Revolutionizing Cefixime Production: A Deep Dive into the D-7-ACA Low-Cost Synthesis Route
Introduction to Patent CN110041346B
The pharmaceutical industry is constantly seeking more efficient pathways for the production of essential antibiotics, and the recent disclosure of patent CN110041346B offers a compelling solution for the manufacture of cefixime, a widely prescribed third-generation cephalosporin. This patent details a novel, low-cost preparation method that fundamentally shifts the starting material paradigm from the traditional and expensive GCLE (7-phenylacetamido-3-chloromethyl cephalosporanic acid p-methoxybenzyl ester) to the more economically viable D-7-ACA (deacetoxycephalosporanic acid derivative). By leveraging the significantly lower molecular weight of D-7-ACA, which is approximately 230.24 compared to nearly 490 for GCLE, this process achieves an effective mole ratio that is more than two times higher per unit of mass. This technical breakthrough not only addresses the critical issue of raw material costs but also streamlines the synthetic route, reducing the number of operational steps and potentially lowering the environmental footprint associated with waste generation. For procurement and R&D leaders, understanding this shift is vital for future-proofing supply chains against volatile pricing of complex beta-lactam intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of cefixime has relied heavily on GCLE as the primary starting scaffold. While chemically effective, the GCLE route suffers from inherent economic and logistical inefficiencies that burden the supply chain. The most glaring issue is the high molecular weight of GCLE, which dictates that a substantial portion of the mass purchased is essentially a leaving group or protecting moiety that contributes nothing to the final molecular architecture of the drug. Furthermore, the synthesis of GCLE itself often requires multi-step protection and deprotection strategies, adding length and cost to the upstream supply chain. Traditional methods also frequently involve harsh enzymatic hydrolysis steps or complex side-chain grafting sequences that require precise control and expensive reagents. These factors combine to create a high baseline cost of goods sold (COGS) and limit the flexibility of manufacturers to respond to market price fluctuations. Additionally, the reliance on a single, high-value starting material like GCLE creates a bottleneck; any disruption in GCLE availability can halt entire production lines, posing a significant risk to supply continuity for global pharmaceutical buyers.
The Novel Approach
In stark contrast, the methodology outlined in CN110041346B introduces a streamlined approach that bypasses these traditional bottlenecks by utilizing D-7-ACA directly. This novel route capitalizes on the maturity of D-7-ACA production technology, which has seen continuous cost reductions and quality improvements in recent years. The process initiates with a direct acylation of the 7-amino group of D-7-ACA using a cefixime active ester, specifically the MICA active ester, to form a protected first intermediate. This is followed by a targeted halogenation of the 3-hydroxymethyl group and a subsequent olefination to install the critical vinyl functionality. By eliminating the need for the bulky phenylacetyl and benzhydryl protecting groups found in the GCLE route, this new method drastically reduces the molecular baggage carried through the synthesis. The result is a shorter, more direct pathway that maximizes atom economy. For a reliable cefixime API intermediate supplier, adopting this route translates to a more robust and cost-competitive manufacturing platform that can better withstand market pressures while maintaining stringent quality standards required for regulatory compliance.
Mechanistic Insights into the D-7-ACA Coupling and Olefination Sequence
The core of this innovative synthesis lies in the precise orchestration of four distinct chemical transformations, beginning with the nucleophilic acylation of the beta-lactam nucleus. In the first step, D-7-ACA reacts with the cefixime active ester (MICA) in a biphasic system of tetrahydrofuran and water, facilitated by triethylamine as a base. This reaction must be carefully controlled at temperatures between -10°C and 30°C to prevent hydrolysis of the sensitive beta-lactam ring while ensuring complete conversion of the amine. The use of HPLC monitoring to confirm that D-7-ACA residue is lower than 0.5% demonstrates a rigorous approach to impurity control, ensuring that unreacted starting material does not carry over into subsequent steps where it could complicate purification.
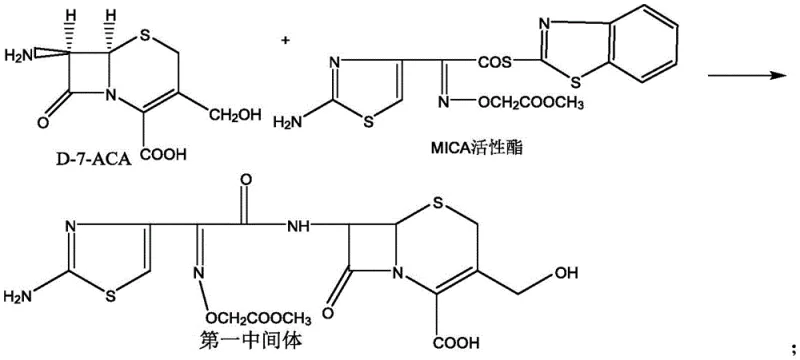
Following the formation of the first intermediate, the process moves to the activation of the 3-position side chain. The hydroxymethyl group is converted to a chloromethyl group using phosphorus pentachloride in dichloromethane and dimethylacetamide. This chlorination is a critical activation step, transforming a poor leaving group (hydroxyl) into a reactive handle for the subsequent carbon-carbon bond-forming reaction. The reaction is maintained at 0°C to manage the exotherm and prevent degradation of the cephalosporin core. The resulting second intermediate serves as the precursor for the installation of the vinyl group, a defining feature of cefixime that distinguishes it from earlier generations of cephalosporins.
The most chemically sophisticated step in this sequence is the conversion of the chloromethyl group to the vinyl group, achieved through a tandem iodination and Wittig-type reaction. In this stage, the second intermediate is treated with sodium iodide, triphenylphosphine, and formaldehyde. The sodium iodide likely facilitates a Finkelstein-type substitution to generate a more reactive iodomethyl species in situ, which then reacts with triphenylphosphine to form a phosphonium salt. Subsequent treatment with formaldehyde and base generates the ylide, which undergoes olefination to yield the desired ethylene double bond. This one-pot or sequential strategy avoids the isolation of unstable intermediates and streamlines the workflow. The reaction conditions are tightly regulated, heating to 25-65°C for the phosphonium formation and then cooling to -10-20°C for the olefination to control stereochemistry and minimize polymerization side reactions.
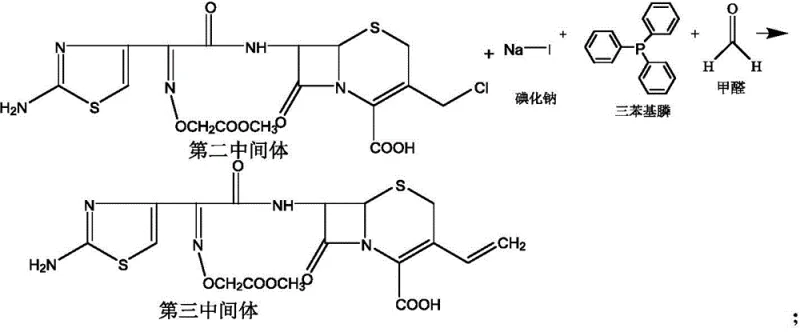
Finally, the synthesis concludes with the hydrolysis of the ester protecting group on the side chain. Using an alkali liquor such as sodium hydroxide, the methoxycarbonylmethoxy group is cleaved to reveal the free carboxylic acid, yielding the final cefixime molecule. The pH is carefully adjusted to between 1.0 and 3.0 to induce crystallization of the cefixime trihydrate. This final purification step is crucial for removing triphenylphosphine oxide and other organic impurities, ensuring the final API meets the high-purity cefixime standards demanded by global pharmacopeias.
How to Synthesize Cefixime Efficiently
Implementing this synthesis route requires strict adherence to the reaction parameters defined in the patent to ensure safety and yield. The process is designed to be scalable, utilizing standard reactor setups capable of handling low-temperature exotherms and solvent exchanges. Operators must be trained to monitor reaction progress via HPLC at critical junctures, particularly ensuring the first intermediate residue is below 1.0% before proceeding to the olefination step. The detailed standardized synthesis steps, including specific reagent ratios and workup procedures, are outlined below to guide process development teams in replicating this high-efficiency route.
- React D-7-ACA with cefixime active ester (MICA) in THF/water with triethylamine to form the first intermediate.
- Treat the first intermediate with phosphorus pentachloride in dichloromethane to convert the hydroxymethyl group to chloromethyl.
- Perform a sequential iodination and Wittig reaction using sodium iodide, triphenylphosphine, and formaldehyde to install the vinyl group.
- Hydrolyze the ester protecting group using alkali liquor, adjust pH, and crystallize to obtain pure cefixime trihydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to the D-7-ACA based route represents a strategic opportunity to optimize cost structures and mitigate supply risks. The primary driver of value is the raw material efficiency; because D-7-ACA has a molecular weight roughly half that of GCLE, purchasing one kilogram of D-7-ACA provides more than twice the molar capacity for producing cefixime compared to one kilogram of GCLE. This fundamental stoichiometric advantage translates directly into substantial cost savings in raw material procurement without compromising on the quality of the final output. Furthermore, D-7-ACA is a commodity chemical produced at massive scales for various cephalosporin antibiotics, ensuring a stable and competitive supply market compared to the more specialized GCLE.
- Cost Reduction in Manufacturing: The elimination of expensive protecting groups and the reduction in molecular weight of the starting material lead to a drastic simplification of the cost basis. By avoiding the purchase of pre-functionalized, high-molecular-weight scaffolds like GCLE, manufacturers can significantly reduce the cost of goods sold. Additionally, the use of common reagents such as phosphorus pentachloride and formaldehyde, rather than specialized enzymes or rare catalysts, further lowers the operational expenditure. This cost structure allows for greater margin flexibility in a competitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Relying on D-7-ACA diversifies the supply chain risk. Since D-7-ACA is a foundational intermediate for a wide range of beta-lactam antibiotics, its production is widespread and robust, reducing the likelihood of shortages that can occur with niche intermediates. The shortened process route also means fewer unit operations and less time spent in production, which enhances the overall throughput and responsiveness of the manufacturing facility to sudden spikes in demand. This reliability is critical for maintaining uninterrupted supply to downstream formulation partners.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like THF, dichloromethane, and acetone which are easily recovered and recycled in modern solvent recovery systems. The reduction in synthetic steps inherently reduces the volume of waste generated per kilogram of product, aligning with green chemistry principles and reducing the burden on wastewater treatment facilities. This environmental efficiency not only lowers disposal costs but also ensures compliance with increasingly stringent environmental regulations, safeguarding the facility's license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the technical specifications and beneficial effects described in patent CN110041346B, providing clarity for stakeholders evaluating the feasibility of this technology for their own production lines or sourcing strategies.
Q: Why is D-7-ACA preferred over GCLE for cefixime production?
A: D-7-ACA has a molecular weight of approximately 230.24, which is roughly half that of GCLE (489.96). This results in an effective mole ratio that is more than two times higher for the same mass of starting material, significantly reducing raw material costs and improving atom economy.
Q: How does this process control impurities during the olefination step?
A: The process utilizes a controlled sequence involving sodium iodide and triphenylphosphine at specific temperature ranges (25-65°C followed by cooling to -10-20°C). HPLC monitoring ensures the residual intermediate is lower than 1.0% before proceeding, minimizing side reactions and ensuring high purity.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the route uses commodity chemicals like phosphorus pentachloride, sodium iodide, and formaldehyde. The reaction conditions operate within standard industrial temperature ranges (-20°C to 65°C) and utilize common solvents like THF, DCM, and acetone, making it highly scalable and supply-chain friendly.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefixime Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the D-7-ACA synthesis route for the global cefixime market. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical, large-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced process control systems capable of maintaining the stringent purity specifications required for beta-lactam antibiotics. We are committed to delivering high-purity cefixime intermediates and APIs that meet international regulatory standards, leveraging our technical expertise to optimize yield and minimize impurities at every stage of the synthesis.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective technology for their supply chains. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how the D-7-ACA route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and economical supply of this critical antibiotic.
