Optimizing Cangrelor Intermediate Production: A Technical Breakdown of Patent CN105481922B
Optimizing Cangrelor Intermediate Production: A Technical Breakdown of Patent CN105481922B
The global demand for potent antiplatelet agents continues to drive innovation in the synthesis of critical pharmaceutical intermediates. Specifically, the preparation of Cangrelor intermediate (Formula I), a pivotal precursor for the reversible P2Y12 receptor antagonist Cangrelor, has been the subject of intense process optimization. Patent CN105481922B discloses a novel, robust preparation method designed to overcome significant defects found in prior art, offering a pathway characterized by mild reaction conditions, superior yields, and enhanced environmental compatibility. This technical insight report analyzes the strategic value of this patented route for R&D directors and supply chain leaders seeking reliable cangrelor intermediate supplier partnerships. By shifting away from hazardous reagents and complex purification protocols, this methodology represents a substantial leap forward in the commercial scale-up of complex nucleoside analogs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cangrelor and its key intermediates has been plagued by severe safety hazards and economic inefficiencies. Early literature, such as the route disclosed by The Medicines Company (J. Med. Chem. 1999), relied heavily on dangerous reagents like sodium hydride (NaH) and expensive, scarce iodine-based raw materials. Furthermore, the construction of the pyrimidine ring in these legacy processes often suffered from low yields, typically ranging between 50% and 75%, which drastically impacts overall process economics. Perhaps most critically, the synthesis of essential precursors like 2-mercaptoadenosine frequently necessitated the use of highly toxic carbon disulfide (CS2), resulting in the generation of lethal hydrogen sulfide (H2S) gas. These factors create immense regulatory burdens and safety risks, making such routes unsuitable for modern, green manufacturing standards.
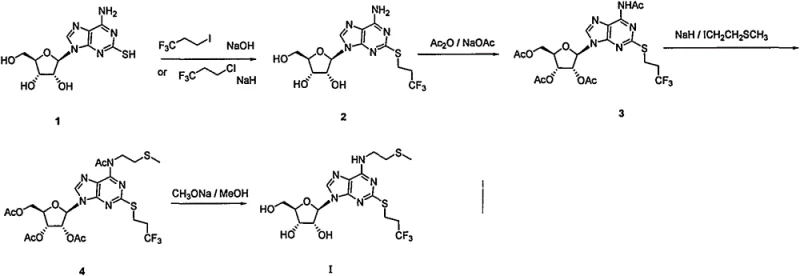
Additionally, other reported methods, such as those found in patent CN105061431, attempted to address raw material sourcing but introduced new bottlenecks. These alternative routes often required more than one equivalent of expensive protecting group reagents like SBA and excessive amounts of catalysts such as TMSOTF (trimethylsilyl trifluoromethanesulfonate). The reliance on stoichiometric or excess amounts of costly Lewis acids not only inflates the raw material costs but also complicates the downstream separation and purification processes. The removal of these metal or acid residues requires extensive washing and chromatography, which reduces throughput and increases solvent waste, thereby failing to meet the rigorous cost reduction in pharmaceutical intermediates manufacturing targets set by generic drug producers.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the process outlined in CN105481922B introduces a streamlined, six-step synthetic strategy that prioritizes safety and efficiency. The core innovation lies in the construction of the heterocyclic core and the subsequent glycosylation using readily available tetraacetyl ribose. By utilizing a specific sequence of ammonolysis, acid-catalyzed coupling, selective reduction, and orthoformate-mediated cyclization, the inventors have achieved a route that operates under significantly milder conditions. The elimination of toxic gases like H2S and the avoidance of pyrophoric reagents like NaH fundamentally alters the risk profile of the manufacturing process. This approach ensures that the production of high-purity cangrelor intermediate can be conducted in standard stainless steel reactors without the need for specialized containment systems required for toxic gas handling.
Furthermore, the novel approach demonstrates exceptional versatility in reaction conditions, allowing manufacturers to optimize for either speed or cost depending on their specific facility capabilities. For instance, the coupling reaction can be catalyzed by a variety of accessible acids including methanesulfonic acid, trifluoromethanesulfonic acid, or even sulfuric acid, providing flexibility in procurement. The reduction step offers multiple viable pathways, including catalytic hydrogenation (H2/Pd-C or H2/Ni) or chemical reduction using zinc powder or hydrazine hydrate. This redundancy in chemical logic ensures reducing lead time for high-purity cangrelor intermediates, as supply chain disruptions for a single specific reagent will not halt production. The final deprotection step utilizes simple aqueous base hydrolysis, yielding the target compound with reported HPLC purity exceeding 99%, which minimizes the need for recrystallization.
Mechanistic Insights into Acid-Catalyzed Glycosylation and Cyclization
The heart of this synthetic success lies in the precise control of the glycosylation and ring-closure mechanisms. In the second step, the coupling of the amino-pyrimidine (Compound III) with tetraacetyl ribose (Compound IV) is driven by strong acid catalysis. The mechanism likely involves the protonation of the anomeric acetate or the formation of an oxocarbenium ion intermediate, which is then attacked by the nucleophilic amine of the pyrimidine ring. The use of solvents like xylene or toluene at elevated temperatures (40°C to 140°C) facilitates the removal of acetic acid byproduct, driving the equilibrium towards the formation of the N-glycosidic bond in Compound V. This step is critical for establishing the correct stereochemistry at the anomeric center, which is preserved throughout the subsequent transformations to ensure biological activity in the final API.
Following the reduction of the nitro group to an amine (Compound VI), the construction of the fused imidazole ring (purine system) is achieved through condensation with triethyl orthoformate. This cyclization reaction is a classic example of formamidine synthesis, where the orthoformate acts as a C1 synthon. Under acidic conditions, the orthoformate reacts with the vicinal diamine functionality on the pyrimidine ring to close the five-membered imidazole ring, releasing ethanol as a byproduct. This intramolecular condensation is highly efficient, reportedly achieving yields around 90-91%. The mechanistic elegance here is that it avoids the need for harsh formylating agents or multi-step protections, directly assembling the purine scaffold (Compound VII) in a single operation. This efficiency is paramount for maintaining high overall yield in a linear synthesis.
How to Synthesize Cangrelor Intermediate Efficiently
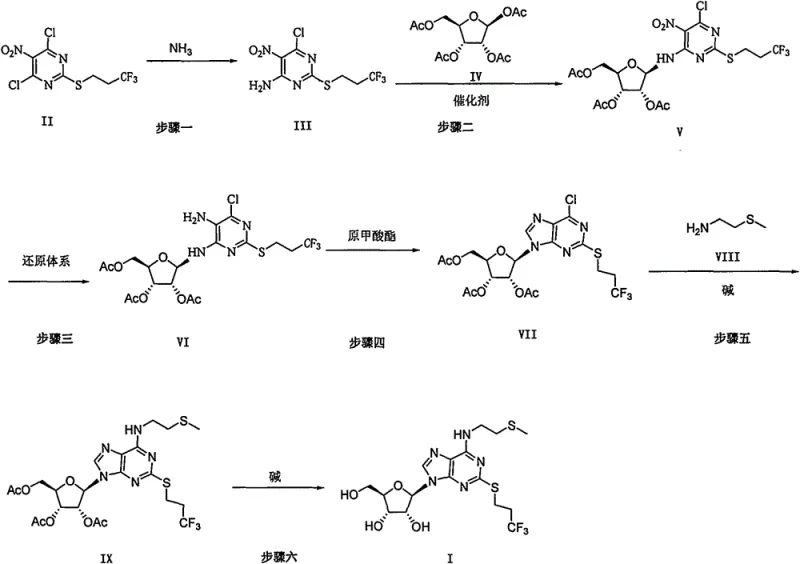
The synthesis of Cangrelor Intermediate (Formula I) via this patented route involves a logical progression of functional group manipulations that are well-suited for industrial application. The process begins with the selective amination of a dichloro-nitro-pyrimidine, followed by the critical attachment of the ribose sugar moiety. Subsequent steps involve the reduction of the nitro group to enable ring closure, followed by the introduction of the side chain amine and final deprotection. Each step has been optimized to minimize side reactions and maximize isolation yields, ensuring that the cumulative yield of the entire sequence remains commercially viable. For a detailed breakdown of the specific reaction parameters, stoichiometry, and workup procedures required to execute this synthesis in a GMP environment, please refer to the standardized guide below.
- Perform ammonolysis on Compound II using ammonia in a sealed tube to obtain Compound III.
- Couple amino Compound III with tetraacetyl ribose (IV) using an acid catalyst to form Compound V.
- Reduce the nitro group in Compound V using a reduction system (e.g., H2/Pd-C or Zn/AcOH) to yield amino Compound VI.
- Cyclize amino Compound VI with orthoformate to construct the purine ring, forming Compound VII.
- Couple Compound VII with 2-methylthioethylamine (VIII) in the presence of a base to afford Compound IX.
- Remove the acetyl protecting groups from Compound IX using a base to obtain the final Cangrelor intermediate (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CN105481922B synthesis route offers tangible strategic benefits beyond mere chemical novelty. The primary advantage is the drastic simplification of the supply chain risk profile. By eliminating the need for specialized, hazardous reagents like carbon disulfide and sodium hydride, facilities can reduce their insurance premiums and safety compliance costs. Moreover, the reliance on commodity chemicals such as ammonia, tetraacetyl ribose, and common organic solvents (ethanol, toluene, acetonitrile) ensures that raw material sourcing is stable and less susceptible to geopolitical fluctuations that often affect specialty fluorinated or iodinated reagents. This stability is crucial for maintaining continuous production schedules for high-purity API intermediates.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the elimination of expensive catalysts and the improvement in overall yield. Previous methods required stoichiometric amounts of costly Lewis acids like TMSOTF, which represent a significant line item in the bill of materials. By replacing these with catalytic amounts of cheaper Bronsted acids (like sulfuric or p-toluenesulfonic acid), the direct material costs are significantly reduced. Additionally, the higher yields reported in each step (often exceeding 90%) mean less raw material is wasted per kilogram of final product, leading to substantial cost savings in the overall manufacturing budget without compromising quality.
- Enhanced Supply Chain Reliability: The versatility of the reaction conditions provides a buffer against supply disruptions. For example, the reduction step can be performed using catalytic hydrogenation if hydrogen infrastructure is available, or alternatively using zinc powder or hydrazine if catalyst supply is constrained. This flexibility allows manufacturing sites to adapt quickly to market changes. Furthermore, the avoidance of custom-synthesized, scarce intermediates means that the production of Cangrelor intermediate is not bottlenecked by a single upstream supplier, thereby enhancing the reliability of the supply chain for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior. The absence of toxic gas generation (H2S) removes the need for complex scrubbing systems and specialized ventilation, simplifying the engineering requirements for scale-up. The use of aqueous workups and standard solvent recovery techniques aligns with green chemistry principles, reducing the volume of hazardous waste generated. This environmental friendliness facilitates faster regulatory approvals and permits for new manufacturing lines, accelerating the time-to-market for generic versions of Cangrelor and ensuring long-term sustainability of the production process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within patent CN105481922B, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of this supply source.
Q: How does this new synthesis route improve safety compared to prior art?
A: Unlike previous methods that utilized highly toxic carbon disulfide (CS2) and generated hazardous hydrogen sulfide (H2S) gas, this patented route avoids these dangerous reagents entirely, significantly enhancing operational safety and environmental compliance.
Q: What are the key advantages regarding raw material availability?
A: The process utilizes widely available starting materials such as tetraacetyl ribose and common amines, avoiding the reliance on scarce and expensive iodine-based reagents or specialized mercaptoadenosine precursors required in older methodologies.
Q: Does this method support large-scale commercial production?
A: Yes, the reaction conditions are mild (temperatures ranging from 30°C to 150°C) and utilize standard solvents like ethanol, toluene, and acetonitrile, making the process highly amenable to scale-up from kilogram to multi-ton manufacturing without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cangrelor Intermediate Supplier
The technical robustness of the CN105481922B pathway underscores the potential for high-quality, cost-effective production of Cangrelor intermediate. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such complex laboratory patents into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Cangrelor intermediate meets the exacting standards required for cardiovascular drug formulation.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples to validate the superior quality of our intermediates. Let us help you secure a stable, compliant, and economical supply chain for your next-generation antiplatelet therapies.
