grignard reagent from ortho-fluorobenzyl bromide, followed by reaction with cyclopropanecarbonitrile. A critical bottleneck in this traditional sequence is the subsequent alpha-bromination step, which necessitates the use of N-bromosuccinimide (NBS) and benzoyl peroxide in carbon tetrachloride (CCl4). This approach is fraught with difficulties: CCl4 is a known carcinogen and ozone-depleting substance, subjecting manufacturers to strict environmental regulations and high disposal costs. Furthermore, the use of NBS is economically inefficient due to the high cost of the reagent and the generation of succinimide byproducts that complicate downstream purification. The overall yield of such traditional methods is often suboptimal, reported around 22.7%, and typically requires labor-intensive column chromatography for purification, which is a major impediment to commercial scale-up of complex pharmaceutical intermediates.
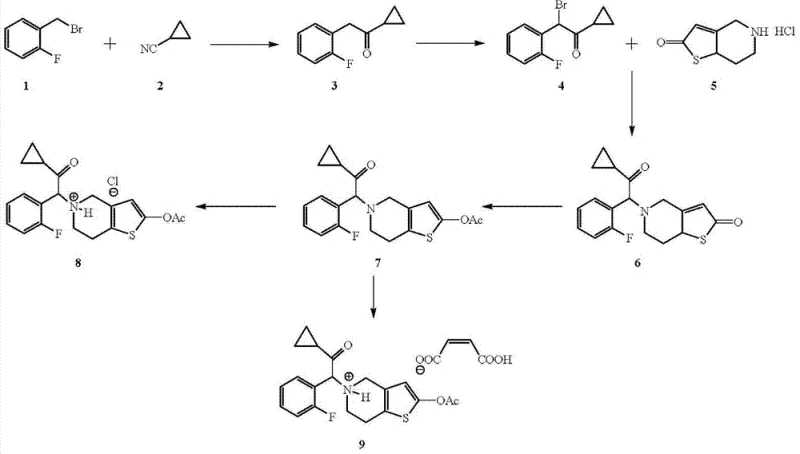
The Novel Approach
In stark contrast, the methodology disclosed in CN103524530A offers a streamlined and economically superior alternative. The core innovation lies in the direct alpha-bromination of 1-cyclopropyl-2-(2-fluorophenyl) ethyl ketone using an aqueous system of hydrobromic acid and hydrogen peroxide. This generates elemental bromine in situ, which reacts efficiently with the ketone substrate in alcohol solvents like ethanol or isopropanol, completely avoiding chlorinated solvents. This green chemistry approach not only drastically reduces raw material costs but also simplifies the workup procedure to basic extraction and concentration. Following bromination, the intermediate undergoes nucleophilic substitution with 2-oxo-4,5,6,7-tetrahydro-thieno[3,2-c]pyridine hydrochloride, followed by acetylation. The entire sequence is designed to maximize throughput and minimize waste, providing a reliable prasugrel intermediate supplier with a distinct competitive advantage in terms of production speed and regulatory compliance.
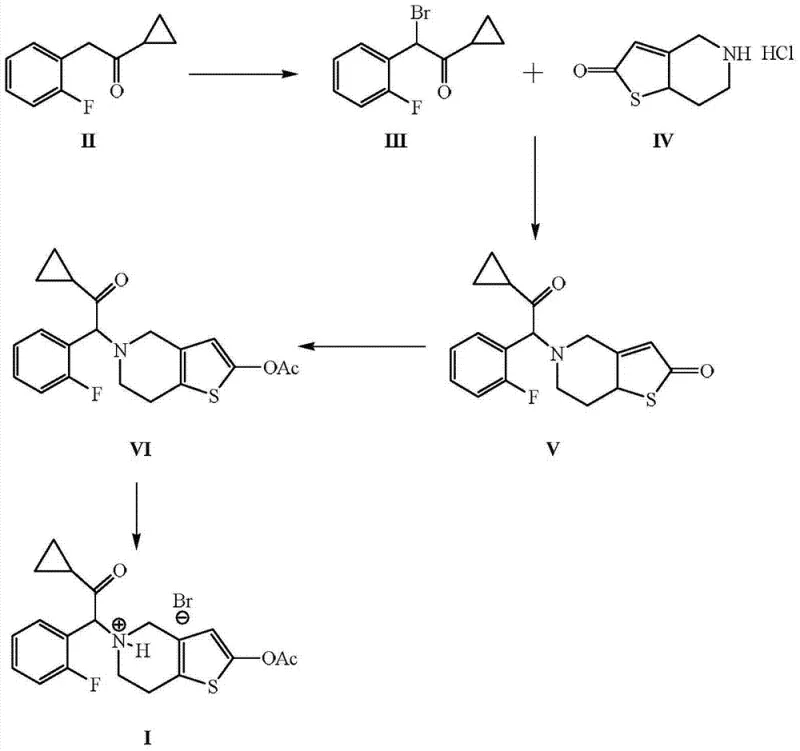
Mechanistic Insights into In-Situ Bromination and Purification
The mechanistic elegance of this process centers on the controlled generation of electrophilic bromine species. In the first step, the reaction between hydrobromic acid (HBr) and hydrogen peroxide (H2O2) generates molecular bromine (Br2) within the reaction medium. This electrophile attacks the alpha-position of the ketone (Compound II), facilitated by the enolizable nature of the substrate. The use of protic solvents like ethanol stabilizes the transition state and solubilizes the inorganic salts formed, ensuring a homogeneous reaction environment that promotes high conversion rates. Unlike radical bromination with NBS which can lead to poly-brominated side products, this ionic mechanism offers superior regioselectivity for the mono-bromo derivative (Compound III). The stoichiometry is carefully balanced, typically employing a molar ratio of ketone to HBr of 1:1 to 1:5, ensuring complete consumption of the starting material without excessive halogen waste.
Purity control is another critical aspect where this patent excels, particularly for R&D teams focused on impurity profiles. The protocol specifies a rigorous recrystallization step using acetonitrile for the free base of Prasugrel (Compound VI) prior to salt formation. Acetonitrile acts as a selective solvent that dissolves the target molecule at elevated temperatures (60°C to 80°C) while precipitating structurally related impurities upon cooling. This physical purification method is far more scalable and reproducible than chromatographic techniques. By repeating this crystallization, the process achieves a purity exceeding 99.5% with single impurities below 0.05%. This high level of chemical integrity is essential for the subsequent salt formation with hydrobromic acid, ensuring that the final high-purity prasugrel hydrobromide meets the stringent specifications required for clinical and commercial applications without the need for further tedious purification.
How to Synthesize Prasugrel Hydrobromide Efficiently
The synthesis of this critical antithrombotic intermediate is broken down into four distinct, manageable unit operations that prioritize safety and yield. The process begins with the green bromination of the fluorophenyl ketone, followed by coupling with the thienopyridine core, acetylation, and final salt formation. Each step has been optimized for temperature and solvent conditions to ensure maximum efficiency. For laboratory and pilot plant teams looking to implement this technology, the following guide outlines the standardized operational parameters derived from the patent embodiments. These steps provide a clear roadmap for reducing lead time for high-purity pharmaceutical intermediates while maintaining strict quality control standards throughout the manufacturing campaign.
- Perform alpha-bromination of 1-cyclopropyl-2-(2-fluorophenyl) ethyl ketone using HBr and H2O2 in ethanol to generate the key bromo-intermediate.
- Condense the bromo-intermediate with 2-oxo-4,5,6,7-tetrahydro-thieno[3,2-c]pyridine hydrochloride using a base like sodium carbonate in acetonitrile.
- Acetylate the resulting ketone intermediate with acetic anhydride and triethylamine, followed by recrystallization in acetonitrile to achieve >99.5% purity.
- Convert the purified free base into the final hydrobromide salt using aqueous hydrobromic acid in a methanol/MTBE solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits beyond mere chemical yield. The elimination of specialized, high-cost reagents like NBS and benzoyl peroxide significantly lowers the Bill of Materials (BOM) cost. Moreover, the substitution of carbon tetrachloride with ethanol removes the heavy regulatory burden associated with handling and disposing of Class 1 solvents, thereby reducing environmental compliance costs and insurance premiums. The simplified purification workflow, which relies on crystallization rather than column chromatography, allows for faster batch turnover times and higher equipment utilization rates. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting global demand fluctuations.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of expensive halogenating agents. N-bromosuccinimide is a premium reagent with a high price point, whereas hydrobromic acid and hydrogen peroxide are commodity chemicals available in bulk at a fraction of the cost. Additionally, the removal of succinimide byproduct generation simplifies the waste stream treatment, further lowering operational expenditures. By avoiding the need for silica gel chromatography, the process saves on consumable costs and solvent usage, leading to substantial overall savings in the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of niche reagents. This new method relies on universally available raw materials such as ethanol, acetonitrile, and aqueous acids, which are less susceptible to market volatility compared to specialized organic peroxides. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent grades, ensures that production schedules are less likely to be disrupted by minor variations in raw material quality. This reliability is crucial for maintaining consistent inventory levels and meeting Just-In-Time delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: From a scale-up perspective, the process is inherently safer and easier to manage. Exothermic risks associated with peroxide initiators are mitigated by the controlled addition of hydrogen peroxide. The use of non-chlorinated solvents aligns with modern Green Chemistry principles, facilitating easier permitting for new manufacturing lines. The ability to produce high-purity material through simple filtration and crystallization means that the process can be scaled from kilograms to tons without the engineering complexities associated with large-scale chromatography, ensuring a smooth transition from pilot plant to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on process capabilities and quality outcomes. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the new bromination method improve safety compared to traditional routes?
A: The novel process replaces hazardous reagents like N-bromosuccinimide (NBS) and benzoyl peroxide with safer, in-situ generated bromine from hydrobromic acid and hydrogen peroxide. Furthermore, it eliminates the use of toxic carbon tetrachloride (CCl4) solvents, substituting them with greener alternatives like ethanol, significantly reducing environmental and operational risks.
Q: What purity levels can be achieved with this synthesis route?
A: By utilizing acetonitrile recrystallization during the intermediate stages and specifically for the final free base, this method consistently achieves purity levels greater than 99.5%, with single impurities controlled below 0.05%, meeting stringent regulatory requirements for API intermediates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It avoids complex purification steps like column chromatography, relying instead on standard filtration and crystallization techniques. The use of readily available, cost-effective reagents and simplified workup procedures makes it highly amenable to multi-kilogram and ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prasugrel Hydrobromide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green bromination route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of Prasugrel Hydrobromide meets or exceeds the >99.5% purity benchmark. Our commitment to quality assurance ensures that our clients receive intermediates that are ready for immediate downstream processing, minimizing delays in their own API synthesis timelines.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data. Let us help you optimize your supply chain for Prasugrel production with a solution that balances economic efficiency, environmental responsibility, and uncompromising quality.
