Advanced Solvent-Free Synthesis of PinB(DMA) for Commercial Scale Pharmaceutical Intermediates
Advanced Solvent-Free Synthesis of PinB(DMA) for Commercial Scale Pharmaceutical Intermediates
The landscape of organic synthesis is constantly evolving, driven by the need for more robust, efficient, and scalable reagents that can withstand the rigors of industrial manufacturing. A pivotal advancement in this domain is detailed in patent CN102093399A, which introduces a novel boronizing agent known as dimethylamino boronic acid pinacol ester, or PinB(DMA). This compound represents a significant leap forward in boronation chemistry, addressing long-standing challenges related to reagent stability and process safety. Unlike conventional boron sources that often demand cryogenic conditions and stringent anhydrous environments, PinB(DMA) offers a unique combination of low water sensitivity and structural stability. This technical breakthrough not only streamlines the synthetic workflow for creating valuable boronic acid derivatives but also opens new avenues for cost-effective production of complex pharmaceutical intermediates. As the industry seeks reliable solutions for high-volume API manufacturing, the adoption of such stable reagents becomes a strategic imperative for maintaining supply chain continuity and operational efficiency.
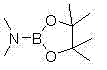
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of high-value boronic acid esters has relied heavily on traditional boronating agents such as trimethyl borate, triisopropyl borate, or methoxy boronic acid pinacol ester. While these reagents have served the industry for decades, they possess inherent drawbacks that complicate large-scale operations. Primarily, these compounds are extremely sensitive to moisture and prone to facile hydrolysis, which imposes severe constraints on their packaging, storage, and transportation. In a practical manufacturing setting, this sensitivity necessitates the use of specialized containment systems and rigorous environmental controls to prevent degradation before the reagent even reaches the reactor. Furthermore, the high reactivity of these traditional agents often mandates harsh reaction conditions, specifically ultra-low temperatures ranging from cryogenic levels to minus twenty degrees Celsius, to control exotherms and selectivity. These energy-intensive requirements not only inflate operational costs but also introduce significant safety risks and engineering complexities when attempting to scale up from laboratory benchtop to multi-ton commercial production.
The Novel Approach
In stark contrast to the fragility of legacy reagents, the novel synthesis pathway for PinB(DMA) outlined in the patent data presents a robust and operationally simple alternative. The core innovation lies in the direct reaction between tris(dimethylamino)borane and pinacol under solvent-free conditions, eliminating the need for volatile organic solvents that complicate downstream purification. By operating at moderate temperatures between 50°C and 70°C, the process avoids the energy penalties associated with cryogenic cooling while maintaining excellent control over the reaction kinetics. The resulting product, PinB(DMA), is characterized by its remarkable stability and low sensitivity to water, which drastically simplifies handling procedures and reduces the risk of batch failure due to environmental exposure. This shift from moisture-sensitive liquids to a more stable ester structure allows for a more flexible supply chain, where materials can be stored and transported with standard precautions rather than exotic containment strategies, thereby enhancing the overall reliability of the manufacturing process for downstream users.
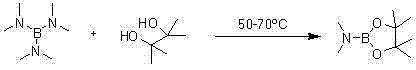
Mechanistic Insights into Solvent-Free Boronation Synthesis
The mechanistic elegance of the PinB(DMA) synthesis lies in its atom-economic design and the elimination of solvent interference. The reaction proceeds through a nucleophilic substitution where the hydroxyl groups of the molten pinacol attack the boron center of the tris(dimethylamino)borane. This interaction facilitates the displacement of dimethylamine groups, which are released as a gas, driving the equilibrium forward without the need for additional dehydrating agents or azeotropic distillation setups. The absence of solvent is particularly advantageous from a kinetic perspective, as it increases the effective concentration of the reactants, thereby accelerating the reaction rate and allowing for shorter cycle times. Moreover, the gaseous byproduct serves as a self-indicating marker for reaction progress; the cessation of gas evolution provides a clear, visual, and physical signal that the conversion is nearing completion, simplifying process monitoring and reducing the reliance on complex inline analytical tools during the bulk reaction phase.
From an impurity control standpoint, the simplicity of the reaction matrix translates directly into superior product purity. Since no external solvents are introduced, there is no risk of solvent-derived impurities or residual solvent peaks complicating the final specification of the boronating agent. The post-reaction workup involves a straightforward vacuum distillation under reduced pressure, typically around minus 0.1 MPa, to isolate the product fraction boiling between 45°C and 50°C. This thermal separation is highly effective at removing any unreacted starting materials or higher-boiling oligomers, yielding a product with a GC purity exceeding 98%. The structural integrity of the pinacol ester ring further contributes to this purity profile, as the steric bulk of the gem-dimethyl groups protects the boron-oxygen bonds from hydrolytic cleavage, ensuring that the final reagent remains stable during storage and subsequent use in sensitive cross-coupling reactions where trace impurities could otherwise poison catalysts or derail synthetic sequences.
How to Synthesize PinB(DMA) Efficiently
The synthesis of Dimethylamino Boronic Acid Pinacol Ester is a streamlined process that leverages solvent-free chemistry to achieve high efficiency and purity. By utilizing readily available raw materials like tris(dimethylamino)borane and pinacol, manufacturers can bypass the complexities of traditional esterification methods. The procedure is designed for scalability, relying on thermal activation and vacuum distillation rather than complex chromatographic purification. For research and development teams looking to implement this route, the following guide outlines the critical parameters for success, emphasizing the importance of temperature control and inert atmosphere protection to maximize yield and safety. Detailed standardized synthesis steps follow below to ensure reproducibility across different scales of operation.
- Mix tris(dimethylamino)borane and pinacol in a molar ratio of 1.05-1.2: 1.0 under an inert gas atmosphere.
- Heat the mixture to 50-70°C and react for 2-5 hours without solvent until gas evolution ceases.
- Distill the reaction mixture under vacuum (-0.1 MPa) and collect the fraction boiling at 45-50°C to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to PinB(DMA) represents a strategic opportunity to optimize both cost structures and logistical resilience. The elimination of solvents in the synthesis process is a primary driver for cost reduction, as it removes the expense of purchasing, recovering, and disposing of large volumes of organic liquids. This solvent-free approach significantly lowers the environmental footprint of the manufacturing process, aligning with increasingly stringent global regulations on volatile organic compound emissions and waste management. Furthermore, the high stability of the final product reduces the risk of spoilage during transit and storage, minimizing inventory write-offs and ensuring that capital is not tied up in degraded materials. These factors combine to create a more predictable and economical supply model for essential boronating reagents.
- Cost Reduction in Manufacturing: The economic benefits of this novel synthesis route are substantial, primarily driven by the drastic simplification of the production workflow. By operating under solvent-free conditions, the process eliminates the need for expensive solvent recovery infrastructure and the associated energy costs for distillation and drying. The high yield reported in the patent embodiments, reaching up to 98%, ensures that raw material utilization is maximized, directly lowering the cost of goods sold. Additionally, the mild reaction conditions reduce energy consumption compared to cryogenic processes, contributing to lower utility bills and a smaller carbon footprint. These efficiencies allow suppliers to offer competitive pricing while maintaining healthy margins, providing a tangible financial advantage for downstream purchasers of API intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the instability of key reagents, but PinB(DMA) mitigates this risk through its inherent chemical robustness. Traditional boronating agents require cold chain logistics and specialized packaging to prevent hydrolysis, which can lead to delays and increased shipping costs. In contrast, the low water sensitivity of PinB(DMA) allows for standard storage and transportation protocols, reducing the complexity of logistics and the likelihood of shipment rejections due to quality deviations. This stability ensures that manufacturers can maintain consistent inventory levels without the fear of rapid degradation, thereby securing the production schedule for critical pharmaceutical intermediates and reducing the lead time for high-purity specialty chemicals.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden bottlenecks, particularly regarding heat transfer and waste treatment. The PinB(DMA) synthesis is inherently scalable because it avoids the use of solvents that would otherwise require massive reactors to accommodate dilute concentrations. The simple vacuum distillation workup is easily adaptable to large-scale industrial columns, facilitating the commercial scale-up of complex pharmaceutical intermediates without the need for bespoke engineering solutions. Moreover, the absence of solvent waste streams simplifies environmental compliance, reducing the burden on wastewater treatment facilities and lowering the costs associated with hazardous waste disposal. This alignment with green chemistry principles makes the process attractive for companies aiming to meet sustainability goals while expanding production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of PinB(DMA) in industrial settings. These insights are derived directly from the patented technology and are intended to clarify the operational benefits and application scope of this advanced boronating agent. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this reagent into existing synthetic routes for drug substance manufacturing.
Q: What are the stability advantages of PinB(DMA) over traditional boronating agents?
A: Unlike traditional reagents such as trimethyl borate which are highly moisture-sensitive and require low-temperature storage, PinB(DMA) exhibits low water sensitivity and high structural stability, simplifying logistics and handling.
Q: Can PinB(DMA) be used for large-scale API intermediate production?
A: Yes, the solvent-free synthesis process described in patent CN102093399A is highly scalable, offering simple post-treatment via vacuum distillation and achieving yields up to 98%, making it ideal for industrial manufacturing.
Q: What types of boronic esters can be synthesized using PinB(DMA)?
A: PinB(DMA) is versatile and effective for synthesizing alkyl, aryl, and heterocyclic boronic acid pinacol esters, serving as a critical building block for Suzuki-Miyaura cross-coupling reactions in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable PinB(DMA) Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates relies on a partnership grounded in technical expertise and manufacturing excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We are committed to delivering PinB(DMA) with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to handle complex chemistries under GMP-like conditions positions us as a trusted partner for global innovators seeking to secure their supply chains for critical boron-based building blocks.
We invite you to explore how our optimized synthesis of PinB(DMA) can drive value in your organization. By leveraging our technical capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to enhance your production efficiency and secure a reliable source of high-quality chemical intermediates for your next generation of therapeutic products.
