Advanced Nickel-Catalyzed Synthesis of CF2O-Containing Liquid Crystal Monomers for Industrial Scale-Up
The global demand for high-performance liquid crystal materials, particularly those exhibiting high dielectric anisotropy and low viscosity for TFT-LCD applications, continues to drive innovation in synthetic chemistry. A pivotal advancement in this sector is detailed in patent CN103086850A, which discloses a highly efficient preparation method for CF2O-containing compounds, specifically targeting the synthesis of 4-(4-n-propyl)phenyl-3,5-difluoro-1',1'-difluorobenzyl-3,4,5-trifluorophenyl ether. This patent addresses the critical bottlenecks associated with traditional difluoro-methoxy bridge formation, offering a route that is not only chemically superior but also economically viable for large-scale production. By shifting the synthetic paradigm from late-stage fluorination to an early-stage etherification followed by nickel-catalyzed coupling, the technology promises to redefine the supply chain dynamics for electronic chemical manufacturers seeking reliable sources of advanced monomer intermediates.
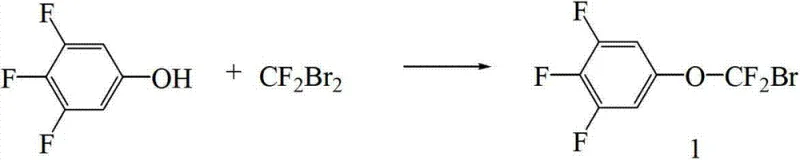
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of liquid crystal monomers containing the difluoro-methoxy (CF2O) central bridged bond has been plagued by significant technical and economic hurdles. Conventional methodologies, such as the DAST (diethylaminosulfur trifluoride) fluorination route, rely on converting ester groups into difluoro-methoxy bonds at low temperatures, often requiring cryogenic conditions around -70°C. This approach suffers from inherently low reaction yields, typically hovering around 35%, and necessitates the use of prohibitively expensive fluorinating reagents that drastically inflate the cost of goods. Furthermore, alternative methods involving the direct reaction of polycyclic Grignard reagents with dibromodifluoromethane often result in complex post-processing operations and substantial purification losses due to the formation of difficult-to-separate byproducts, rendering these processes unfavorable for industrial amplification.
The Novel Approach
In stark contrast, the methodology outlined in CN103086850A introduces a strategic reversal of the synthetic sequence that effectively circumvents these legacy inefficiencies. The novel approach prioritizes the formation of the difluoro-methoxy bridge on a simpler phenolic precursor, specifically 3,4,5-trifluorophenol, before engaging in the complex carbon-carbon bond formation. By first synthesizing 5-(bromo-difluoro-methoxy)-1,2,3-trifluorobenzene through a controlled Williamson etherification with difluorodibromomethane, the process establishes the critical CF2O linkage under mild alkaline conditions. This intermediate is subsequently coupled with a biphenyl Grignard reagent using a nickel catalyst, a transformation that proceeds with high conversion rates and atom economy. This shift not only simplifies the operational workflow but also significantly enhances the overall purity of the final product, making it a superior choice for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Nickel-Catalyzed Cross-Coupling
The core of this technological breakthrough lies in the sophisticated application of nickel-catalyzed cross-coupling chemistry to forge the bond between the fluorinated aromatic rings. The reaction utilizes anhydrous nickel chloride (NiCl2) or nickel acetylacetonate (Ni(acac)2) as a catalyst within a tetrahydrofuran (THF) solvent system, operating at moderate temperatures between 50°C and 70°C. Mechanistically, the nickel catalyst facilitates the oxidative addition into the carbon-bromine bond of the 5-(bromo-difluoro-methoxy) intermediate, generating a reactive organonickel species. This species then undergoes transmetallation with the 3,5-difluoro-4'-propylbiphenyl-4-magnesium bromide Grignard reagent, followed by reductive elimination to yield the target CF2O-containing biphenyl ether. This catalytic cycle is remarkably robust, tolerating the multiple fluorine substituents on the aromatic rings which typically deactivate substrates in palladium-catalyzed systems, thereby ensuring high reaction efficiency.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over non-catalytic nucleophilic substitutions. The specificity of the nickel catalyst minimizes homocoupling of the Grignard reagent, a common side reaction that generates symmetric biphenyl impurities difficult to remove. Additionally, by pre-forming the O-CF2Br moiety on the phenol ring, the process avoids the harsh conditions required to install the CF2O group directly onto the sensitive biphenyl core, which often leads to decomposition or rearrangement products. The use of inert gas protection (nitrogen or argon) throughout the coupling further suppresses oxidation of the Grignard reagent, ensuring that the reaction profile remains clean and that the final crude product exhibits high GC purity, often exceeding 80% prior to final crystallization, thus reducing the burden on downstream purification units.
How to Synthesize 4-(4-n-propyl)phenyl-3,5-difluoro-1',1'-difluorobenzyl-3,4,5-trifluorophenyl ether Efficiently
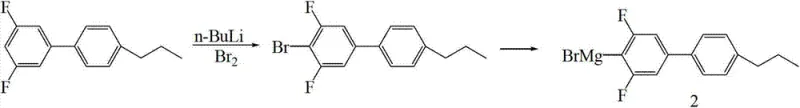
The execution of this synthesis requires precise control over stoichiometry and environmental conditions to maximize the benefits of the patented route. The process begins with the preparation of the key intermediate, 5-(bromo-difluoro-methoxy)-1,2,3-trifluorobenzene, by reacting 3,4,5-trifluorophenol with difluorodibromomethane in the presence of sodium hydride and sodium in DMAC solvent. Once this intermediate is secured, it is coupled with the Grignard reagent derived from 4-bromo-3,5-difluoro-4'-propylbiphenyl. The detailed standardized synthesis steps, including specific molar ratios, temperature ramps, and workup procedures necessary to achieve the reported 81% yield, are outlined below for technical reference.
- Prepare 5-(bromo-difluoro-methoxy)-1,2,3-trifluorobenzene by reacting 3,4,5-trifluorophenol with difluorodibromomethane under alkaline conditions in an amide solvent.
- Generate the Grignard reagent 3,5-difluoro-4'-propylbiphenyl-4-magnesium bromide from the corresponding bromo-biphenyl precursor using magnesium turnings in THF.
- Perform the final coupling reaction between the bromo-difluoro-methoxy intermediate and the Grignard reagent using anhydrous NiCl2 as a catalyst in THF at 50-70°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic advantages beyond mere chemical elegance. The elimination of cryogenic requirements and expensive fluorinating agents like DAST directly impacts the cost structure, allowing for more competitive pricing models in the volatile electronic chemicals market. Furthermore, the simplified post-processing steps, which avoid complex recrystallization of low-melting sulfonate salts seen in older methods, reduce the cycle time per batch. This efficiency gain enhances the responsiveness of the supply chain, enabling manufacturers to react more swiftly to fluctuations in demand for TFT-LCD materials without compromising on quality or delivery reliability.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost reagents with commodity chemicals and the dramatic improvement in reaction yield. By avoiding the use of DAST and utilizing difluorodibromomethane, a more accessible industrial gas, the raw material costs are substantially lowered. Additionally, the high atom economy of the nickel-catalyzed coupling means that less starting material is wasted as byproduct, leading to significant cost savings in raw material consumption per kilogram of final product produced.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as 3,4,5-trifluorophenol and standard Grignard precursors mitigates the risk of supply disruptions often associated with specialized fluorinating agents. The robustness of the reaction conditions, which do not require extreme低温 (low temperatures) or exotic catalysts, ensures that production can be maintained consistently across different manufacturing sites. This stability is crucial for maintaining continuous supply lines to major display panel manufacturers who require uninterrupted delivery of high-purity monomers.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the process is inherently greener, generating less hazardous waste compared to sulfur-based fluorination methods. The solvents used, primarily THF and DMAC, are well-understood in industrial settings and can be efficiently recovered and recycled, minimizing the environmental footprint. The mild reaction temperatures and absence of highly toxic byproducts simplify the waste treatment protocols, facilitating easier regulatory compliance and smoother scale-up from pilot plant to multi-ton commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this CF2O-containing compound synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and strategic value of adopting this new methodology for industrial partners.
Q: How does this new method improve upon the traditional DAST fluorination route?
A: The traditional DAST method requires expensive fluorinating reagents and operates at extremely low temperatures (-70°C), resulting in lower yields around 35%. The new nickel-catalyzed approach utilizes readily available difluorodibromomethane and operates at mild temperatures (50-70°C), significantly improving atom economy and reducing production costs.
Q: What represents the key innovation in the synthetic strategy for CF2O bridges?
A: The core innovation lies in reversing the synthetic order. Instead of introducing the difluoro-methoxy group onto a complex biphenyl system late in the synthesis, this method first installs the O-CF2Br moiety onto the simpler trifluorophenol ring. This intermediate is then coupled with the biphenyl Grignard reagent, minimizing side reactions and purification losses.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is specifically designed for industrial suitability. It eliminates the need for cryogenic conditions and hazardous reagents like DAST. The use of standard solvents like THF and DMAC, combined with robust nickel catalysis, ensures high conversion rates and simplified post-processing, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-n-propyl)phenyl-3,5-difluoro-1',1'-difluorobenzyl-3,4,5-trifluorophenyl ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of mastering complex fluorination chemistries to meet the rigorous demands of the electronic materials sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the specific economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical solution available.
