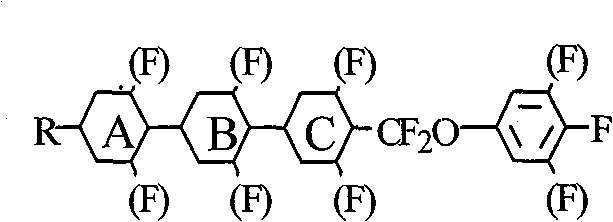
Advanced One-Pot Synthesis of Difluoromethoxy Bridge Compounds for High-Performance LCDs
The rapid evolution of Thin Film Transistor Liquid Crystal Display (TFT-LCD) technology has placed unprecedented demands on the performance of liquid crystal materials, specifically requiring lower viscosity and higher dielectric anisotropy to enable faster response times and reduced driving voltages. Patent CN101565343B addresses these critical industry needs by disclosing a highly efficient preparation method for inducing difluoromethoxy central bridge bonds (-CF2O-) within complex organic molecules. This specific chemical moiety is pivotal for creating monomer liquid crystals that exhibit the precise electro-optical properties required for next-generation displays. The invention utilizes a novel 'one-pot' synthetic strategy that leverages dimercaptopropane-trifluoromethane sulfonic acid to generate trifluoromethane sulfonate intermediates in situ, subsequently reacting them with phenols and fluorinating agents at low temperatures without the need for intermediate separation. This approach represents a significant technological leap forward, offering a robust pathway for the industrial production of high-value electronic chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of liquid crystal compounds containing the difluoromethoxy center bridged bond was plagued by significant economic and technical hurdles that hindered large-scale adoption. The dibromodifluoromethane method, for instance, relies on Grignard or lithium reagents which are not only sensitive to moisture but also suffer from low yields when applied to polycyclic systems, while the source material itself is relatively rare and expensive. Alternatively, the DAST method involves the conversion of esters to monothioesters using Lawesson's reagent followed by fluorination, a process characterized by the prohibitive cost of DAST and consistently suboptimal yields that drive up the final price of the liquid crystal material. Furthermore, the traditional two-step dimercaptopropane-trifluoromethayl sulfonic acid method requires the isolation and recrystallization of the trifluoromethanyl sulfonate salt; this purification step is technically difficult due to the low melting point of the salt and results in substantial material loss, ultimately compromising the overall efficiency and cost-effectiveness of the manufacturing process.
The Novel Approach
The methodology outlined in patent CN101565343B fundamentally reengineers this synthetic landscape by integrating the formation of the sulfonate intermediate and its subsequent fluorination into a single, continuous 'one-pot' operation. By eliminating the isolation and recrystallization steps associated with the traditional sulfonate method, the process avoids the inherent yield penalties of solid-liquid separations and the difficulties of handling low-melting salts. The reaction proceeds by first generating the trifluoromethanyl sulfonate under reflux conditions with water removal, followed by direct cooling and the addition of phenols, pyridine hydrogen fluoride, and bromine. This seamless transition between reaction stages not only simplifies the operational workflow but also ensures that the reactive intermediate is consumed immediately, minimizing decomposition and side reactions. Consequently, this approach delivers a simple, highly efficient, and industrially scalable route that significantly lowers the barrier to entry for producing high-performance liquid crystal monomers.
Mechanistic Insights into One-Pot Trifluoromethylation and Fluorination
The core of this chemical transformation lies in the in situ generation of a highly reactive trifluoromethyl sulfonate species from an organic acid precursor, typically an alkyl formate, substituted cyclohexyl formic acid, or substituted benzoic acid. In the presence of 1,3-dimercaptopropane and trifluoromethanesulfonic acid, the carboxylic acid undergoes activation to form the sulfonate ester without ever leaving the solution phase. This intermediate is then subjected to cryogenic conditions, typically ranging from -90°C to -60°C using liquid nitrogen-ethanol or acetone cooling baths, to stabilize the system before the introduction of the nucleophilic phenol component. The subsequent addition of pyridine hydrogen fluoride (HFPy) and bromine facilitates the critical fluorination and oxidative steps required to establish the difluoromethoxy bridge, effectively replacing the carbonyl oxygen functionality with the desired -CF2O- linkage while maintaining the structural integrity of the complex polycyclic backbone.
Impurity control in this rigorous synthetic pathway is achieved through precise stoichiometric management and optimized workup procedures that leverage the distinct solubility profiles of the reaction components. The patent specifies the use of sodium bicarbonate aqueous solutions to neutralize the acidic reaction mixture, followed by extraction and washing with sodium bisulfite to remove excess bromine and oxidative byproducts. Final purification is accomplished via column chromatography using silica gel as the stationary phase and petroleum ether or normal hexane as the mobile phase, followed by recrystallization. This multi-stage purification protocol ensures that the final product meets the stringent purity requirements of the electronics industry, with experimental embodiments demonstrating gas chromatographic purity levels reaching 99.5%, thereby effectively suppressing the formation of ionic impurities or unreacted starting materials that could degrade the performance of the final liquid crystal display.
How to Synthesize Difluoromethoxy Bridge Compounds Efficiently
The synthesis of these high-value intermediates requires strict adherence to the patented one-pot protocol to maximize yield and ensure reproducibility across different batches. The process begins with the dehydration reaction of the organic acid and dimercaptopropane-trifluoromethayl sulfonic acid in a toluene or toluene-isooctane mixed solvent system under reflux, utilizing a water trap to drive the equilibrium forward until anhydrous conditions are confirmed. Once the intermediate is formed, the reaction vessel must be cooled rapidly to cryogenic temperatures before the sequential addition of the phenol derivative, triethylamine, pyridine hydrogen fluoride, and bromine solution, ensuring that the exothermic nature of the fluorination is carefully managed. For a comprehensive understanding of the specific molar ratios, temperature gradients, and workup details required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- React organic acid with 1,3-dimercaptopropane and trifluoromethanesulfonic acid in toluene/octane under reflux to form the trifluoromethyl sulfonate intermediate in situ.
- Cool the reaction mixture to cryogenic temperatures (-70°C to -90°C) and directly add fluorophenols, pyridine-HF, and bromine without isolating the intermediate salt.
- Perform aqueous workup with sodium bicarbonate, followed by silica gel chromatography and recrystallization to obtain the high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot synthetic methodology offers profound advantages in terms of cost structure and operational reliability compared to legacy manufacturing routes. By consolidating what was previously a multi-step process involving isolation and recrystallization into a single continuous flow, the method drastically reduces the consumption of solvents, energy, and labor hours associated with intermediate handling and purification. This streamlining of the process flow translates directly into a more favorable cost of goods sold (COGS), allowing suppliers to offer competitive pricing for high-purity electronic chemicals without compromising on quality margins. Furthermore, the elimination of difficult recrystallization steps for low-melting salts removes a major bottleneck in production scheduling, enhancing the predictability of lead times and ensuring a more stable supply of critical raw materials for downstream LCD panel manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the significant increase in overall yield resulting from the avoidance of intermediate isolation losses. In traditional methods, the physical handling and recrystallization of the trifluoromethanyl sulfonate salt often result in substantial material attrition, whereas the one-pot method retains nearly all theoretical mass within the reaction vessel until the final product is formed. Additionally, the reduction in unit operations decreases the demand for auxiliary equipment and utilities, such as drying ovens and filtration systems, leading to substantial overhead savings that accumulate over large-scale production runs.
- Enhanced Supply Chain Reliability: From a logistics perspective, the simplified reaction scheme reduces the complexity of the manufacturing timeline, thereby mitigating the risk of delays caused by equipment bottlenecks or purification failures. The use of readily available starting materials such as organic acids and common fluorinating agents, rather than rare or highly specialized reagents like dibromodifluoromethane, ensures a more resilient supply chain that is less susceptible to market volatility. This stability is crucial for maintaining continuous production schedules for TFT-LCD mixtures, where any interruption in the supply of key monomers can halt entire assembly lines.
- Scalability and Environmental Compliance: The robustness of the one-pot method makes it inherently easier to scale from laboratory benchtop quantities to multi-ton commercial production without the need for complex process re-engineering. The consolidated waste stream, resulting from fewer purification steps and solvent exchanges, simplifies effluent treatment and reduces the environmental footprint of the manufacturing facility. This alignment with green chemistry principles not only aids in regulatory compliance but also appeals to end-users who are increasingly prioritizing sustainable sourcing practices in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of the difluoromethoxy bridge synthesis method described in patent CN101565343B. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on reaction conditions, purity standards, and applicability to various liquid crystal structures. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this technology into their existing production pipelines.
Q: What are the primary limitations of the conventional DAST method for CF2O bridge synthesis?
A: The conventional DAST method suffers from the high cost of the Lawesson reagent and DAST itself, coupled with generally lower yields compared to the novel one-pot sulfonate approach described in patent CN101565343B.
Q: How does the one-pot method improve yield compared to the traditional two-step sulfonate process?
A: The traditional two-step process requires the isolation and recrystallization of the trifluoromethyl sulfonate salt, which leads to significant purification losses. The one-pot method eliminates this isolation step, thereby retaining the intermediate in solution and drastically reducing material loss.
Q: What purity levels can be achieved with this new synthetic route?
A: According to the experimental embodiments in the patent, the one-pot method consistently achieves gas chromatographic purity levels of 99.5%, making it highly suitable for demanding electronic applications like TFT-LCDs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethoxy Compound Supplier
As the global demand for high-performance display materials continues to surge, partnering with a chemically proficient manufacturer is essential for securing a competitive edge in the market. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex one-pot fluorination processes described in recent patents can be executed with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the 99.5% GC purity benchmarks required for advanced TFT-LCD applications, guaranteeing that every batch meets the exacting standards of the optoelectronics industry.
We invite potential partners to engage with our technical procurement team to discuss how our advanced synthesis capabilities can optimize your supply chain for liquid crystal intermediates. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into how our implementation of the one-pot method can reduce your overall material costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific molecular targets, ensuring a seamless transition from development to full-scale commercialization.
