Advanced Microwave-Assisted Synthesis of Paraben Esters for Commercial Scale-Up
Advanced Microwave-Assisted Synthesis of Paraben Esters for Commercial Scale-Up
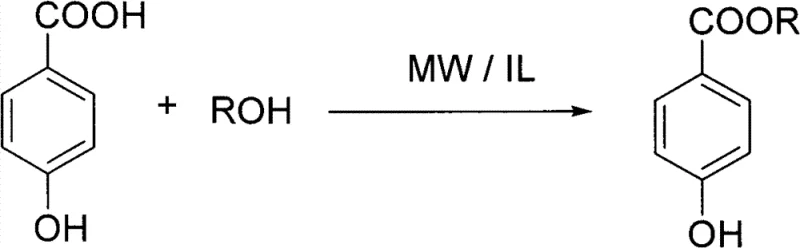
The global demand for high-purity preservatives and pharmaceutical intermediates continues to drive innovation in green chemical manufacturing processes. A pivotal advancement in this sector is detailed in Chinese Patent CN101982453B, which discloses a novel preparation method for paraben ester compounds promoted by sulfonic acid-type ionic liquids. This technology represents a significant departure from traditional mineral acid-catalyzed esterification, addressing critical pain points regarding equipment corrosion, environmental pollution, and product quality. By leveraging the unique physicochemical properties of task-specific ionic liquids combined with microwave irradiation, this process achieves exceptional reaction efficiency under remarkably mild conditions. For procurement leaders and R&D directors seeking a reliable paraben ester supplier, understanding the mechanistic advantages of this patented route is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of nipagin esters, or parabens, has relied heavily on the Fischer esterification process utilizing concentrated sulfuric acid as a homogeneous catalyst. While chemically effective, this legacy approach presents severe operational drawbacks that impact both capital expenditure and product integrity. The highly corrosive nature of sulfuric acid necessitates the use of specialized, expensive reactor linings and piping, leading to accelerated equipment degradation and frequent maintenance downtime. Furthermore, the harsh acidic environment promotes undesirable side reactions, including sulfonation of the aromatic ring and oxidation, which often result in deeply colored crude products requiring extensive and costly purification steps. The disposal of acidic waste streams also poses significant environmental compliance challenges, increasing the overall ecological footprint of the manufacturing facility.
The Novel Approach
In stark contrast, the methodology described in patent CN101982453B utilizes sulfonic acid-functionalized ionic liquids as dual-purpose reaction media and catalysts. This innovative system eliminates the need for volatile organic solvents and corrosive mineral acids, thereby creating a inherently safer and cleaner reaction environment. The process operates at moderate temperatures between 80°C and 120°C, significantly reducing energy consumption compared to high-temperature reflux methods. Crucially, the ionic liquid catalyst can be easily separated from the organic product through simple aqueous extraction and subsequently recycled, drastically reducing raw material costs. The integration of microwave radiation further accelerates the reaction kinetics, shortening production cycles from many hours to merely 0.5 to 3 hours, which directly enhances throughput capacity for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Sulfonic Acid Ionic Liquid Catalysis
The catalytic efficacy of this system stems from the Brønsted acidic nature of the sulfonic acid groups (-SO3H) tethered to the imidazolium cation of the ionic liquid. These acidic protons activate the carbonyl oxygen of the p-hydroxybenzoic acid, increasing its electrophilicity and facilitating nucleophilic attack by the alcohol. Unlike traditional liquid acids, the ionic liquid creates a structured solvent cage that stabilizes the transition state, thereby lowering the activation energy of the esterification. The polar nature of the ionic liquid also ensures excellent solubility for the polar starting materials while allowing the less polar ester product to be easily extracted, driving the equilibrium forward. This dual functionality as both solvent and catalyst simplifies the reaction mixture complexity and minimizes the formation of tar-like byproducts often seen in heterogeneous acid catalysis.
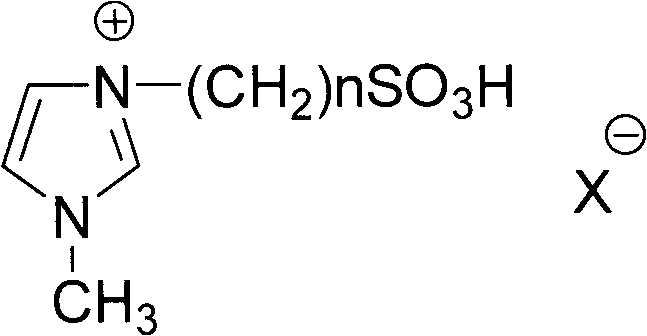
Impurity control is inherently superior in this ionic liquid system due to the mild reaction conditions and the absence of strong oxidizing agents. Traditional sulfuric acid methods often lead to charring and polymerization of organic substrates, generating complex impurity profiles that are difficult to remove. In the ionic liquid promoted reaction, the temperature is strictly controlled via microwave feedback, preventing thermal runaway and localized overheating. The specific interaction between the ionic liquid anions and the reaction intermediates suppresses side reactions such as ether formation or dehydration of the alcohol. Consequently, the crude product obtained is of high purity, often appearing as a white solid immediately after solvent removal, which significantly reduces the burden on downstream purification units and ensures consistent quality for high-purity food additives and pharmaceutical ingredients.
How to Synthesize Paraben Esters Efficiently
The operational protocol for this synthesis is designed for simplicity and scalability, making it highly attractive for industrial adoption. The process begins with the premixing of p-hydroxybenzoic acid and the selected sulfonic acid ionic liquid, followed by the addition of the alcohol substrate. The reaction vessel is then subjected to microwave irradiation, where precise temperature control ensures optimal conversion rates without degradation. Post-reaction workup involves a straightforward liquid-liquid extraction strategy that leverages the water solubility of the ionic liquid to separate it from the organic ester phase. Detailed standardized synthetic steps see the guide below for specific molar ratios and processing parameters derived from the patent examples.
- Mix p-hydroxybenzoic acid with a sulfonic acid functionalized ionic liquid (such as N-(4-sulfobutyl)imidazolium hydrogen sulfate) and stir at room temperature.
- Add the corresponding alcohol (e.g., n-butanol, isobutanol) and subject the mixture to microwave radiation at 80-120°C for 0.5 to 3 hours.
- Cool the reaction, add water, extract the product with ether, wash with saturated sodium bicarbonate and brine, dry, and evaporate solvent to obtain the pure ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this ionic liquid-based technology offers profound strategic benefits beyond mere chemical yield. The elimination of corrosive sulfuric acid translates directly into substantial cost savings by extending the lifespan of reactor vessels and reducing the frequency of equipment replacement. Furthermore, the ability to recycle the ionic liquid catalyst multiple times without significant loss of activity creates a closed-loop system that minimizes raw material waste and lowers the variable cost per kilogram of production. The simplified workup procedure, which avoids complex neutralization and washing steps required for mineral acids, reduces labor hours and wastewater treatment volumes, contributing to a leaner and more cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the recyclability of the expensive ionic liquid component. Since the catalyst remains in the aqueous phase after extraction, it can be recovered via evaporation and drying, effectively amortizing its initial cost over numerous batches. Additionally, the reduction in reaction time from traditional multi-hour refluxes to under three hours increases asset utilization rates, allowing existing infrastructure to produce higher volumes without capital expansion. The avoidance of hazardous waste disposal fees associated with spent mineral acids further improves the overall margin profile for cost reduction in food additive manufacturing.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions, which are less sensitive to minor fluctuations in temperature or mixing compared to heterogeneous catalysis. The use of readily available starting materials like p-hydroxybenzoic acid and common alcohols ensures that raw material sourcing remains stable and unaffected by niche reagent shortages. The shortened production cycle time means that lead times for customer orders can be significantly compressed, enabling a more responsive just-in-time delivery model that aligns with the dynamic demands of the global personal care and pharmaceutical markets.
- Scalability and Environmental Compliance: Scaling this process from laboratory to pilot and commercial plant is facilitated by the homogeneous nature of the reaction mixture and the efficiency of microwave heating technologies which are increasingly available at industrial scales. The process generates minimal hazardous waste, as the primary byproduct is water, and the ionic liquid is retained within the system. This aligns perfectly with modern environmental, social, and governance (ESG) goals, reducing the regulatory burden and ensuring long-term operational licenses in regions with strict environmental protection laws regarding acidic effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and claims presented in the underlying patent documentation, providing a factual basis for evaluating the feasibility of adopting this route for your specific product portfolio. Understanding these details is crucial for technical teams assessing the compatibility of this method with existing facility capabilities.
Q: Can the ionic liquid catalyst be recycled in this paraben synthesis process?
A: Yes, the sulfonic acid ionic liquid serves as both solvent and catalyst and can be recovered from the aqueous phase after extraction. By removing water via rotary evaporation and azeotropic distillation with toluene, the ionic liquid is regenerated for reuse without significant loss of catalytic activity.
Q: What are the typical reaction yields for this microwave-assisted method?
A: According to patent data, the molar yield of the finished paraben products consistently exceeds 85%. Specific examples demonstrate yields ranging from 87.5% to 92.5% depending on the specific alcohol chain length used.
Q: How does this method compare to traditional sulfuric acid catalysis?
A: Unlike traditional methods using corrosive sulfuric acid which cause equipment damage and produce dark-colored byproducts, this ionic liquid method operates under mild conditions (80-120°C), prevents equipment corrosion, simplifies post-treatment, and results in high-purity white solid products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paraben Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green catalytic technologies like the sulfonic acid ionic liquid method for producing high-value esters. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards for pharmaceutical and food-grade applications. We are prepared to leverage this advanced synthesis route to deliver superior product consistency and reliability to our global partners.
We invite you to engage with our technical team to explore how this efficient manufacturing process can optimize your supply chain. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how partnering with our technical procurement team can drive value and efficiency in your paraben ester sourcing strategy.
