Revolutionizing Lacosamide Intermediate Production: A Deep Dive into One-Step Chiral Resolution Technology
The global demand for third-generation antiepileptic drugs continues to surge, with Lacosamide standing out as a pivotal treatment for partial-onset seizures and neuropathic pain due to its favorable tolerability profile. At the heart of manufacturing this critical active pharmaceutical ingredient lies the production of its key chiral building block, R-(-)-2-amino-3-methoxyl-N-benzyl propionamide. Patent CN102516114B introduces a groundbreaking preparation method for the D-tartrate salt of this intermediate, offering a robust solution to the longstanding challenges of chiral separation in fine chemical manufacturing. This technology leverages a novel one-step resolution strategy that not only simplifies the operational workflow but also dramatically enhances the economic viability of producing high-purity pharmaceutical intermediates. By integrating a seamless recycling loop for the unwanted enantiomer, this process addresses both cost efficiency and environmental sustainability, making it an ideal candidate for large-scale industrial adoption by forward-thinking pharmaceutical manufacturers.
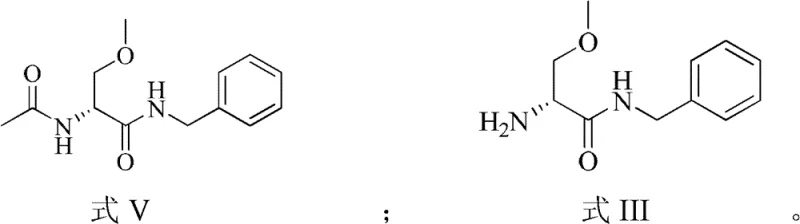
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amino acid derivatives like the Lacosamide intermediate has been plagued by inefficient multi-step protocols that burden production schedules and inflate costs. Traditional approaches often rely on starting from expensive chiral pool materials such as D-Serine, which necessitates a lengthy sequence of protection, functionalization, and deprotection reactions to install the methoxy and benzyl groups. Alternatively, methods starting from achiral substrates typically involve a cumbersome two-stage chiral separation process where salification and fractional crystallization must be conducted in entirely different solvent systems. For instance, prior art such as International Patent Application WO 2011092559 describes a method requiring an initial salification in an aprotic polar solvent followed by a distinct splitting step in a mixed protonic-aprotic solvent system. This disjointed approach demands extensive solvent swapping, complex filtration procedures, and significant energy input for solvent recovery, creating bottlenecks that hinder the commercial scale-up of complex pharmaceutical intermediates. Furthermore, the reliance on multiple solvent types increases the hazard profile of the manufacturing process and complicates waste stream management, posing significant challenges for supply chain heads focused on regulatory compliance and operational safety.
The Novel Approach
In stark contrast to these fragmented legacy methods, the technology disclosed in CN102516114B presents a unified, streamlined pathway that consolidates the resolution process into a single operational unit. The core innovation lies in the discovery that D-tartaric acid can effectively resolve the racemic amine mixture within a consistent ethanol-water solvent system, eliminating the need for intermediate isolation or solvent exchange. This one-step resolution allows for the direct crystallization of the desired R-(-)-2-amino-3-methoxyl-N-benzyl propionamide-D-tartrate salt with high optical purity simply by controlling temperature and seeding. The elegance of this system is further amplified by its integrated recycling capability; the mother liquor, enriched with the unwanted S-enantiomer, is not discarded but instead subjected to a thermal racemization process. This converts the S-isomer back into a racemic mixture, which is then fed back into the resolution reactor. This closed-loop design fundamentally transforms the mass balance of the production line, ensuring that nearly all raw material input is eventually converted into the valuable R-enantiomer product, thereby setting a new benchmark for cost reduction in pharmaceutical intermediate manufacturing.
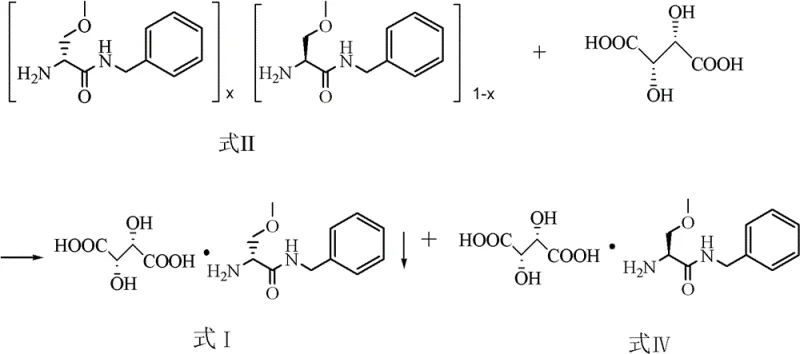
Mechanistic Insights into D-Tartrate Mediated Chiral Resolution
The success of this resolution strategy hinges on the precise thermodynamic control of diastereomeric salt solubility within the ethanol-water binary solvent system. When D-tartaric acid interacts with the racemic (R/S)-2-amino-3-methoxyl-N-benzyl propionamide mixture, it forms two distinct diastereomeric salts: the R-amine-D-tartrate salt and the S-amine-D-tartrate salt. Although these salts share similar chemical functionalities, their three-dimensional crystal lattice energies differ significantly, leading to divergent solubility profiles. In the optimized solvent composition, specifically where ethanol constitutes 70% to 90% of the volume, the R-salt exhibits markedly lower solubility compared to its S-counterpart, particularly at reduced temperatures. By carefully managing the cooling trajectory from a reflux state down to 25°C and introducing specific seed crystals of the R-salt, the system is driven into a state of supersaturation that selectively nucleates the desired diastereomer. This controlled crystallization prevents the co-precipitation of the S-salt, ensuring that the filter cake obtained possesses an HPLC purity exceeding 99% after a simple recrystallization wash. The mechanistic simplicity here avoids the need for chromatographic separation or enzymatic kinetic resolution, which are often cost-prohibitive at the metric-ton scale required for global API supply.
Beyond the primary resolution, the patent details a sophisticated mechanism for raw material conservation through thermal racemization. The filtrate remaining after the isolation of the R-salt is rich in the S-enantiomer tartrate salt. Rather than treating this as waste, the process employs a base-mediated deacidification followed by extraction to recover the free S-amine. This S-rich mixture is then subjected to heating at temperatures between 80°C and 120°C, either under solvent-free conditions or in the presence of water or alcohols. Under these thermal conditions, the chiral center at the alpha-carbon undergoes reversible deprotonation and reprotonation, effectively scrambling the stereochemistry and regenerating a near 1:1 racemic mixture. This thermal racemization is catalyst-free, avoiding the introduction of heavy metal impurities that would require costly downstream purification steps. The regenerated racemate is then seamlessly reintroduced into the resolution cycle. This mechanism ensures that the theoretical yield of the process can approach 100% over multiple cycles, providing a compelling argument for procurement managers seeking to minimize raw material volatility and secure long-term supply chain reliability for critical epilepsy medication ingredients.
How to Synthesize R-(-)-2-Amino-3-Methoxyl-N-Benzyl Propionamide Efficiently
The implementation of this synthesis route requires precise adherence to the solvent ratios and thermal profiles outlined in the patent to ensure consistent optical purity and yield. The process begins with the preparation of the racemic amine feedstock, which is dissolved in a pre-heated mixture of ethanol and water. The ratio of these solvents is critical; too much water reduces the solubility difference between the diastereomers, while too much alcohol keeps both salts in solution, preventing crystallization. Once the solution is homogenized at approximately 60°C, stoichiometric amounts of D-tartaric acid are added to form the salt in situ. The detailed standardized synthetic steps, including specific stirring rates, cooling gradients, and seeding protocols necessary to replicate the high-purity results described in the patent examples, are provided in the guide below.
- Dissolve the racemic (R/S)-2-amino-3-methoxyl-N-benzyl propionamide mixture in a heated ethanol-water solvent system.
- Add D-tartaric acid as the resolving agent and introduce seed crystals to initiate the selective crystallization of the R-enantiomer salt.
- Filter the high-purity R-salt filter cake and subject the remaining mother liquor to thermal racemization for raw material recycling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement directors and supply chain strategists, the transition to this one-step resolution technology represents a significant opportunity to optimize the cost structure and resilience of the API supply chain. The elimination of complex multi-solvent switching steps drastically reduces the consumption of organic solvents, which are not only expensive purchase items but also costly to dispose of in compliance with environmental regulations. By consolidating the resolution into a single vessel operation, manufacturers can reduce the footprint of their production facilities and lower the capital expenditure required for specialized equipment. Furthermore, the ability to recycle the mother liquor means that the effective cost of the starting chiral amine is significantly amortized over multiple production batches. This inherent efficiency translates into a more stable pricing model for the final intermediate, shielding buyers from the volatility often associated with chiral starting materials. The robustness of the ethanol-water solvent system also simplifies logistics, as these are common, non-hazardous solvents that are readily available globally, reducing the risk of supply disruptions compared to specialized aprotic solvents.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the maximization of raw material utility through the racemization recycle loop. In traditional resolution processes where the unwanted enantiomer is discarded, the maximum theoretical yield is capped at 50%, effectively doubling the raw material cost per unit of product. By thermally racemizing the S-enantiomer and refeeding it into the process, this technology pushes the overall yield well beyond the 50% limit, approaching quantitative conversion over time. Additionally, the removal of transition metal catalysts or expensive enzymatic agents from the workflow eliminates the need for rigorous metal scavenging steps and the associated analytical testing for residual impurities. This simplification of the purification train reduces both the direct cost of reagents and the indirect costs associated with quality control labor and extended batch cycle times, delivering substantial cost savings without compromising on the stringent purity specifications required for pharmaceutical grade materials.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of sourcing specialized chiral reagents or the fragility of multi-step synthetic routes. This method mitigates those risks by relying on D-tartaric acid, a commodity chiral pool chemical that is produced in massive quantities globally and is not subject to the same supply constraints as bespoke chiral ligands. The operational simplicity of the one-step resolution also reduces the likelihood of batch failures due to human error or equipment malfunction, as there are fewer unit operations and transfer points where things can go wrong. The use of a standard ethanol-water solvent system further enhances reliability, as these solvents are ubiquitous in chemical manufacturing and do not require specialized storage or handling infrastructure. For a reliable pharmaceutical intermediate supplier, this translates to the ability to guarantee consistent delivery schedules and maintain safety stock levels more effectively, ensuring that downstream API manufacturers never face production stoppages due to intermediate shortages.
- Scalability and Environmental Compliance: Scaling chiral processes from the laboratory to the multi-ton commercial scale is notoriously difficult, often resulting in a loss of optical purity or yield due to heat and mass transfer limitations. However, the crystallization-based nature of this resolution is inherently scalable, as the principles of solubility and nucleation remain consistent regardless of vessel size, provided that agitation and cooling rates are properly managed. The process operates at moderate temperatures (30°C to 60°C) and atmospheric pressure, removing the need for high-pressure reactors or cryogenic cooling systems, which simplifies engineering requirements for plant expansion. From an environmental perspective, the reduction in solvent variety and volume significantly lowers the E-factor (mass of waste per mass of product) of the synthesis. The aqueous waste streams generated are easier to treat biologically compared to those containing halogenated or aprotic solvents, facilitating compliance with increasingly strict environmental discharge regulations and supporting the sustainability goals of modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this chiral intermediate. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit within your manufacturing portfolio. Understanding these nuances is essential for R&D teams planning process validation and for procurement teams negotiating supply agreements.
Q: What distinguishes this one-step resolution method from conventional two-step processes?
A: Unlike traditional methods requiring separate salification and splitting steps in different solvent systems, this patented approach achieves resolution in a single solvent system (ethanol-water), significantly simplifying operations and reducing solvent consumption.
Q: How is the utilization rate of raw materials improved in this process?
A: The process incorporates a thermal racemization step for the mother liquor, converting the unwanted S-enantiomer back into a racemic mixture that can be recycled into the resolution step, thereby maximizing atom economy.
Q: What are the optimal conditions for the resolution reaction?
A: The resolution is optimally performed at temperatures between 30°C and 60°C using an ethanol-water mixed solvent with an ethanol volume percentage of 70% to 90%, ensuring a balance between yield and optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-(-)-2-Amino-3-Methoxyl-N-Benzyl Propionamide-D-Tartrate Supplier
As the pharmaceutical industry continues to demand higher purity and greater efficiency in the synthesis of life-saving medications, NINGBO INNO PHARMCHEM stands ready to deploy this advanced resolution technology for the commercial production of R-(-)-2-amino-3-methoxyl-N-benzyl propionamide-D-tartrate. Our facility is equipped with the specialized crystallization and thermal processing capabilities required to execute this one-step resolution and racemization cycle with precision. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot quantities for clinical trials or metric tons for market launch, our output remains consistent. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art chiral HPLC and polarimetry to verify the optical integrity of every batch, guaranteeing that the intermediate meets the exacting standards necessary for Lacosamide API synthesis.
We invite global partners to engage with our technical team to explore how this optimized manufacturing route can enhance your supply chain economics. By leveraging our expertise in chiral resolution and process intensification, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for your upcoming projects. Together, we can secure a sustainable and cost-effective supply of this critical chiral building block, driving value across the entire pharmaceutical value chain.
