Advanced Manufacturing of Tenofovir Alafenamide Intermediate via Optimized Condensation
Introduction to Next-Generation TAF Intermediate Manufacturing
The global demand for effective antiretroviral therapies continues to drive innovation in the synthesis of key pharmaceutical building blocks, specifically for the treatment of HIV-infected diseases. Patent CN109942632B discloses a breakthrough preparation method for (R)-9-[2-(phosphoryl phenol methoxyl) propyl] adenine, a critical intermediate in the production of Tenofovir Alafenamide (TAF). This novel approach addresses long-standing inefficiencies in prior art by utilizing an optimized condensation reaction in N-methyl pyrrolidone (NMP) solvent. Unlike traditional methods that suffer from excessive solvent consumption and prolonged reaction times, this technology leverages a precise balance of organic bases and catalysts to achieve high yields and exceptional purity. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a move towards more sustainable and cost-effective manufacturing protocols that do not compromise on the stringent quality standards required for antiviral drug substances.
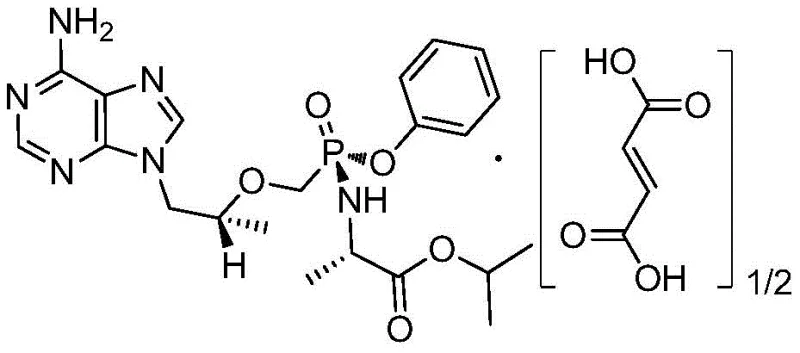
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this adenine derivative has been plagued by significant operational bottlenecks that hinder efficient commercial scale-up. One prominent prior art method, disclosed in WO2013052094, relies on acetonitrile as the primary solvent and requires refluxing for more than 48 hours to reach completion. This extended duration not only ties up reactor capacity but also incurs substantial energy costs. Furthermore, the process demands a large volume of solvent, typically around 8 volumes relative to the starting material, which complicates downstream solvent recovery and increases the environmental footprint. Another conventional route utilizes dicyclohexylcarbodiimide (DCC) as a condensing agent; while chemically viable, this generates stoichiometric amounts of N,N-Dicyclohexylurea (DCU) waste. The removal of DCU requires rigorous filtration and subsequent acid-base adjustments, which often introduce inorganic salt impurities that are notoriously difficult to purge from the final crystalline product, thereby jeopardizing the purity profile essential for regulatory approval.
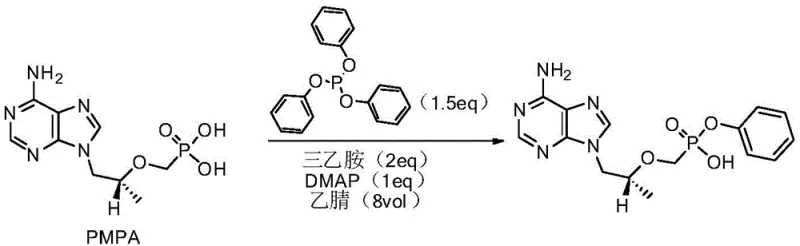
The Novel Approach
In stark contrast, the methodology outlined in CN109942632B introduces a paradigm shift by replacing problematic solvents and reagents with a more robust system. By employing N-methyl pyrrolidone (NMP) as the reaction medium, the process achieves superior solubility and thermal stability, allowing the reaction to proceed at elevated temperatures between 95°C and 140°C. This thermal advantage dramatically accelerates the kinetics, reducing the reaction window from days to merely 3 to 25 hours depending on the specific temperature setpoint. The new protocol also optimizes the molar ratios of triphenyl phosphite and catalyst, ensuring that the condensation proceeds with high atom economy. Crucially, the post-treatment is simplified to a straightforward extraction and pH adjustment sequence, eliminating the need for complex chromatographic purifications or extensive washing steps associated with urea byproduct removal. This streamlined workflow directly translates to reduced operational expenditure and a more reliable supply chain for high-purity pharmaceutical intermediates.
Mechanistic Insights into NMP-Mediated Condensation Catalysis
The core of this technological advancement lies in the synergistic interaction between the phosphorus source, the nucleophilic catalyst, and the high-boiling polar aprotic solvent. In this mechanism, triphenyl phosphite acts as the phosphorylating agent, reacting with the hydroxyl group of the PMPA starting material. The presence of 4-dimethylamino pyridine (DMAP) serves as a potent nucleophilic catalyst, activating the phosphorus center and facilitating the displacement of the phenoxy leaving group. Unlike lower boiling solvents that limit the thermal energy available to overcome activation barriers, NMP allows the system to operate at temperatures up to 140°C without requiring pressurized vessels. This thermal headroom ensures that the reaction equilibrium is driven strongly towards the product side, minimizing the accumulation of unreacted starting materials. Additionally, the choice of organic bases such as triethylamine or DIPEA plays a critical role in scavenging the acidic byproducts generated during the condensation, maintaining a neutral to slightly basic environment that protects the sensitive adenine moiety from degradation.
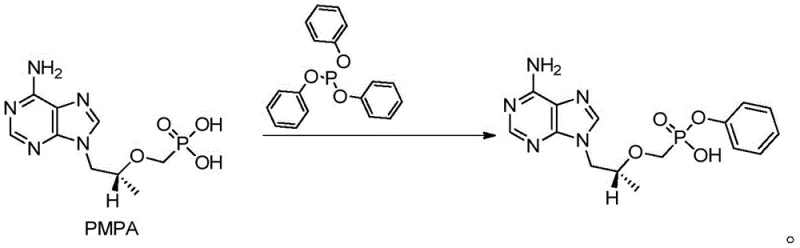
Impurity control is another pillar of this mechanistic design, particularly regarding the final isolation step. The patent specifies a precise pH adjustment range of 2 to 4 using concentrated hydrochloric acid during the work-up phase. This specific acidity window is engineered to protonate the product selectively, inducing precipitation while keeping potential organic impurities and residual catalysts in the aqueous or organic phase. By avoiding the harsh conditions associated with DCC-mediated couplings, which often require strong acids or bases for urea removal, this method preserves the structural integrity of the chiral center at the propyl chain. The result is a product with an HPLC purity exceeding 99%, demonstrating that the mechanistic choices made in solvent and reagent selection directly correlate with a cleaner impurity profile. For quality assurance teams, this implies a more predictable and controllable manufacturing process with fewer out-of-specification batches.
How to Synthesize (R)-9-[2-(phosphoryl phenol methoxyl) propyl] adenine Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and thermal management to maximize the benefits of the patented process. The standard operating procedure involves charging the reactor with the PMPA starting material, followed by the addition of the DMAP catalyst and triphenyl phosphite in the NMP solvent matrix. An organic base is then introduced to initiate the condensation, and the mixture is heated to the target temperature range. Monitoring the reaction progress via HPLC is recommended to ensure the starting material content drops below 0.1% before proceeding to work-up. The detailed standardized synthesis steps, including specific addition rates and cooling protocols, are provided in the guide below to ensure reproducibility across different manufacturing scales.
- Charge reactor with PMPA, DMAP catalyst, triphenyl phosphite, and organic base (DIPEA or TEA) in N-methyl pyrrolidone (NMP) solvent.
- Heat the mixture to 95-140°C and maintain reaction for 3 to 25 hours depending on temperature profile until HPLC shows <0.1% starting material.
- Cool reaction, extract with dichloromethane, adjust aqueous phase pH to 2-4 using hydrochloric acid to precipitate pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this novel synthesis route offers compelling advantages that extend beyond mere technical feasibility. The reduction in reaction time from over 48 hours to as little as 3 hours represents a massive increase in asset utilization, allowing manufacturers to produce significantly more batches per year with the same equipment footprint. This efficiency gain is critical for meeting the surging global demand for HIV treatments without necessitating capital-intensive plant expansions. Furthermore, the elimination of difficult-to-remove byproducts like DCU simplifies the supply chain for raw materials, as there is no longer a dependency on specialized filtration aids or extensive washing solvents. The overall process robustness ensures a consistent supply of high-quality intermediates, mitigating the risk of production delays that can ripple through the entire pharmaceutical value chain.
- Cost Reduction in Manufacturing: The transition to NMP solvent and the optimization of catalyst loading significantly lowers the variable costs associated with production. By reducing solvent volume from 8 volumes to as low as 1-5 mL/g, the expense related to solvent purchase, storage, and recovery is drastically curtailed. Additionally, the avoidance of expensive condensing agents like DCC and the reduction in energy consumption due to shorter reaction times contribute to a leaner cost structure. These savings can be passed down the supply chain, offering competitive pricing for the final API without sacrificing margin.
- Enhanced Supply Chain Reliability: The simplicity of the reagent profile enhances supply security, as triphenyl phosphite and NMP are commodity chemicals with stable global availability. Unlike processes relying on moisture-sensitive acyl chlorides or specialized coupling reagents that may face supply disruptions, this method utilizes robust, shelf-stable inputs. The shortened cycle time also means that inventory turnover is faster, allowing suppliers to respond more agilely to fluctuations in market demand and reducing the lead time for delivering high-purity pharmaceutical intermediates to downstream partners.
- Scalability and Environmental Compliance: The process is inherently designed for scale, avoiding the safety hazards associated with handling large quantities of thionyl chloride or generating massive amounts of solid urea waste. The simplified work-up involving extraction and pH adjustment generates less hazardous waste, aligning with increasingly strict environmental regulations. This green chemistry aspect not only reduces disposal costs but also future-proofs the manufacturing site against tightening regulatory frameworks, ensuring long-term operational continuity for the production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this technology outperforms legacy routes. Understanding these nuances is essential for technical teams evaluating process transfer and for procurement officers assessing supplier capabilities.
Q: What are the critical advantages of using NMP over acetonitrile in this synthesis?
A: N-methyl pyrrolidone (NMP) allows for significantly reduced solvent volumes (1-5 mL/g vs 8 vol) and enables higher reaction temperatures (up to 140°C), which drastically shortens reaction time from over 48 hours to as little as 3 hours while maintaining high yield.
Q: How does this method improve impurity profiles compared to DCC-mediated routes?
A: By avoiding dicyclohexylcarbodiimide (DCC), this process eliminates the formation of N,N-Dicyclohexylurea (DCU) waste, removing the need for complex filtration and acid-base regulation steps that often leave inorganic salt residues in the final API intermediate.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method utilizes commercially available reagents, requires low equipment specifications compared to anhydrous acyl chloride methods, and features a simple work-up procedure involving extraction and pH adjustment, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-9-[2-(phosphoryl phenol methoxyl) propyl] adenine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving antiviral drugs depends on the reliability and quality of the supply chain. Our technical team has extensively analyzed the pathway described in CN109942632B and possesses the expertise to execute this optimized condensation reaction with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from pilot scale to full industrial manufacturing. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for GMP API synthesis.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your Tenofovir Alafenamide projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial viability.
