Revolutionizing LCD Material Synthesis: High-Yield Production of Trans,Trans-Alkylcyclohexyl Cyclohexanol Intermediates
Introduction to Advanced Liquid Crystal Intermediate Synthesis
The rapid evolution of Thin Film Transistor Liquid Crystal Displays (TFT-LCD) has placed unprecedented demands on the purity and performance of underlying liquid crystal materials. As the industry shifts towards high-resolution and low-power consumption displays, the need for specialized intermediates with precise stereochemical configurations has become critical. Patent CN101704724A introduces a groundbreaking methodology for the preparation of high-proportion trans,trans-4-(4'-alkylcyclohexyl)cyclohexyl alcohol compounds, which serve as vital precursors for alkyl bicyclohexane ether liquid crystals. These compounds are renowned for their low rotational viscosity and excellent mutual solubility, properties essential for enhancing the response speed of modern display panels. By leveraging a novel reduction-isomerization sequence, this technology addresses long-standing inefficiencies in the supply chain of electronic chemical manufacturing, offering a robust pathway for reliable liquid crystal intermediate supplier partnerships.
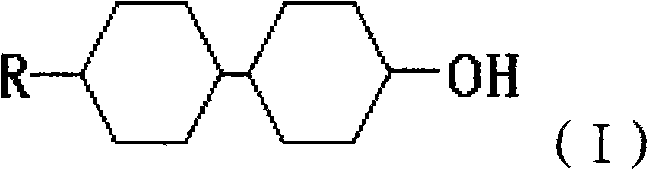
The significance of this patent lies in its ability to produce the thermodynamically stable trans,trans isomer with exceptional selectivity, directly impacting the quality of the final liquid crystal mixture. Traditional methods often struggle with cis/trans separation, leading to significant material loss and increased costs. This new approach not only simplifies the synthetic route but also aligns with green chemistry principles by avoiding hazardous oxidation steps associated with older technologies. For procurement managers and R&D directors alike, understanding the mechanistic advantages of this route is key to securing a competitive edge in the display materials market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trans,trans-4-(4'-alkylcyclohexyl)cyclohexyl alcohol relied heavily on the hydrogenation of trans-alkylcyclohexyl phenols. This conventional pathway, while chemically feasible, suffers from severe stereoselectivity issues, typically yielding a mixture dominated by the cis-isomer. To isolate the desired trans,trans product, manufacturers were forced to employ multiple, labor-intensive recrystallization steps, often resulting in overall yields as low as 20%. Furthermore, alternative routes involving the oxidation of alcohol mixtures to ketones utilized toxic重 oxidants like chromic acid or potassium permanganate, generating substantial hazardous waste and complicating environmental compliance. The reliance on cryogenic conditions in some lithium-ammonia reduction methods further hindered scalability, making industrial implementation difficult and costly. These bottlenecks created a fragile supply chain vulnerable to raw material fluctuations and regulatory pressures.
The Novel Approach
In stark contrast, the methodology disclosed in CN101704724A utilizes a direct reduction of trans-4-alkylcyclohexyl cyclohexyl ketones followed by a Lewis acid-catalyzed isomerization. This strategic shift eliminates the need for problematic phenol hydrogenation and subsequent oxidative steps. By employing borohydride reducing agents in a controlled solvent system, the process achieves high conversion rates under mild conditions. The subsequent introduction of a Lewis acid, such as aluminum trichloride, facilitates a stereoselective transposition that enriches the trans-isomer content to over 90% directly from the reaction mixture. This streamlined workflow drastically reduces the number of unit operations, minimizes solvent consumption, and significantly improves the overall mass balance of the production process, representing a paradigm shift in cost reduction in display material manufacturing.
Mechanistic Insights into Borohydride Reduction and Lewis Acid Isomerization
The core of this innovative synthesis lies in the precise control of stereochemistry during the reduction and isomerization phases. Initially, the trans-4-alkylcyclohexyl cyclohexyl ketone undergoes nucleophilic attack by the borohydride anion. The choice of solvent system, typically a combination of tetrahydrofuran (THF) and ethanol, plays a pivotal role in modulating the reactivity of the reducing agent and ensuring homogeneous reaction conditions. Following the initial reduction, the crude alcohol mixture contains a significant proportion of the less stable cis-isomer. The introduction of the Lewis acid catalyst triggers a carbocation-mediated rearrangement. The aluminum center coordinates with the hydroxyl group, facilitating the departure of water or the formation of an alkoxy-aluminum complex that allows for bond rotation and equilibration towards the thermodynamically favored trans,trans configuration.

This mechanistic pathway ensures that impurities and by-products are minimized, as the reaction conditions are tuned to favor the desired stereoisomer without requiring harsh thermal treatments. The acidic hydrolysis step subsequently quenches the reaction, releasing the free alcohol and regenerating the catalyst species. From an impurity control perspective, this method is superior because it avoids the formation of complex oxidation by-products common in older routes. The high selectivity reduces the burden on downstream purification, allowing for simpler recrystallization protocols to achieve the stringent purity specifications required for high-performance TFT-LCD applications. This level of control is essential for maintaining consistent batch-to-batch quality in commercial scale-up of complex electronic chemicals.
How to Synthesize Trans,Trans-4-(4'-alkylcyclohexyl)cyclohexanol Efficiently
Implementing this synthesis requires careful attention to temperature control and reagent stoichiometry to maximize the trans-isomer ratio. The process begins with the dissolution of the ketone starting material in an organic solvent, followed by the controlled addition of the borohydride solution at temperatures ranging from 0°C to 40°C. Once the reduction is complete, the reaction mixture is cooled, and the Lewis acid is introduced to drive the isomerization. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to guide process engineers in replicating this high-efficiency route.
- Dissolve trans-4-alkylcyclohexyl cyclohexyl ketone in THF and reduce with sodium borohydride in ethanol at 0-40°C.
- Add aluminum trichloride (Lewis acid) at 0-5°C to induce stereoselective transposition reaction.
- Perform acidic hydrolysis, extract with toluene, and recrystallize from ethanol to achieve >99.8% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible benefits that extend beyond mere chemical yield. The elimination of multi-step recrystallization and hazardous oxidation processes translates directly into reduced operational expenditures and lower waste disposal costs. By simplifying the manufacturing workflow, companies can mitigate risks associated with complex supply chains and reduce the lead time for high-purity liquid crystal intermediates. The robustness of the reaction conditions also enhances supply continuity, as the process is less sensitive to minor variations in raw material quality compared to cryogenic or high-pressure alternatives.
- Cost Reduction in Manufacturing: The dramatic increase in yield from approximately 20% in traditional methods to over 80% in this new process fundamentally alters the cost structure of the intermediate. By maximizing the conversion of raw materials into the desired product, manufacturers can significantly reduce the cost per kilogram of the final API or electronic material. Furthermore, the avoidance of expensive heavy metal oxidants and the reduction in solvent usage for recrystallization contribute to substantial cost savings. This efficiency allows for more competitive pricing strategies in the global market for display materials.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents such as sodium borohydride and aluminum trichloride ensures that the supply chain is not dependent on exotic or hard-to-source catalysts. The mild reaction temperatures eliminate the need for specialized cryogenic equipment, making the process adaptable to a wider range of manufacturing facilities. This flexibility enhances the resilience of the supply network, ensuring consistent delivery schedules even during periods of high market demand or logistical disruptions.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction times and temperatures that are easily managed in large-scale reactors. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, reducing the compliance burden on manufacturing sites. The ability to produce high-purity materials with a smaller environmental footprint positions suppliers as preferred partners for multinational corporations committed to sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the patent data and are intended to clarify the feasibility and advantages of adopting this technology for industrial production. Understanding these details is crucial for making informed decisions about process integration and supplier selection.
Q: What is the primary advantage of this new synthesis route over traditional phenol hydrogenation?
A: The new route bypasses the low-yield recrystallization steps of the phenol route, increasing overall yield from ~20% to over 80% while eliminating heavy metal oxidation waste.
Q: How is the high trans-isomer ratio (>90%) achieved in this process?
A: The process utilizes a specific Lewis acid (AlCl3) mediated isomerization step immediately following reduction, which thermodynamically favors the formation of the stable trans,trans configuration.
Q: Is this process scalable for industrial TFT-LCD material production?
A: Yes, the reaction operates at mild temperatures (0-40°C) using common solvents like THF and ethanol, making it highly suitable for large-scale commercial manufacturing without cryogenic requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans,Trans-4-(4'-alkylcyclohexyl)cyclohexanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of next-generation display technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101704724A are fully realized in practical manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of liquid crystal intermediate meets the exacting standards required by the electronics industry. Our commitment to technical excellence ensures that our clients receive materials that consistently perform in their final formulations.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can support your production goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
