Advanced Synthesis of Thiazolyl-pyrazolopyrimidine Intermediates for CRF-1 Antagonist Production
The pharmaceutical industry's relentless pursuit of novel therapeutics for neurological disorders has placed CRF-1 receptor antagonists at the forefront of research, particularly for treating depression and alcohol dependence. Patent CN102171212A introduces a groundbreaking advancement in this field by disclosing a novel class of thiazolyl-pyrazolopyrimidine compounds, specifically designated as Formula I, which serve as critical synthetic intermediates. These intermediates are pivotal in the efficient manufacture of final active pharmaceutical ingredients (APIs) such as Formula II, which exhibit potent antagonistic activity. The patent delineates a sophisticated synthetic pathway that not only enhances the overall yield but also addresses significant safety and environmental concerns associated with legacy manufacturing processes. By leveraging a copper-catalyzed coupling strategy, this technology offers a robust platform for the commercial scale-up of complex pharmaceutical intermediates, ensuring a steady supply of high-quality materials for downstream drug development.
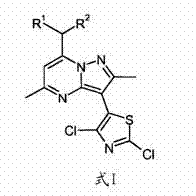
Furthermore, the structural versatility of these intermediates, where R1 and R2 can be independently ethyl or n-propyl groups, allows for the fine-tuning of physicochemical properties essential for drug efficacy. The transition from traditional stoichiometric methods to this catalytic approach represents a paradigm shift in process chemistry, emphasizing sustainability without compromising on the stringent purity requirements demanded by regulatory bodies. For reliable pharmaceutical intermediate suppliers, mastering this specific molecular architecture is key to supporting the global pipeline of neuropsychiatric medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in WO 2008/036579, often relied on harsh reaction conditions that posed significant challenges for industrial application. Traditional routes frequently necessitated the use of mutagenic substances and environmentally toxic catalysts, creating substantial hurdles for waste management and operator safety. Moreover, conventional coupling reactions often suffered from poor atom economy, requiring stoichiometric amounts of expensive metal reagents which drove up the cost of goods sold (COGS). The instability of key reagents, particularly dichlorothiazole derivatives under strong basic conditions or elevated temperatures, frequently led to degradation products that were difficult to separate, thereby compromising the purity profile of the final API. These inefficiencies resulted in prolonged cycle times and complex purification workflows, such as extensive column chromatography, which are impractical for multi-ton manufacturing scales.
The Novel Approach
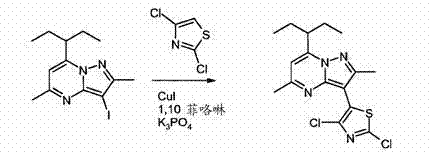
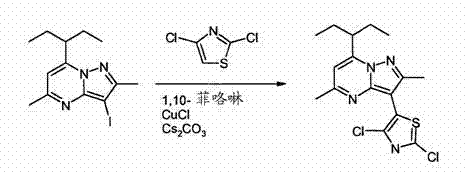
The innovative process disclosed in CN102171212A overcomes these historical bottlenecks by employing a mild yet highly effective copper-catalyzed arylation protocol. This novel approach utilizes catalytic quantities of copper halides, such as CuCl, in conjunction with stable nitrogen-based ligands like 1,10-phenanthroline. This combination facilitates the coupling of the pyrazolopyrimidine core with the thiazole moiety under significantly milder conditions, typically between 80°C and 120°C, preventing the thermal degradation observed in older methods. Crucially, the selection of appropriate bases, specifically cesium carbonate (Cs2CO3) or potassium phosphate (K3PO4), ensures that the sensitive dichlorothiazole reagent remains intact throughout the reaction. This strategic optimization leads to superior yields and a cleaner reaction profile, drastically reducing the burden on downstream purification units and enabling a more streamlined path to high-purity pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed Arylation
The core of this technological breakthrough lies in the mechanistic efficiency of the copper-catalyzed C-N or C-C bond formation, depending on the specific substitution pattern targeted. In this specific embodiment, the reaction likely proceeds through a Cu(I)/Cu(III) catalytic cycle where the 1,10-phenanthroline ligand plays a dual role: stabilizing the copper center against oxidation and facilitating the oxidative addition of the aryl halide. The use of 2-MeTHF (2-methyltetrahydrofuran) mixed with DMAC (N,N-dimethylacetamide) as a co-solvent system is particularly ingenious. 2-MeTHF offers excellent solubility for organic substrates while being derived from renewable resources, aligning with green chemistry principles, whereas DMAC helps solubilize the inorganic base. The synergy between the solvent system and the catalyst ensures that the activation energy for the coupling is lowered, allowing the reaction to proceed to completion with minimal formation of homocoupling byproducts or dehalogenated impurities.
Impurity control is further enhanced by the precise regulation of reaction temperature and base strength. As noted in the patent data, stronger bases like lithium tert-butoxide cause rapid decomposition of the thiazole ring, whereas the moderate basicity of Cs2CO3 provides the necessary deprotonation without inducing side reactions. This selectivity is paramount for maintaining the integrity of the chloro-substituents on the thiazole ring, which are essential for the subsequent nucleophilic substitution step. By minimizing the generation of closely related impurities, the process inherently supports the production of materials that meet rigorous specification limits, a critical factor for cost reduction in API manufacturing where purification often accounts for a majority of processing time.
How to Synthesize Thiazolyl-pyrazolopyrimidine Efficiently
The synthesis of these valuable intermediates is executed through a carefully orchestrated two-step sequence that maximizes throughput and minimizes waste. The first step involves the coupling of a 3-iodo-pyrazolo[1,5-a]pyrimidine derivative with 2,4-dichlorothiazole under inert atmosphere to prevent catalyst deactivation. Following the isolation of the dichloro-intermediate, the second step employs a nucleophilic aromatic substitution where morpholine displaces one of the chlorine atoms. Detailed standard operating procedures for this synthesis, including specific molar ratios and temperature ramps, are outlined below to guide process engineers in replicating these results.
- React a 3-iodo-pyrazolo[1,5-a]pyrimidine derivative with 2,4-dichlorothiazole using a copper halide catalyst (e.g., CuCl), 1,10-phenanthroline ligand, and a base like Cs2CO3 in a 2-MeTHF/DMAC solvent system.
- Isolate the resulting 2,4-dichloro-thiazolyl intermediate via filtration and crystallization, ensuring removal of copper residues and unreacted starting materials.
- Perform nucleophilic substitution by reacting the dichloro-intermediate with excess morpholine in 2-propanol or 2-MeTHF, utilizing morpholine as both reactant and base to form the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits beyond mere technical feasibility. The shift from stoichiometric to catalytic metal usage fundamentally alters the cost structure of the synthesis, removing the dependency on bulk quantities of expensive heavy metals. Furthermore, the simplified work-up procedures, which often allow for direct crystallization from the reaction solvent, eliminate the need for resource-intensive solvent swaps and chromatographic separations. This operational simplicity enhances the resilience of the supply chain by reducing the number of unit operations and potential failure points, ensuring consistent delivery schedules for downstream clients.
- Cost Reduction in Manufacturing: The implementation of catalytic copper systems significantly lowers raw material expenses by reducing the loading of precious metal catalysts from stoichiometric levels to mere mole percentages. Additionally, the ability to use morpholine as both a reactant and a base in the second step eliminates the need for purchasing and handling separate inorganic bases, further streamlining the bill of materials. The high purity achieved directly from crystallization reduces the loss of product during purification, effectively increasing the overall mass balance and yield per batch, which drives down the unit cost of the final intermediate.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as 2-MeTHF, DMAC, and common copper salts, are commodity chemicals with robust global supply chains, mitigating the risk of shortages associated with exotic or specialized reagents. The stability of the intermediates and the tolerance of the reaction to minor variations in conditions provide a buffer against supply fluctuations, ensuring that production timelines remain unaffected by raw material quality variances. This reliability is crucial for maintaining continuous manufacturing campaigns required to support large-scale clinical trials and commercial launches.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful transition from gram-scale experiments to multi-kilogram batches without loss of efficiency. The avoidance of mutagenic reagents and the use of greener solvents like 2-MeTHF simplify the environmental permitting process and reduce the costs associated with hazardous waste disposal. This alignment with modern environmental, social, and governance (ESG) goals makes the supply chain more sustainable and attractive to partners who prioritize green manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing clarity on critical process parameters.
Q: How does this new synthetic route improve safety compared to previous methods?
A: The patented process avoids the use of mutagenic substances and environmentally toxic catalysts found in prior art (e.g., WO 2008/036579). By utilizing catalytic amounts of copper with stable ligands like 1,10-phenanthroline and avoiding strong, degrading bases at high temperatures, the method significantly enhances operator safety and environmental compliance.
Q: What are the key advantages of using 2-propanol in the amination step?
A: Using 2-propanol as a solvent unexpectedly shortens reaction time by approximately three-fold compared to other systems. It allows for direct crystallization of the final product from the reaction mixture, eliminating the need for solvent exchange and simplifying the work-up process while maintaining high purity (>99%).
Q: Can this process be scaled for commercial API production?
A: Yes, the process is designed for scalability. The use of robust solvents like 2-MeTHF and DMAC, combined with efficient crystallization protocols that avoid complex chromatography, makes it highly suitable for commercial scale-up of complex pharmaceutical intermediates from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiazolyl-pyrazolopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation neurological therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of thiazolyl-pyrazolopyrimidine intermediate meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific drug development needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can accelerate your timeline to market while maximizing value.
