Advanced Synthesis of Bis-3-butenyl Fluorinated Terphenyls for Next-Gen LCD Applications
Advanced Synthesis of Bis-3-butenyl Fluorinated Terphenyls for Next-Gen LCD Applications
The rapid evolution of display technologies, particularly in the realms of STN-LCD and TFT-LCD panels, demands liquid crystal materials that offer superior response speeds, wider operating temperature ranges, and enhanced optical anisotropy. Patent CN102153441A introduces a significant breakthrough in this domain by disclosing a novel class of fluorinated terphenyl liquid crystal compounds containing dual 3-butenyl side chains. These molecules are engineered to address the critical performance bottlenecks of legacy materials, specifically targeting improvements in viscosity and dielectric anisotropy. For R&D directors and procurement specialists in the electronic chemicals sector, understanding the synthetic accessibility and structural advantages of these monomers is paramount. The patent outlines a robust, four-step synthetic pathway that leverages well-established organometallic chemistry to construct the complex terphenyl core with high precision. This report provides a deep technical analysis of the patented methodology, evaluating its potential for commercial scale-up and its strategic value in the supply chain of high-performance display materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of liquid crystal monomers has been plagued by a trade-off between optical performance and physical stability. Conventional terphenyl derivatives often suffered from high viscosity, which directly impedes the response time of the liquid crystal cell, a critical metric for video and gaming applications. Furthermore, many existing compounds exhibited poor low-temperature solubility, leading to crystallization issues in cold environments, or lacked sufficient UV stability, causing degradation over the lifespan of the display device. Prior art structures, such as those referenced in the background of the patent, frequently demonstrated inadequate clearing points or insufficient birefringence values, limiting their utility in high-resolution, fast-switching panels. The synthesis of these older generations often involved harsh conditions, multiple protection-deprotection sequences, or expensive catalysts that drove up the cost of goods sold (COGS) and complicated waste management protocols. These inefficiencies created a significant barrier for procurement managers seeking reliable sources of high-performance intermediates without inflating production budgets.
The Novel Approach
The methodology described in CN102153441A represents a paradigm shift towards efficiency and modularity in liquid crystal synthesis. By utilizing a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction as the cornerstone step, the inventors have established a convergent route that assembles the terphenyl backbone from readily available precursors. This approach allows for the precise placement of fluorine atoms, which are crucial for lowering viscosity and tuning dielectric properties, without compromising the thermal stability of the molecule. The subsequent functionalization steps—reduction, halogenation, and Grignard coupling—are executed under relatively mild conditions, typically ranging from ambient temperature to moderate reflux, thereby minimizing energy consumption and safety risks. This streamlined process not only enhances the overall yield but also simplifies the purification workflow, as evidenced by the successful isolation of high-purity crystals via recrystallization in the provided embodiments. For supply chain stakeholders, this translates to a more predictable manufacturing timeline and reduced dependency on exotic reagents.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling and Grignard Alkylation
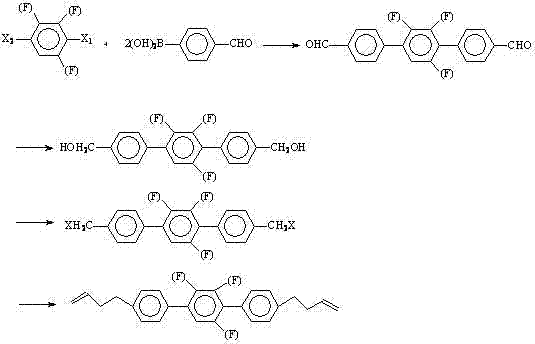
The core of this synthetic strategy relies on the robustness of the Suzuki coupling mechanism to form the carbon-carbon bonds linking the phenyl rings. In the first step, p-formylphenylboronic acid reacts with a difluoro-diiodobenzene derivative (Compound II) in the presence of a palladium catalyst, such as tetrakis(triphenylphosphine)palladium(0), and a base like potassium carbonate or sodium hydroxide. The mechanism proceeds through the oxidative addition of the aryl iodide to the Pd(0) species, followed by transmetallation with the boronate complex and reductive elimination to forge the biaryl linkage. This step is critical for establishing the rigid rod-like structure essential for liquid crystallinity. The choice of solvent system, often a mixture of toluene, ethanol, and water, facilitates the solubility of both organic and inorganic components, ensuring homogeneous reaction kinetics. Following the formation of the dialdehyde intermediate, the pathway diverges into standard functional group interconversions. The reduction of the aldehyde groups to hydroxymethyl groups using potassium borohydride is a highly chemoselective transformation that preserves the sensitive fluorine substituents and the aromatic integrity of the core.
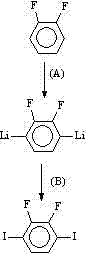
Subsequent halogenation converts the alcohol functionalities into reactive leaving groups, typically chlorides or bromides, using reagents like triphenylphosphine-carbon tetrabromide or thionyl chloride. This activation is a prerequisite for the final chain-extension step. The installation of the terminal 3-butenyl chains is achieved via a Grignard reaction, where the dihalide intermediate reacts with allylmagnesium chloride or bromide. This nucleophilic substitution effectively extends the molecular length and introduces the flexible alkenyl tails that promote mesophase formation. The patent highlights that this final coupling can be performed with or without additional copper catalysts, offering flexibility in process optimization. Crucially, the synthesis of the central difluoro-diiodobenzene intermediate itself is detailed, involving a directed lithiation of difluorobenzene followed by iodination. This upstream control ensures the regiochemical purity of the starting material, which is vital for preventing the formation of isomeric impurities that could degrade the electro-optical performance of the final liquid crystal mixture.
How to Synthesize Bis-3-butenyl Fluorinated Terphenyl Efficiently
The synthesis of these advanced liquid crystal monomers requires precise control over reaction parameters to ensure high purity and yield. The patented process delineates a clear, four-stage protocol that begins with the assembly of the aromatic core and concludes with the attachment of the alkenyl side chains. Operators must pay close attention to the stoichiometry of the Grignard reagent and the temperature profiles during the exothermic coupling steps to prevent side reactions. The use of anhydrous conditions is imperative during the organometallic stages to avoid quenching of the reactive intermediates. While the general procedure is robust, minor adjustments in solvent ratios or catalyst loading may be necessary when scaling from laboratory glassware to pilot plant reactors. For a comprehensive, step-by-step guide including specific molar equivalents, reaction times, and workup procedures, please refer to the standardized synthesis instructions provided below.
- Perform a palladium-catalyzed Suzuki cross-coupling between p-formylphenylboronic acid and a difluoro-diiodobenzene derivative to form the terphenyl backbone.
- Reduce the resulting dialdehyde intermediate to a diol using potassium borohydride or sodium borohydride under controlled thermal conditions.
- Convert the hydroxymethyl groups to halomethyl groups using triphenylphosphine-carbon tetrabromide or thionyl chloride.
- Execute a final Grignard coupling with allyl magnesium halide to install the terminal 3-butenyl chains, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers tangible benefits for organizations aiming to optimize their liquid crystal supply chains. The reliance on commodity chemicals such as toluene, ethanol, and tetrahydrofuran reduces the risk of supply disruptions associated with specialized solvents. Furthermore, the avoidance of cryogenic conditions in the main coupling steps (unlike some lithiation-heavy routes) lowers the barrier for manufacturing in facilities with standard cooling infrastructure. This accessibility directly contributes to cost reduction in display material manufacturing by minimizing capital expenditure on specialized equipment. The high conversion rates reported in the patent embodiments suggest that raw material utilization is efficient, reducing the volume of waste streams that require treatment. For procurement managers, this means a more stable pricing model and a lower total cost of ownership for the intermediate. Additionally, the modular nature of the synthesis allows for the potential production of various analogues by simply swapping the boronic acid or the dihalide component, providing supply chain agility to meet diverse customer specifications without retooling entire production lines.
- Cost Reduction in Manufacturing: The elimination of complex protection-deprotection sequences and the use of catalytic rather than stoichiometric amounts of expensive palladium species significantly lower the direct material costs. The ability to perform reactions at moderate temperatures (e.g., 50-110°C for the coupling step) reduces energy consumption compared to high-temperature processes. Moreover, the straightforward purification via recrystallization minimizes the need for resource-intensive chromatographic separations, further driving down operational expenses.
- Enhanced Supply Chain Reliability: The starting materials, including p-formylphenylboronic acid and various fluorinated benzenes, are widely available from global chemical suppliers, mitigating the risk of single-source dependency. The robustness of the Suzuki coupling against moisture and oxygen, relative to other organometallic reactions, increases the tolerance of the process, leading to fewer batch failures and more consistent delivery schedules. This reliability is crucial for maintaining the continuous operation of downstream LCD panel fabrication lines.
- Scalability and Environmental Compliance: The process generates primarily inorganic salts and organic solvents as byproducts, which are manageable within standard industrial waste treatment frameworks. The absence of heavy metal contaminants in the final product, ensured by rigorous filtration and washing steps described in the patent, aligns with stringent environmental regulations for electronic materials. The scalability is evidenced by the use of standard unit operations like reflux, filtration, and distillation, which translate seamlessly from kilogram to multi-ton production scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorinated terphenyl compounds. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these nuances is essential for integrating this technology into existing product portfolios.
Q: What are the key physical properties of this fluorinated terphenyl monomer?
A: According to patent CN102153441A, the compound exhibits a high clearing point, low viscosity, large birefringence (Δn), and excellent low-temperature solubility, making it ideal for TN, STN, and VA mode liquid crystal displays.
Q: How does the new synthesis route improve upon conventional methods?
A: The patented method utilizes mild reaction conditions (20-150°C) and avoids harsh reagents where possible. It employs a modular Suzuki coupling followed by standard functional group transformations, resulting in higher conversion rates and easier purification compared to older multi-step condensations.
Q: Is this process scalable for industrial production?
A: Yes, the patent explicitly states the method is suitable for industrialized production. The use of common solvents like toluene, THF, and ethanol, along with commercially available catalysts like tetrakis(triphenylphosphine)palladium(0), facilitates easy scale-up from gram to tonnage quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Terphenyl Supplier
As the demand for high-performance liquid crystal materials continues to surge, partnering with a manufacturer that possesses deep technical expertise and scalable capabilities is essential. NINGBO INNO PHARMCHEM stands at the forefront of fine chemical synthesis, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive organometallic reactions required for synthesizing complex fluorinated terphenyls, ensuring stringent purity specifications are met for every batch. With rigorous QC labs and a commitment to process optimization, we guarantee the consistency and quality necessary for advanced electronic applications.
We invite you to collaborate with our technical team to explore how this patented technology can enhance your product offerings. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to support your R&D and sourcing strategies, ensuring a seamless transition from laboratory concept to commercial reality.
