Industrial Scale-Up of Glabridin: Overcoming Cryogenic and Chromatographic Barriers
Industrial Scale-Up of Glabridin: Overcoming Cryogenic and Chromatographic Barriers
The global demand for high-purity cosmetic active ingredients, particularly those with potent whitening and antioxidant properties like glabridin, has necessitated a re-evaluation of traditional synthetic methodologies. Patent CN103030647A introduces a robust, six-step synthetic pathway that fundamentally addresses the bottlenecks of prior art, specifically targeting the transition from laboratory curiosity to industrial viability. This technical breakthrough leverages a protected acetophenone starting material to navigate through a Willgerodt-Kindler reaction and a novel one-pot Friedel-Crafts cyclization, ultimately delivering the target isoflavone with a total yield exceeding 20 percent. For R&D directors and procurement specialists alike, this patent represents a critical pivot point, offering a route that eliminates the need for hazardous solvents like dichloroethane and avoids the prohibitive costs associated with cryogenic chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
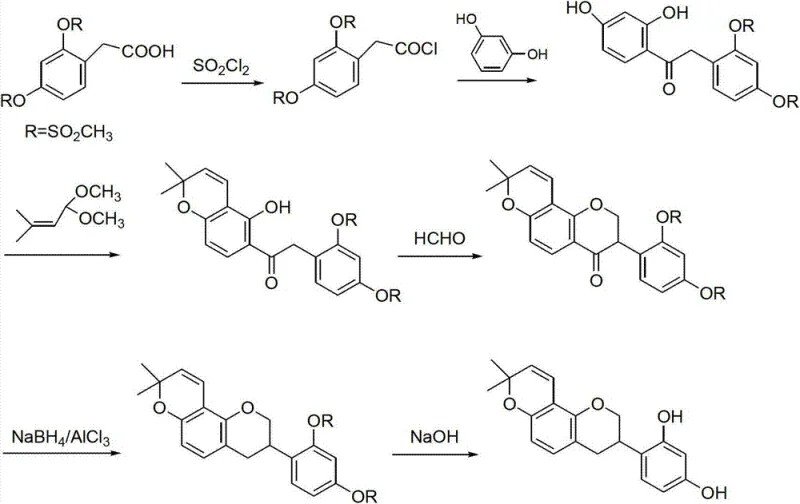
Historically, the chemical synthesis of glabridin has been plagued by operational complexities that render large-scale manufacturing economically unfeasible. As illustrated in the prior art referenced within the patent documentation, earlier attempts often relied on starting materials such as 2,4-dimethylsulfonyloxyphenylacetic acid, which necessitated a six-step sequence yielding only 25.8 percent overall. However, the critical failure point of this legacy route lies in its purification strategy; every single reaction step required column chromatographic separation. In an industrial context, column chromatography is a notorious bottleneck, consuming vast quantities of silica gel and solvents while drastically limiting throughput. Furthermore, the reliance on highly toxic solvents such as dichloroethane poses severe environmental and safety compliance risks, creating substantial liabilities for modern chemical manufacturers aiming for green chemistry standards.
Another significant barrier identified in the background technology is the route reported by Keepyung Nahm et al., which utilizes 2,4-dihydroxyacetophenone. While chemically valid, this ten-step synthesis achieves a dismal overall yield of merely 6.2 percent. The most prohibitive aspect of this methodology is the key condensation step between intermediates 1 and 2, which must be conducted at a cryogenic temperature of -78°C. Maintaining such low temperatures on a metric ton scale requires specialized refrigeration equipment and immense energy consumption, effectively pricing the final product out of the competitive cosmetic market. These conventional methods collectively highlight a desperate industry need for a process that operates under ambient or mild thermal conditions without sacrificing purity.
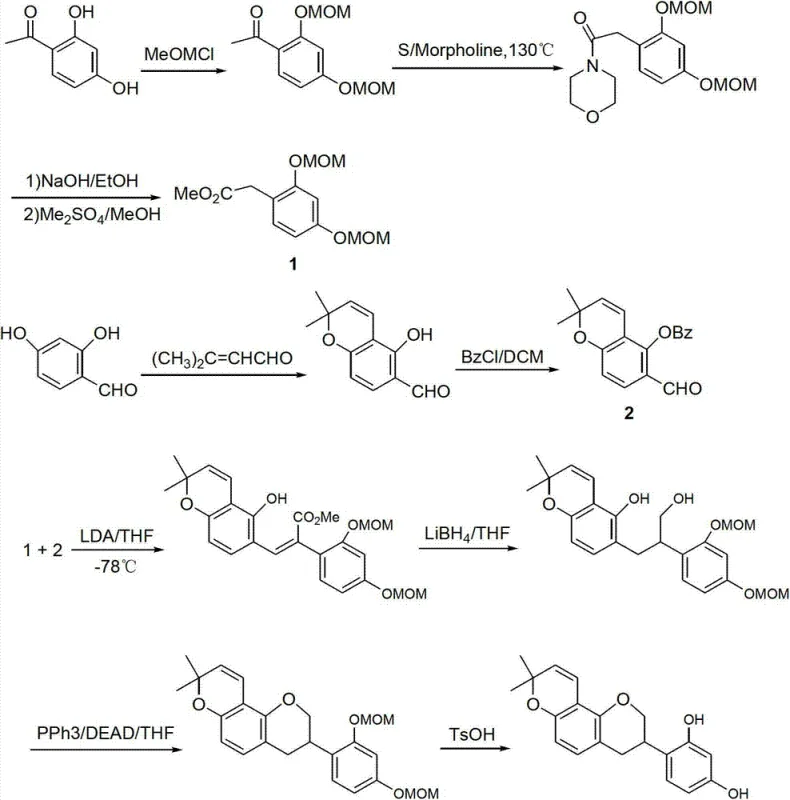
The Novel Approach
The methodology disclosed in CN103030647A offers a decisive solution to these historical inefficiencies by restructuring the synthetic logic around accessibility and operational simplicity. By initiating the synthesis with phenol-hydroxyl protected acetophenone, the route successfully bypasses the need for cryogenic temperatures entirely. A standout feature of this novel approach is the construction of the isoflavone core via a one-pot Friedel-Crafts reaction. Instead of isolating unstable intermediates, the process allows the reaction between the phenylacetic acid derivative and resorcinol to proceed directly in the presence of boron trifluoride etherate, followed by in-situ activation with methanesulfonyl chloride. This telescoping of steps not only reduces solvent usage but also minimizes material loss during transfer and isolation, directly contributing to the improved overall yield of not less than 20 percent.
Mechanistic Insights into Willgerodt-Kindler and Friedel-Crafts Cascade
The success of this synthetic strategy hinges on the precise execution of the Willgerodt-Kindler reaction in the initial stage, transforming the acetophenone derivative into the corresponding phenylacetic acid. This transformation involves the reaction of the ketone with elemental sulfur and morpholine under reflux conditions, forming a thioamide intermediate which is subsequently hydrolyzed under alkaline conditions. The mechanistic elegance here lies in the tolerance of the protecting groups; the alkoxy substituents remain stable throughout the harsh sulfur-mediated conditions, ensuring that the downstream functionality required for the final glabridin structure is preserved. Following hydrolysis and pH adjustment, the resulting acid is obtained in high purity through simple filtration, avoiding the chromatographic burdens of previous methods.
Subsequent steps focus on the rigorous control of stereochemistry and impurity profiles, particularly during the formation of the dihydropyran ring. The cyclization reaction between the isoflavanone intermediate and 3-methyl-2-butenal is carefully managed using solvents like pyridine or toluene with phenylboronic acid catalysis. This step is critical for establishing the characteristic prenyl-derived ring system of glabridin. The patent specifies that by optimizing the molar ratios and reaction times—typically refluxing for 6 to 14 hours depending on the solvent system—the formation of regioisomers is minimized. The final deprotection step utilizes reagents such as boron tribromide or hydrobromic acid to cleave the alkyl protecting groups, revealing the free phenolic hydroxyls essential for the biological activity of the final API intermediate.
How to Synthesize Glabridin Efficiently
The synthesis of glabridin via this patented route offers a clear roadmap for process chemists aiming to establish a reliable supply chain for this high-value cosmetic active. The protocol is designed to maximize yield while minimizing the need for complex purification equipment, making it an ideal candidate for technology transfer from pilot plant to full commercial production. The following guide outlines the critical operational phases derived from the patent examples, emphasizing the specific conditions required to achieve the reported efficiencies.
- Perform Willgerodt-Kindler reaction on 2,4-dialkoxy acetophenone with sulfur and morpholine, followed by alkaline hydrolysis to yield phenylacetic acid.
- Execute a one-pot Friedel-Crafts reaction with resorcinol using boron trifluoride etherate and methanesulfonyl chloride to form the isoflavone core.
- Conduct Pd/C catalytic hydrogenation, followed by cyclization with prenyl aldehyde, carbonyl reduction, and final deprotection to obtain glabridin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates directly into enhanced margin potential and supply security. The elimination of cryogenic requirements removes a significant capital expenditure barrier, allowing production to occur in standard stainless steel reactors without the need for specialized low-temperature cooling jackets or liquid nitrogen infrastructure. Furthermore, the shift from column chromatography to recrystallization and filtration as the primary purification methods drastically reduces the consumption of silica gel and organic solvents. This reduction in consumable usage not only lowers the direct cost of goods sold but also simplifies waste management protocols, aligning with increasingly stringent environmental regulations regarding solvent disposal.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of unit operations. By avoiding the use of exotic reagents and eliminating the need for repeated chromatographic separations, the manufacturing overhead is significantly reduced. The use of common, bulk-available reagents such as sulfur, morpholine, and resorcinol ensures that raw material costs remain stable and predictable, shielding the production budget from the volatility often associated with specialty fine chemicals. Additionally, the higher overall yield means that less starting material is required to produce the same amount of final product, effectively amplifying the purchasing power of the raw material budget.
- Enhanced Supply Chain Reliability: Supply continuity is a critical concern for downstream formulators in the cosmetic and pharmaceutical industries. This synthetic route enhances reliability by relying on a short, six-step sequence compared to the ten-step alternatives, thereby reducing the cumulative risk of batch failure at any single stage. The robustness of the reaction conditions, which tolerate standard heating and atmospheric pressure, ensures that production schedules are not disrupted by equipment limitations or sensitive parameter deviations. This stability allows for more accurate lead time forecasting and the ability to scale production volumes rapidly in response to market demand spikes.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this process offers a cleaner profile that facilitates easier regulatory approval for commercial plants. The replacement of toxic dichloroethane with safer solvents like ethanol, ethyl acetate, or toluene reduces the hazard classification of the manufacturing site. The ability to perform key transformations in a one-pot manner reduces the volume of wastewater generated between steps. These factors collectively lower the cost of environmental compliance and waste treatment, making the facility more sustainable and resilient against future regulatory tightening regarding volatile organic compound emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glabridin synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications for industrial adoption.
Q: How does this new synthesis route improve upon previous methods regarding scalability?
A: Unlike previous routes requiring -78°C cryogenic conditions or extensive column chromatography at every step, this method utilizes standard reflux conditions and recrystallization, making it highly suitable for multi-kilogram production.
Q: What is the overall yield efficiency of the patented glabridin synthesis process?
A: The patent reports a total yield of not less than 20 percent across six steps, which is a significant improvement over the 6.2 percent yield observed in the 10-step alternative route.
Q: Are the reagents used in this process readily available for industrial procurement?
A: Yes, the process relies on common industrial reagents such as sulfur powder, morpholine, resorcinol, and palladium on carbon, ensuring a stable and cost-effective supply chain without reliance on exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glabridin Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this optimized synthesis are fully realized in the final product. We operate stringent purity specifications and maintain rigorous QC labs equipped to detect and quantify trace impurities, guaranteeing that every batch of glabridin meets the exacting standards required for high-end cosmetic formulations and pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific innovation and commercial reliability.
