Advanced Synthesis of Stable Isotope Labeled Quinoxaline-2-Carboxylic Acid for Veterinary Residue Analysis
Introduction to Patent CN110218195B
The pharmaceutical and agrochemical industries face increasing regulatory pressure to monitor veterinary drug residues, specifically carbadox and olaquindox, in the food supply chain. Patent CN110218195B introduces a breakthrough methodology for the synthesis of stable isotope labeled quinoxaline-2-carboxylic acid, a critical internal standard required for high-precision liquid chromatography-tandem mass spectrometry (LC-MS/MS) analysis. This compound, characterized by its specific deuterium labeling pattern, allows for the accurate quantification of toxic metabolites that pose carcinogenic risks. The invention addresses a significant market gap where such high-purity standards were previously dependent on costly imports with limited supply continuity. By establishing a robust domestic synthesis route, this technology not only enhances analytical reliability but also drastically reduces the economic burden associated with residue monitoring programs.
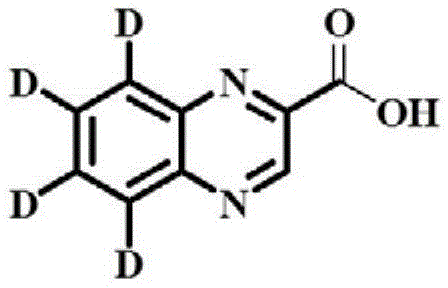
The core innovation lies in the molecular architecture of the target compound, which retains high isotope abundance exceeding 99% while maintaining chemical purity above 98%. This level of isotopic enrichment is essential for distinguishing the internal standard from the native analyte during mass spectrometric detection, thereby eliminating matrix interference effects. The structural integrity of the deuterium labels on the benzene ring is preserved through carefully controlled reaction conditions that prevent hydrogen-deuterium exchange. For analytical laboratories and quality control divisions, access to such reliably synthesized standards is paramount for meeting international safety guidelines and ensuring the integrity of the global food trade.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of stable isotope labeled quinoxaline-2-carboxylic acid has been plagued by significant supply chain vulnerabilities and exorbitant costs. Prior to this invention, the market relied almost exclusively on imported standards, creating a bottleneck for domestic testing agencies and veterinary drug manufacturers. The lack of a localized synthesis capability meant that lead times were unpredictable, and pricing was subject to volatile international trade dynamics. Furthermore, existing literature offered no viable synthetic pathways that could balance cost-efficiency with the rigorous purity requirements demanded by analytical chemistry. Traditional methods often suffered from complex multi-step sequences that led to substantial isotope loss, rendering the final product unsuitable for high-sensitivity MS detection. This technological void hindered the effective implementation of residue monitoring protocols, potentially compromising public health safety measures.
The Novel Approach
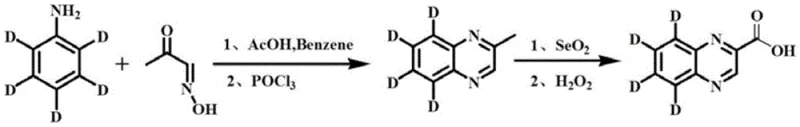
The patented methodology revolutionizes the production landscape by introducing a concise, two-step synthetic route that maximizes yield and isotopic fidelity. As illustrated in the reaction scheme below, the process begins with the condensation of stable isotope-labeled aniline and methylglyoxal-1-oxime, followed by a strategic oxidation sequence. This approach eliminates the need for complex protecting group strategies or exotic reagents that typically drive up manufacturing expenses. The use of selenium dioxide for the selective oxidation of the methyl group to an aldehyde, followed by hydrogen peroxide oxidation to the carboxylic acid, demonstrates exceptional chemoselectivity. This ensures that the sensitive quinoxaline ring system remains intact while the side chain is functionalized precisely. By streamlining the synthesis into fewer operational units, the process inherently reduces waste generation and energy consumption, aligning with modern green chemistry principles.
Furthermore, the novel approach leverages readily available starting materials, such as deuterated aniline, which are commercially accessible at reasonable price points compared to fully customized isotopic building blocks. The reaction conditions, utilizing common solvents like benzene or toluene and standard acids like acetic acid, facilitate easy scale-up from laboratory benchtop to industrial reactor volumes. This accessibility transforms the compound from a niche, high-cost specialty chemical into a commoditized intermediate that can be produced with consistent quality. For procurement managers, this shift represents a transition from a fragile, single-source supply model to a resilient, scalable manufacturing framework that guarantees long-term availability.
Mechanistic Insights into SeO2-Mediated Oxidation and Cyclization
The chemical elegance of this synthesis is rooted in the precise control of reaction mechanisms to preserve the isotopic label. In the first step, the condensation of methylglyoxal-1-oxime with deuterated aniline under acid catalysis initiates the formation of the quinoxaline heterocycle. The subsequent addition of phosphorus oxychloride (POCl3) serves a dual purpose: it acts as a potent dehydrating agent to drive the cyclization equilibrium forward and facilitates the aromatization of the dihydro-intermediate. This step is critical because incomplete cyclization would lead to impurities that are difficult to separate and could interfere with downstream oxidation. The acidic environment must be carefully managed to prevent the protonation of the nitrogen atoms, which could deactivate the ring towards further substitution, while simultaneously ensuring that the deuterium atoms on the aromatic ring do not undergo exchange with protic solvent molecules.
The second stage involves a sophisticated oxidative transformation where selenium dioxide (SeO2) selectively oxidizes the benzylic methyl group at the 2-position of the quinoxaline ring to an aldehyde functionality. This Riley oxidation is highly specific for allylic and benzylic positions, leaving the electron-deficient pyrazine ring untouched. Following the formation of the aldehyde intermediate, hydrogen peroxide is introduced to effect the final oxidation to the carboxylic acid. This two-stage oxidation strategy is superior to direct strong oxidation, which might degrade the heterocyclic core or cause decarboxylation. The mechanism ensures that the electron-withdrawing nature of the emerging carboxyl group does not destabilize the adjacent nitrogen atoms. Throughout this sequence, the deuterium labels on the fused benzene ring remain chemically inert, resulting in a final product with isotope abundance greater than 99%, a metric that is non-negotiable for quantitative mass spectrometry applications.
How to Synthesize Deuterated Quinoxaline-2-Carboxylic Acid Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high purity. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions, which simplifies the engineering requirements for production facilities. Operators must focus on precise stoichiometric control, particularly the molar ratio of selenium dioxide to the methyl quinoxaline intermediate, to prevent over-oxidation or the formation of selenium byproducts that are difficult to remove. The purification protocol, involving pH swings between alkaline and acidic conditions, exploits the amphoteric nature of the quinoxaline derivative to separate it from neutral organic impurities and inorganic salts. Detailed standardized operating procedures for each unit operation are essential to maintain the stringent quality specifications required for analytical standards.
- Condense stable isotope-labeled aniline with methylglyoxal-1-oxime under acid catalysis, followed by cyclization using phosphorus oxychloride to form the labeled quinoxaline core.
- Oxidize the resulting 2-methyl quinoxaline intermediate using selenium dioxide to generate the aldehyde, followed by hydrogen peroxide oxidation to yield the final carboxylic acid.
- Purify the crude product through pH adjustment and solvent extraction to achieve >98% purity and >99% isotope abundance.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain strategists, the adoption of this synthesis method offers transformative benefits that extend beyond simple unit cost savings. The primary advantage lies in the localization of the supply chain; by enabling domestic production of a previously imported critical reagent, organizations can mitigate the risks associated with geopolitical trade tensions and logistics disruptions. This shift enhances supply security, ensuring that analytical laboratories have uninterrupted access to the internal standards necessary for regulatory compliance testing. The simplification of the synthetic route also translates to a more robust manufacturing process with fewer failure points, leading to higher batch success rates and more predictable delivery schedules. This reliability is crucial for maintaining the continuity of large-scale residue monitoring programs that operate on tight regulatory timelines.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the utilization of low-cost, commodity-grade raw materials such as methylglyoxal-1-oxime and deuterated aniline, rather than expensive, custom-synthesized precursors. By eliminating the need for complex multi-step protection and deprotection sequences, the process significantly reduces the consumption of solvents, reagents, and labor hours per kilogram of product. The high overall yield achieved through the two-step sequence minimizes material waste, directly improving the cost-of-goods-sold (COGS) profile. Furthermore, the avoidance of precious metal catalysts or exotic oxidants removes a major variable cost component, stabilizing the pricing structure against fluctuations in the commodities market. These efficiencies collectively enable a substantial reduction in the final purchase price for end-users without compromising on quality.
- Enhanced Supply Chain Reliability: From a logistics perspective, the simplicity of the reaction conditions allows for flexible manufacturing scheduling and rapid response to demand spikes. The use of standard chemical reactors and common solvents means that production can be easily scaled or shifted between different facilities without extensive retooling. This flexibility creates a resilient supply network capable of absorbing shocks from raw material shortages or equipment maintenance downtime. Additionally, the high purity of the crude product reduces the burden on downstream purification processes, shortening the overall cycle time from raw material intake to finished goods shipment. For supply chain heads, this translates to reduced inventory holding costs and improved cash flow dynamics due to faster throughput times.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind from the outset. The reaction steps operate at moderate temperatures and pressures, reducing the energy footprint and safety risks associated with high-energy chemical transformations. The waste stream is primarily composed of manageable inorganic salts and organic solvents that can be recovered and recycled, aligning with increasingly strict environmental regulations. The elimination of heavy metal catalysts simplifies wastewater treatment and reduces the liability associated with hazardous waste disposal. This environmental compatibility not only lowers compliance costs but also enhances the corporate sustainability profile of the manufacturer, a factor that is becoming increasingly important in vendor selection criteria for multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this stable isotope labeled compound. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this standard into their analytical workflows. The answers provided reflect the consensus of the technical community regarding the efficacy and reliability of this specific synthetic pathway.
Q: What is the primary application of stable isotope labeled quinoxaline-2-carboxylic acid?
A: It serves as a critical internal standard for the precise detection of carbadox and olaquindox residues in food products using LC-MS/MS, ensuring regulatory compliance.
Q: How does this synthesis method improve upon traditional sourcing?
A: Unlike reliance on expensive imports, this domestic synthesis utilizes cheap, readily available raw materials and a concise two-step route to significantly lower production costs.
Q: What represents the key challenge in maintaining isotope abundance?
A: The process ensures that deuterium atoms on the benzene ring remain stable during acidic cyclization and oxidative conditions, preventing isotope dilution.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Stable Isotope Labeled Quinoxaline-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality reference standards play in ensuring food safety and regulatory compliance. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications, backed by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex isotopic labeling projects allows us to offer tailored solutions that address the specific challenges of veterinary drug residue analysis. By partnering with us, you gain access to a supply chain that prioritizes quality, reliability, and technical excellence.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our manufacturing capabilities can support your operational goals. Request a Customized Cost-Saving Analysis today to understand the economic benefits of switching to our domestically produced standards. Our team is ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to transparency and partnership. Let us help you secure a stable supply of high-purity intermediates that empower your analytical capabilities and safeguard your brand reputation.
