Advanced Synthesis of 1,1-Difluorotetrahydronaphthalene Derivatives for Next-Generation Electronic Chemical Manufacturing
The rapid evolution of flat-panel display technology, particularly Thin Film Transistor Liquid Crystal Displays (TFT-LCDs), demands materials that can simultaneously offer fast response times, low operating voltages, and exceptional environmental stability. Patent CN102888227A introduces a groundbreaking class of 1,1-difluorotetrahydronaphthalene liquid crystal compounds that address these critical industry pain points. Unlike traditional biphenyl-based mesogens which often suffer from high melting points and limited solubility, these novel structures incorporate a rigid tetrahydronaphthalene core bridged by two fluorine atoms. This specific architectural modification not only enhances the dielectric anisotropy required for low-voltage driving but also significantly improves the charge retention rate, a parameter vital for preventing image flicker in high-resolution screens. As a reliable electronic chemical supplier, understanding the nuanced synthesis and application of such high-performance monomers is essential for maintaining competitiveness in the optoelectronic materials market.
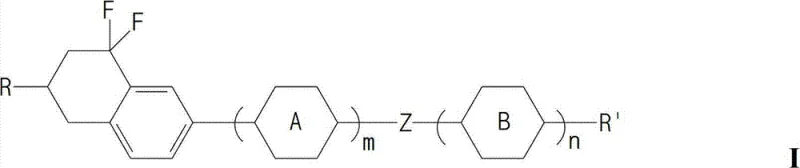
The structural integrity of these compounds allows for precise tuning of the birefringence (Δn) and viscosity, enabling formulators to optimize response times without sacrificing thermal stability. The patent details a comprehensive library of derivatives where the terminal alkyl chains and linking groups can be varied to match specific host mixture requirements. This flexibility ensures that the material can be seamlessly integrated into existing manufacturing lines for smartphones, tablets, and large-format televisions. By leveraging this technology, manufacturers can achieve a significant reduction in threshold voltage, directly translating to lower power consumption for end-user devices, a key selling point in the current energy-conscious consumer electronics landscape.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional liquid crystal materials often rely on simple biphenyl or phenyl-cyclohexane cores which, while easy to synthesize, face inherent physical limitations. One major drawback is the strong conjugation in biphenyl systems, which leads to higher melting points and poorer solubility in low-viscosity hosts, restricting the operating temperature range of the final display. Furthermore, conventional compounds frequently exhibit insufficient dielectric anisotropy, necessitating higher driving voltages that increase power draw and heat generation. From a supply chain perspective, older synthetic routes often involve harsh conditions or unstable intermediates that complicate scale-up and introduce impurities detrimental to voltage holding ratios (VHR). These limitations create a bottleneck for developing next-generation displays that require faster switching speeds and wider viewing angles, forcing R&D teams to seek more sophisticated molecular architectures.
The Novel Approach
The innovative strategy presented in this patent utilizes a 1,1-difluorotetrahydronaphthalene scaffold that fundamentally alters the electronic and steric properties of the mesogen. The introduction of the gem-difluoro group at the bridgehead position effectively breaks the conjugation between the aromatic rings, resulting in a marked depression of the melting point and enhanced mutual solubility with other liquid crystal components. This structural feature also generates a strong lateral dipole moment, which drastically increases the dielectric anisotropy without compromising the rotational viscosity. Consequently, the novel approach enables the formulation of mixtures with lower saturation voltages and improved UV resistance, addressing the degradation issues common in outdoor or high-brightness applications. This represents a paradigm shift in electronic chemical manufacturing, moving towards highly engineered fluorinated cores that deliver superior electro-optical performance.
Mechanistic Insights into DAST Fluorination and Suzuki Coupling
The synthetic pathway relies heavily on two critical transformations: the fluorination of the ketone intermediate and the final carbon-carbon bond formation. The fluorination step employs Diethylaminosulfur Trifluoride (DAST) to convert the carbonyl group of the tetralone precursor into the gem-difluoro moiety. This reaction proceeds via a nucleophilic attack of the fluoride ion on the sulfur-activated carbonyl carbon, followed by elimination. Controlling the exotherm during this step is crucial, as DAST is moisture-sensitive and the reaction can be vigorous. The patent specifies refluxing in methylene chloride until GC analysis confirms complete conversion, ensuring that no residual ketone remains to act as an impurity that could degrade the VHR of the final product. This high-fidelity transformation is key to achieving the desired dielectric properties.
Following fluorination, the assembly of the final molecule is achieved through a Palladium-catalyzed Suzuki-Miyaura cross-coupling reaction. This step links the fluorinated tetralin bromide with a functionalized boronic acid derivative. The mechanism involves the oxidative addition of the aryl bromide to the Pd(0) catalyst, transmetallation with the boronate species activated by base, and reductive elimination to form the biaryl bond. The choice of a toluene/ethanol/water solvent system facilitates the solubility of both organic and inorganic phases, promoting efficient mass transfer. This catalytic cycle is highly robust, allowing for the coupling of complex, multi-ring systems with high yields (up to 80% in reported embodiments). The ability to tolerate various functional groups during this coupling provides the versatility needed to generate the diverse library of compounds described in the patent.
How to Synthesize 1,1-Difluorotetrahydronaphthalene Efficiently
The preparation method outlined in the patent offers a scalable, six-step sequence that balances yield with operational simplicity, making it highly attractive for commercial production. The process begins with the formation of a Grignard reagent from a bromoalkane, which is then reacted with bromobenzene acetaldehyde to build the carbon skeleton. Subsequent bromination and malonate condensation set up the ring closure, which is effected using polyphosphoric acid (PPA) to form the tetralone core. The critical fluorination and coupling steps follow to finalize the structure. This route avoids exotic reagents and utilizes standard unit operations like extraction and crystallization, facilitating technology transfer from lab to pilot plant. For a detailed breakdown of the specific reaction conditions and stoichiometry, please refer to the standardized guide below.
- Perform Grignard reaction between bromoalkane and bromobenzene acetaldehyde at -5°C to 0°C to form intermediate alcohol A.
- Convert intermediate A to dibromide B using bromine and triphenylphosphine catalyst at low temperature.
- Condense intermediate B with diethyl malonate using sodium metal in ethanol to obtain carboxylic acid derivative C.
- Execute ring-closure cyclization of C using polyphosphoric acid (PPA) at 75-85°C to form the tetralone core D.
- Fluorinate the carbonyl group of D using DAST reagent to generate the key 1,1-difluoro intermediate E.
- Complete the synthesis via Palladium-catalyzed Suzuki coupling of E with corresponding boronic acid F to yield the final liquid crystal compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain management perspective, the synthesis route described in CN102888227A offers distinct advantages regarding cost stability and raw material security. The starting materials, such as bromobenzene acetaldehyde and common bromoalkanes, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source bottlenecks. Furthermore, the synthetic steps utilize well-established reactions like Grignard addition and Suzuki coupling, which are widely understood by contract manufacturing organizations (CMOs), ensuring that the technology can be licensed or outsourced with minimal friction. The high yields reported in the embodiments, particularly in the final coupling step, suggest a material-efficient process that minimizes waste generation and maximizes output per batch, directly impacting the cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The elimination of complex, multi-step protection-deprotection sequences often found in older fluorinated LC syntheses leads to a streamlined process flow. By utilizing direct fluorination with DAST and efficient PPA cyclization, the number of isolation steps is minimized, which reduces solvent consumption and labor hours. Although specific percentage savings cannot be quantified without a full techno-economic analysis, the qualitative reduction in unit operations inherently lowers the manufacturing overhead. Additionally, the high purity achievable through simple recrystallization reduces the need for expensive preparative HPLC purification, further driving down production costs.
- Enhanced Supply Chain Reliability: The robustness of the Suzuki coupling step, which tolerates a wide range of substituents, allows for the use of diverse boronic acid building blocks. This modularity means that if a specific side-chain precursor faces supply constraints, alternative analogues can often be synthesized using the same core intermediate without revalidating the entire process. This flexibility ensures continuity of supply for downstream panel manufacturers. Moreover, the stability of the final 1,1-difluorotetrahydronaphthalene products implies a longer shelf life, reducing inventory write-offs and allowing for strategic stockpiling during periods of high demand.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like toluene, ethanol, and methylene chloride which have established recovery and recycling protocols in modern chemical plants. The use of PPA for cyclization, while requiring careful handling, avoids the generation of heavy metal waste associated with some Lewis acid catalysts. The high atom economy of the coupling reaction and the efficient conversion in the fluorination step contribute to a greener manufacturing profile. This alignment with environmental, social, and governance (ESG) goals is increasingly important for securing contracts with major electronics brands that prioritize sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating these materials into their product portfolios.
Q: What are the key performance advantages of 1,1-difluorotetrahydronaphthalene liquid crystals?
A: These compounds exhibit exceptional structural stability, high charge retention rates, and large dielectric anisotropy. The introduction of the difluoro bridge disrupts biphenyl conjugation, lowering melting points and improving solubility while enhancing UV resistance.
Q: How does the DAST fluorination step impact the overall yield?
A: The use of DAST (diethylaminosulfur trifluoride) allows for efficient conversion of the carbonyl group to the difluoro moiety under reflux conditions. In optimized embodiments, this step achieves yields around 85%, contributing to a robust overall process efficiency exceeding 80%.
Q: Are these compounds suitable for VA-TFT and IPS display modes?
A: Yes, experimental data confirms their applicability across TN, IPS, FFS, ADS, and VA-TFT patterns. They effectively reduce threshold voltage and adjust optical anisotropy (Δn), making them versatile additives for complex liquid crystal mixtures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Difluorotetrahydronaphthalene Supplier
The technical potential of 1,1-difluorotetrahydronaphthalene derivatives is clear, but realizing this potential requires a manufacturing partner with deep expertise in complex organic synthesis and strict quality control. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace impurity analysis via GC-MS and HPLC, which is critical for maintaining the high voltage holding ratios required in premium display applications. We understand that in the electronic chemical sector, even ppm-level impurities can dictate the success or failure of a display panel.
We invite you to collaborate with us to leverage this patented technology for your next-generation display projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and formulation needs. Please contact us to request specific COA data for our available grades and to discuss route feasibility assessments for custom analogues. By partnering with us, you secure not just a chemical supplier, but a strategic ally committed to advancing the performance and reliability of your electronic materials supply chain.
