Advanced Enzymatic Route for Cefixime Production: Technical Analysis and Commercial Viability
Introduction to Advanced Cefixime Manufacturing Technologies
The pharmaceutical industry is constantly evolving towards greener and more cost-effective synthetic pathways, particularly for high-volume antibiotics like Cefixime. A pivotal development in this domain is documented in patent CN101319246A, which outlines a refined methodology for the preparation of Cefixime that addresses critical bottlenecks in traditional synthesis. This technical disclosure focuses on optimizing the production of the key intermediate 7-AVCA and the subsequent coupling reactions to form the final active pharmaceutical ingredient. By shifting from harsh chemical deprotection agents to biocatalytic processes and replacing chlorinated solvents with more benign alternatives, this approach offers a compelling value proposition for manufacturers seeking to enhance their supply chain resilience. For R&D directors and procurement specialists, understanding these mechanistic shifts is essential for evaluating potential partners who can deliver high-purity pharmaceutical intermediates with superior economic and environmental profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Cefixime have long been plagued by significant operational and environmental challenges that impact both cost and scalability. Historically, the preparation of the crucial 7-AVCA intermediate relied heavily on the use of trifluoroacetic acid for amino de-protection, a reagent known for its high toxicity, corrosive nature, and substantial procurement costs. Furthermore, conventional processes typically employed dichloromethane and tetrahydrofuran as primary solvents during the acylation and coupling stages, which complicates waste management due to their volatility and difficulty in recovery. The generation of mercaptobenzothiazole as a byproduct in standard coupling reactions often leads to yield losses and purity issues, as recovering this thiol derivative from chlorinated solvent systems is technically demanding and inefficient. These factors collectively contribute to a higher cost of goods sold and increased regulatory scrutiny regarding solvent residues and environmental discharge.
The Novel Approach
The innovative methodology presented in the patent data introduces a paradigm shift by substituting chemical deprotection with an enzymatic process using penicillin acylase, effectively eliminating the need for trifluoroacetic acid. This biological catalysis not only operates under milder conditions but also allows for the enzyme to be reused extensively, reportedly maintaining activity for over 300 cycles, which drastically reduces catalyst consumption costs. In the coupling phase, the process replaces problematic chlorinated solvents with a mixture of water and organic solvents such as alcohols, ketones, or esters, which significantly improves the recoverability of reaction media. This solvent engineering facilitates the complete recovery of the mercaptobenzothiazole byproduct, thereby enhancing atom economy and minimizing waste discharge. The result is a streamlined workflow that improves the hydrolysis and crystallization of subsequent products, ensuring a finished product with superior quality characteristics.
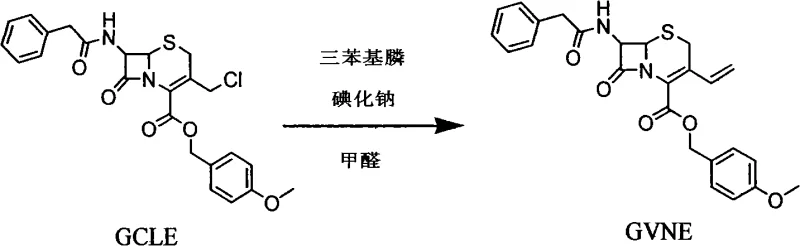
Mechanistic Insights into Enzymatic Deprotection and Coupling
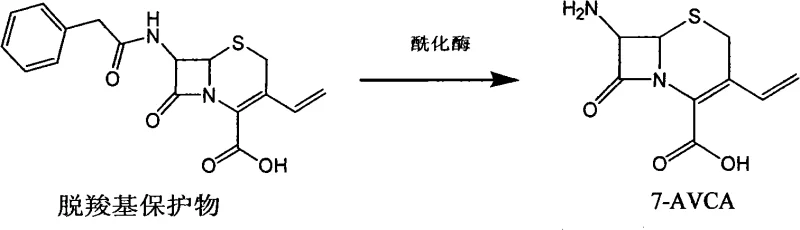
The core technical advancement in this synthesis route lies in the enzymatic hydrolysis step used to generate 7-AVCA from its protected precursor. Unlike chemical hydrolysis which requires strong acids that can degrade sensitive beta-lactam rings, penicillin acylase specifically targets the amide bond of the protecting group under neutral to slightly alkaline conditions. This specificity preserves the integrity of the cephalosporin nucleus while efficiently releasing the free amine required for the subsequent acylation. The reaction is conducted in an aqueous environment, which not only aligns with green chemistry principles but also simplifies the downstream processing by avoiding the need for extensive solvent exchanges. The enzyme's stability and reusability are critical mechanistic features, as the biocatalyst can be filtered and recycled multiple times without significant loss of activity, providing a robust and consistent reaction rate throughout the production campaign.
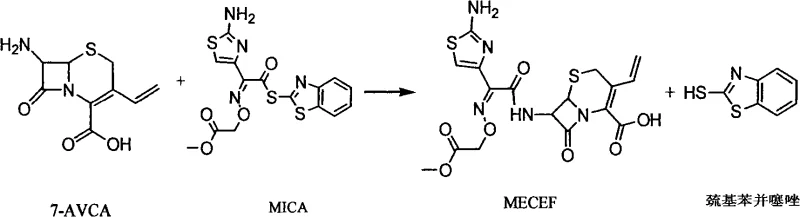
Following the formation of 7-AVCA, the coupling reaction with the MICA side chain is optimized through careful solvent selection to manage the solubility of reactants and byproducts. The use of solvents like methyl alcohol, acetone, or methyl carbonate in the presence of water creates a biphasic or homogeneous system that favors the formation of the cefixime methyl ester intermediate. The presence of water is crucial for dissolving the inorganic bases used to scavenge protons, while the organic component ensures the solubility of the organic substrates. This balanced solvent system allows for the precipitation or easy extraction of the mercaptobenzothiazole byproduct, preventing it from interfering with the final crystallization. The subsequent hydrolysis of the methyl ester to the free acid is performed under controlled alkaline conditions, followed by precise pH adjustment to induce crystallization, yielding Cefixime with high purity and defined particle size distribution suitable for oral formulation.
How to Synthesize Cefixime Efficiently
The synthesis of Cefixime via this enzymatic route involves a sequence of precise unit operations designed to maximize yield and minimize environmental impact. The process begins with the preparation of the GVNE precursor, followed by enzymatic deprotection to yield 7-AVCA, which is then coupled with the activated side chain. Detailed operational parameters such as temperature control, pH maintenance, and solvent ratios are critical for success. For a comprehensive breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to implement this technology in a GMP environment, please refer to the standardized synthesis guide below.
- Prepare the 7-AVCA intermediate by hydrolyzing GVNE with phenol followed by enzymatic deprotection using penicillin acylase in an aqueous system.
- Conduct the acylation reaction between 7-AVCA and MICA side chain in a solvent system comprising alcohols, ketones, or esters with water.
- Hydrolyze the resulting methyl ester intermediate using sodium hydroxide, followed by pH adjustment and crystallization to obtain pure Cefixime.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic synthesis route translates into tangible strategic advantages regarding cost stability and supply continuity. The elimination of trifluoroacetic acid removes a volatile cost component from the bill of materials, as this reagent is subject to significant price fluctuations and strict transportation regulations. Furthermore, the ability to recycle the biocatalyst over hundreds of cycles dramatically lowers the effective cost per kilogram of the intermediate, creating a more predictable cost structure for long-term contracts. The switch to easily recoverable solvents like alcohols and ketones reduces the volume of hazardous waste requiring disposal, leading to substantial savings in waste treatment fees and lowering the overall carbon footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The replacement of expensive chemical reagents with reusable enzymes and the implementation of efficient solvent recovery systems drive down the variable costs of production significantly. By avoiding the use of dichloromethane and tetrahydrofuran, the facility reduces its reliance on solvents that require energy-intensive distillation for recycling, further enhancing operational efficiency. The improved yield and purity of the final product also mean less material is lost to reprocessing or rejection, maximizing the output from each batch of raw materials.
- Enhanced Supply Chain Reliability: Diversifying the raw material base away from hazardous acids and chlorinated solvents mitigates the risk of supply disruptions caused by regulatory crackdowns or logistics constraints. The robustness of the enzymatic process ensures consistent batch-to-batch quality, reducing the likelihood of production delays due to out-of-specification results. This reliability is crucial for maintaining uninterrupted supply lines to downstream formulation partners who depend on timely delivery of high-quality antibiotic ingredients.
- Scalability and Environmental Compliance: The aqueous nature of the deprotection step and the use of greener solvents make this process highly scalable from pilot plant to commercial tonnage without encountering the safety hazards associated with large volumes of toxic chemicals. The simplified waste stream, characterized by lower toxicity and easier treatability, ensures compliance with increasingly stringent environmental regulations in major manufacturing hubs. This future-proofs the supply chain against potential regulatory changes that could otherwise render older, dirtier technologies obsolete.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Cefixime synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and feasibility of the enzymatic route for potential manufacturing partners. Understanding these details is vital for making informed decisions about technology transfer and capacity planning.
Q: How does the enzymatic deprotection method improve upon traditional chemical methods?
A: The enzymatic method replaces toxic and expensive trifluoroacetic acid with penicillin acylase, significantly reducing environmental impact and allowing for enzyme reuse over 300 cycles.
Q: What solvents are recommended for the acylation step to ensure easy recovery?
A: The process utilizes alcohols, ketones, or esters mixed with water, which facilitates the recovery of byproducts like mercaptobenzothiazole and simplifies solvent recycling compared to dichloromethane.
Q: Does this process affect the purity of the final Cefixime product?
A: Yes, the removal of harsh chemical deprotection agents and the efficient recovery of byproducts lead to improved hydrolysis and crystallization, resulting in higher quality finished products meeting USP standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefixime Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the enzymatic preparation of Cefixime are executed with precision and efficiency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of API or intermediate adheres to the highest international standards. We understand the critical importance of consistency in the pharmaceutical supply chain and are committed to delivering products that support your regulatory filings and commercial success.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can align with your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall procurement expenses. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your needs, ensuring a seamless transition from development to commercial supply.
