Optimized Synthesis of Cefbuperazone Intermediates for Commercial Scale Production
The pharmaceutical industry constantly seeks robust synthetic routes for beta-lactam antibiotics that balance high purity with economic viability. Patent CN102212075A introduces a significant advancement in the preparation of cefbuperazone, a third-generation cephalosporin, by optimizing the synthesis of its critical side chain intermediates. This technology addresses long-standing challenges in the functionalization of D-threonine derivatives, specifically focusing on the efficient installation and removal of protecting groups. Unlike legacy processes that rely on hazardous gaseous reagents and corrosive acids, this novel approach employs mild esterification and selective protection strategies using reagents such as tetrahydropyranyl (THP) or trimethylsilyl (TMS) groups. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more sustainable, and cost-effective pharmaceutical intermediate manufacturing. The method ensures that key intermediates like DEPT-OMe and TBLZ-ester are produced with exceptional consistency, laying a solid foundation for the commercial production of high-quality antibiotic APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefbuperazone intermediates has been plagued by inefficient protection strategies that hinder large-scale production. Traditional methods often utilize isobutylene gas in the presence of concentrated sulfuric acid to protect hydroxyl and carboxyl groups simultaneously as tert-butyl ethers. While chemically effective on a small scale, this approach presents severe drawbacks for industrial application. The requirement for handling large volumes of strong mineral acids necessitates specialized, corrosion-resistant reactor vessels, significantly driving up capital expenditure. Furthermore, the deprotection step typically requires trifluoroacetic acid, generating substantial acidic waste streams that complicate environmental compliance. Another critical bottleneck in prior art involves the purification of intermediates; many existing routes rely heavily on silica gel column chromatography to isolate pure products after coupling reactions. This technique is notoriously difficult to scale, consumes vast quantities of solvents, and creates operational bottlenecks that extend lead times and inflate the cost of goods sold for the final antibiotic intermediate.
The Novel Approach
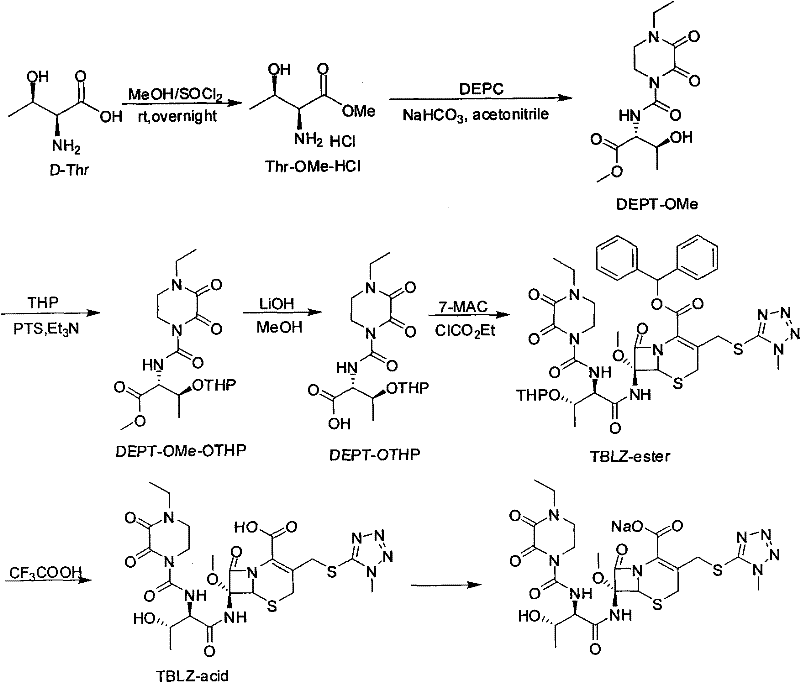
The methodology described in CN102212075A offers a transformative solution by replacing harsh conditions with mild, selective chemistry. The process initiates with the esterification of D-threonine using thionyl chloride in methanol, a standard and highly efficient transformation. The core innovation lies in the subsequent protection of the hydroxyl group. Instead of bulky tert-butyl groups, the patent advocates for the use of THP, TMS, or benzyl groups, which can be installed under mild basic or neutral conditions using catalysts like p-toluenesulfonic acid or imidazole. This shift eliminates the need for hazardous isobutylene gas and concentrated sulfuric acid. Moreover, the new route is designed to avoid column chromatography entirely. By optimizing reaction stoichiometry and workup procedures, such as liquid-liquid extraction and crystallization, the process yields white solids with purity levels consistently exceeding 96%. This streamlining of the workflow not only enhances safety but also drastically simplifies the downstream processing required to obtain the final TBLZ-ester and cefbuperazone acid.
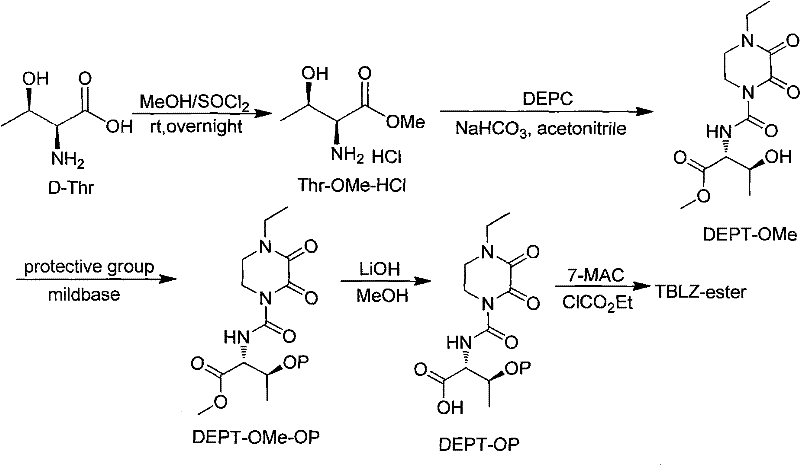
Mechanistic Insights into Selective Hydroxyl Protection and Coupling
The chemical elegance of this synthesis lies in the orthogonal stability of the protecting groups chosen for the D-threonine side chain. The mechanism begins with the nucleophilic attack of the threonine amino group on DEPC (4-ethyl-2,3-dioxopiperazine-1-carbonyl chloride) to form the urea linkage, creating the DEPT-OMe scaffold. The critical step is the protection of the secondary hydroxyl group at the beta-position. When using THP protection, the mechanism involves the acid-catalyzed formation of an oxocarbenium ion from dihydropyran, which is then attacked by the hydroxyl oxygen of the threonine derivative. This forms a stable acetal linkage that withstands the basic conditions of the subsequent ester hydrolysis. Alternatively, silyl protection using TMSCl proceeds via a nucleophilic substitution at the silicon center, facilitated by bases like imidazole or triethylamine which scavenge the generated HCl. These protecting groups are strategically selected because they are stable during the activation of the carboxylic acid for coupling with the 7-MAC nucleus but can be cleanly removed later.
Impurity control is rigorously managed through the choice of deprotection conditions. In the final stages, the TBLZ-ester contains both the side-chain protecting group (e.g., THP or TMS) and the 7-MAC benzhydryl ester protecting group. The patent reveals that these two distinct protecting groups can be removed simultaneously or sequentially under controlled acidic conditions, such as using a mixture of trifluoroacetic acid and anisole. This concurrent deprotection minimizes the number of isolation steps, thereby reducing the opportunity for product degradation or racemization. The resulting cefbuperazone acid precipitates directly from the reaction mixture as a solid, leaving soluble organic impurities and protecting group byproducts in the mother liquor. This "crash-out" purification mechanism is highly effective for achieving the stringent purity specifications required for injectable antibiotics, ensuring that the final product meets pharmacopeial standards without the need for recrystallization from toxic solvents.
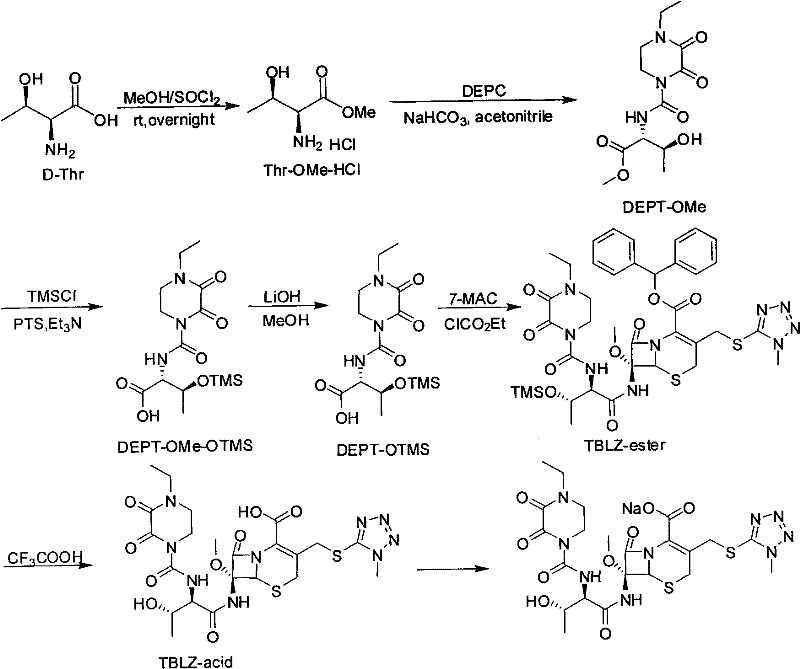
How to Synthesize Cefbuperazone Intermediates Efficiently
The synthesis of these high-value intermediates follows a logical progression designed for maximum yield and minimal waste. The process starts with the conversion of D-threonine to its methyl ester hydrochloride salt, followed by coupling with the piperazine derivative. The crucial divergence from standard protocols occurs in the protection step, where operators can select between silyl or ether-based protecting groups depending on availability and cost. Following protection, the methyl ester is hydrolyzed to the free acid using mild bases like lithium hydroxide or sodium carbonate, preserving the stereochemistry of the threonine moiety. Finally, the activated acid is coupled with the cephalosporin nucleus. For a detailed breakdown of the specific reaction parameters, stoichiometry, and workup procedures validated in the patent examples, please refer to the standardized guide below.
- Perform methyl esterification of D-Threonine using thionyl chloride in anhydrous methanol to generate Thr-OMe-HCl.
- React the threonine ester with DEPC in the presence of a mild base to form the piperazine-urea linkage (DEPT-OMe).
- Protect the hydroxyl group using silyl (TMS), ether (THP), or benzyl groups under mild conditions to prevent side reactions.
- Hydrolyze the methyl ester to the free acid (DEPT-OP) and couple with 7-MAC to form the final TBLZ-ester precursor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible operational efficiencies and risk mitigation. The primary value driver is the elimination of complex purification technologies like preparative HPLC or flash chromatography, which are often the most expensive and time-consuming parts of intermediate manufacturing. By relying on crystallization and extraction, the process becomes inherently more scalable and less dependent on specialized consumables. Furthermore, the reagents employed, such as THP and TMSCl, are commodity chemicals with stable global supply chains, unlike specialized gaseous reagents that may face logistical constraints. This reliability ensures consistent production schedules and reduces the risk of supply disruptions for critical antibiotic components.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from both raw material optimization and operational simplification. By avoiding the use of expensive and hazardous isobutylene gas and concentrated sulfuric acid, the facility reduces its spend on specialized safety infrastructure and waste neutralization. Additionally, the high yields reported in the patent examples, often exceeding 90% for individual steps, mean that less starting material is wasted. The ability to perform telescoped reactions or direct usage of crude intermediates in subsequent steps further lowers solvent consumption and energy costs associated with drying and isolation, leading to a significantly lower cost per kilogram for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the chemical transformations described. The reactions are homogeneous and proceed rapidly at ambient or mildly elevated temperatures, reducing the cycle time per batch. Faster cycle times allow for increased throughput within existing reactor capacity, effectively expanding production capability without capital investment. Moreover, the stability of the intermediates, such as the DEPT-OMe derivatives, allows for flexible inventory management. Manufacturers can stockpile stable intermediates during periods of low demand and rapidly convert them to the final active acid when market demand surges, providing a strategic buffer against supply chain volatility.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route offers a cleaner profile. The replacement of heavy metal catalysts or corrosive mineral acids with organic bases and mild Lewis acids reduces the toxicity of the effluent. The solvent systems primarily consist of methanol, dichloromethane, and ethyl acetate, all of which are readily recoverable through distillation. This aligns with modern green chemistry principles and facilitates easier permitting for plant expansions. The simplified waste stream lowers disposal costs and minimizes the environmental footprint of the manufacturing site, making it a more attractive partner for multinational corporations with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cefbuperazone synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on purity profiles, scalability, and regulatory considerations for potential partners.
Q: What are the advantages of using THP or TMS protection over traditional isobutylene methods?
A: Traditional methods often require harsh sulfuric acid conditions and isobutylene gas, posing significant safety and environmental risks. The patented method utilizes mild reagents like THP or TMSCl, which operate under neutral or slightly basic conditions, drastically reducing equipment corrosion and waste treatment costs while maintaining yields above 90%.
Q: How does this process ensure high purity for the final cefbuperazone acid?
A: The process achieves high purity through selective crystallization and the avoidance of column chromatography. By optimizing the deprotection step to simultaneously remove multiple protecting groups under controlled acidic conditions, impurities remain in the solution while the target cefbuperazone acid precipitates as a high-purity solid, often exceeding 99% purity.
Q: Is this synthesis route scalable for industrial manufacturing?
A: Yes, the route is designed for industrial scalability. It relies on homogeneous reactions that proceed rapidly and completely, eliminating the mass transfer limitations of solid-liquid two-phase systems found in older methods. The use of common solvents like methanol and dichloromethane further facilitates easy solvent recovery and scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefbuperazone Intermediate Supplier
The technological advancements detailed in CN102212075A underscore the complexity and precision required to manufacture high-quality cephalosporin intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate such patented methodologies into commercial reality. Our R&D team is adept at optimizing protection and deprotection sequences to maximize yield while maintaining rigorous quality standards. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials regardless of market fluctuations. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of cefbuperazone intermediate meets the exacting requirements of global regulatory bodies.
We invite pharmaceutical manufacturers and procurement leaders to collaborate with us to leverage this advanced synthesis route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply chain for your antibiotic production needs, driving efficiency and quality from the laboratory to the marketplace.
