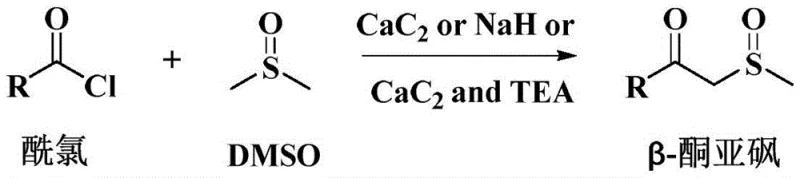
Scalable CaC2-Catalyzed Synthesis of Beta-Ketosulfoxides for Pharmaceutical Intermediates
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is documented in patent CN112939827B, which introduces a novel preparation method for beta-ketosulfoxides utilizing calcium carbide (CaC2) based catalysis. Beta-ketosulfoxides serve as critical precursors in the synthesis of active pharmaceutical ingredients (APIs) and agrochemicals, possessing broad-spectrum biological activities including anti-tumor and antiviral properties. Historically, the synthesis of these valuable compounds has been plagued by low efficiency and harsh reaction conditions. However, this new technology leverages the unique reactivity of CaC2, either alone or in conjunction with triethylamine (TEA), to react acyl chlorides with dimethyl sulfoxide (DMSO). This approach not only solves the longstanding technical problem of excessively low yields but also aligns with modern green chemistry principles by avoiding toxic heavy metals and reducing operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, chemists have struggled to find an optimal route for synthesizing beta-ketosulfoxides that balances yield, safety, and cost. Early methods reported by researchers such as Glen A. Russell relied on strong alkaline solutions like potassium tert-butoxide (KOtBu) or sodium hydride (NaH) to facilitate the reaction between esters and DMSO. While chemically feasible, these approaches present severe limitations, particularly when dealing with high molecular weight aromatic esters, where purity and yield often plummet. Furthermore, alternative oxidative methods involving noble metal catalysts like palladium or titanium, as explored by Sayed H.R. Abdi, introduce prohibitive costs and environmental liabilities due to the requirement for toxic oxidants and heavy metal residues. Perhaps most critically, previous attempts to react acyl chlorides directly with DMSO, considered one of the most economical pathways, resulted in dismal yields ranging from merely 5% to 16%, rendering them commercially unviable for mass production.
The Novel Approach
The methodology outlined in CN112939827B represents a paradigm shift by introducing CaC2 as a catalytic promoter. In the presence of CaC2, or more effectively, a combination of CaC2 and TEA, the reaction between acyl chlorides and DMSO undergoes a dramatic transformation in efficiency. As illustrated in the general reaction scheme below, this system facilitates the formation of the beta-ketosulfoxide backbone with remarkable precision. The yield improvements are substantial, jumping from single digits to a robust range of 45% to 78% under optimized conditions. This novel approach eliminates the need for expensive noble metals and avoids the violent exothermic risks associated with strong bases like NaH, offering a mild, room-temperature protocol that is inherently safer and easier to control on an industrial scale.
Mechanistic Insights into CaC2-Catalyzed Sulfoxidation
The efficacy of the CaC2/TEA system lies in its ability to generate reactive intermediates under mild conditions without the aggressive basicity that leads to substrate decomposition. When CaC2 interacts with the reaction medium, it likely serves as a source of acetylide ions or acts as a solid base that activates the DMSO molecule, facilitating the nucleophilic attack on the acyl chloride carbonyl carbon. The addition of TEA further modulates the reaction environment, stabilizing the transition state and preventing side reactions that typically plague acyl chloride chemistry. Experimental data indicates that a molar ratio of CaC2 to TEA of approximately 1:1 is optimal, maximizing the yield to 78% for substrates like p-nitrobenzoyl chloride. This synergistic effect ensures that the reaction proceeds smoothly at room temperature within just 0.5 hours, minimizing energy consumption and thermal stress on sensitive functional groups.
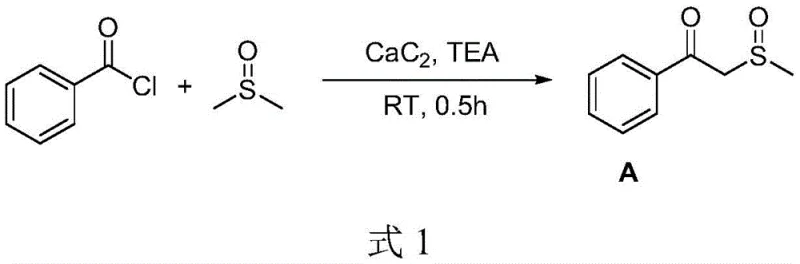
Impurity control is another critical aspect where this mechanism excels. Traditional methods using NaH often result in uncontrollable spraying of reaction materials if the feed rate is not meticulously managed, leading to safety incidents and inconsistent product quality. In contrast, the CaC2-mediated process is characterized by its温和 (mild) nature. The solid nature of CaC2 allows for a controlled release of reactive species, significantly reducing the formation of by-products and simplifying the downstream purification process. For instance, the synthesis of beta-ketosulfoxide derivatives from various substituted benzoyl chlorides, such as the conversion of benzoyl chloride shown below, demonstrates the robustness of this catalytic cycle across different electronic environments, ensuring high purity specifications required for pharmaceutical applications.
How to Synthesize Beta-Ketosulfoxide Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and addition rates to maximize the benefits of the CaC2/TEA catalytic system. The process is designed to be operationally simple, utilizing DMSO both as a reactant and a solvent, which streamlines the workflow. By maintaining an excess of DMSO and optimizing the molar ratios of the catalyst components, manufacturers can achieve consistent high yields. The following guide outlines the standardized procedure derived from the patent examples, providing a clear roadmap for R&D teams looking to adopt this technology for the production of high-purity pharmaceutical intermediates.
- Prepare the reaction mixture by combining DMSO, Calcium Carbide (CaC2), and Triethylamine (TEA) under inert gas protection.
- Slowly add the acyl chloride substrate to the mixture at low temperature (ice-water bath) over 10 minutes.
- Allow the reaction to proceed at room temperature for 0.5 hours, then quench with saturated ammonium chloride and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CaC2-catalyzed method offers compelling economic and logistical advantages over traditional synthetic routes. The primary driver for cost reduction is the replacement of expensive and hazardous reagents with commodity chemicals. Calcium carbide is an inexpensive, widely available industrial material, and triethylamine is a standard organic base, both of which are significantly cheaper than noble metal catalysts or specialized strong bases. This substitution drastically lowers the raw material cost per kilogram of the final product. Furthermore, the elimination of heavy metals removes the need for costly and time-consuming metal scavenging steps during purification, thereby reducing waste disposal costs and simplifying the manufacturing workflow.
- Cost Reduction in Manufacturing: The economic benefits extend beyond raw material costs to overall process efficiency. By operating at room temperature with short reaction times of approximately 0.5 hours, the method significantly reduces energy consumption associated with heating or cryogenic cooling. The high yields achieved, often exceeding 70% for optimized substrates, mean less raw material is wasted, directly improving the atom economy of the process. Additionally, the simplified workup procedure involving standard quenching and extraction reduces labor hours and solvent usage, contributing to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available reagents. Unlike specialized catalysts that may face supply bottlenecks or long lead times, CaC2 and TEA are produced at massive scales globally, ensuring a stable and continuous supply. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, such as using industrial grade CaC2 versus high-purity grades, which further secures the supply chain against fluctuations in reagent availability and pricing.
- Scalability and Environmental Compliance: From a scalability perspective, the mild and controllable nature of the reaction makes it ideal for commercial scale-up. The absence of violent exotherms allows for safer operation in large reactors, reducing the risk of batch failures. Environmentally, the process is greener as it avoids toxic heavy metals and generates acetylene gas as a by-product, which can potentially be captured and utilized, aligning with increasingly stringent environmental regulations and sustainability goals for chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the CaC2-catalyzed synthesis of beta-ketosulfoxides. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and advantages of this method for potential partners and stakeholders.
Q: Why is Calcium Carbide preferred over Sodium Hydride for this synthesis?
A: Calcium Carbide offers a significantly milder reaction profile compared to Sodium Hydride. While NaH can cause violent exothermic reactions and safety hazards if feed rates are not perfectly controlled, CaC2 ensures a stable, controllable process while maintaining high yields up to 78%.
Q: What is the typical yield improvement with the CaC2/TEA system?
A: Traditional acyl chloride and DMSO methods often suffer from yields as low as 5% to 16%. The novel CaC2/TEA catalytic system drastically improves this efficiency, achieving yields between 45% and 78%, making industrial production viable.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the method is highly scalable. It utilizes inexpensive, commodity chemicals like CaC2 and operates at room temperature with short reaction times (0.5h), eliminating the need for expensive noble metal catalysts or complex cryogenic setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Ketosulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the CaC2-catalyzed synthesis method for producing high-quality beta-ketosulfoxides. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a manufacturing setting. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the exacting standards required for API intermediates and fine chemicals. We are equipped to handle the specific nuances of this chemistry, from optimizing the CaC2/TEA ratios to managing the safe handling of acetylene by-products.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data for our beta-ketosulfoxide portfolio and discuss route feasibility assessments that can accelerate your development timeline while reducing overall production costs.
