Advancing Ketone Synthesis: A Green Blue-Light Copper Catalysis Strategy for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct ketone motifs, which serve as pivotal scaffolds in steroids, quinones, and flavonoids. Patent CN115650837A introduces a transformative approach for preparing these valuable ketone compounds through a 1,4-addition reaction of alpha,beta-unsaturated enones. This technology leverages a synergistic combination of hypervalent iodine reagents and copper catalysis under visible blue light irradiation. By shifting away from harsh oxidative conditions, this method offers a greener, milder pathway that maintains high functional group tolerance. For R&D teams focused on complex molecule synthesis, this represents a significant leap forward in accessing diverse chemical space without compromising safety or purity standards.
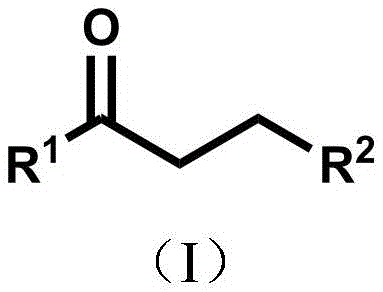
This innovation addresses the critical need for reliable pharmaceutical intermediate supplier capabilities by enabling the synthesis of structures that were previously difficult to access efficiently. The core breakthrough lies in the generation of carbon-centered radicals from carboxylic acid derivatives via hypervalent iodine decarboxylation. These radicals act as nucleophiles in a Michael-type addition, allowing for the introduction of a wide array of alkyl, aryl, and heteroaryl groups. This versatility is crucial for medicinal chemists optimizing lead compounds, as it facilitates rapid structure-activity relationship (SAR) studies with minimal process development overhead.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ketone compounds has relied heavily on the oxidation of alcohols using strong oxidizing agents such as hydrogen peroxide or tert-butyl hydroperoxide. These traditional oxidants are inherently unstable and prone to spontaneous decomposition, creating significant safety hazards in large-scale manufacturing environments. Furthermore, alternative strategies involving Grignard reagents for 1,4-addition to enones often suffer from poor regioselectivity, where competitive 1,2-addition leads to unwanted alcohol byproducts. The use of pyrophoric organometallic reagents also necessitates stringent anhydrous conditions and specialized handling equipment, driving up operational costs and complicating supply chain logistics for high-purity OLED material or API production.
The Novel Approach
The methodology disclosed in the patent circumvents these issues by employing a photoredox-mediated radical coupling strategy. Instead of unstable peroxides or hazardous Grignard reagents, the process utilizes stable hypervalent iodine species that release carbon radicals upon single-electron transfer. This shift not only eliminates the risk of thermal runaway associated with peroxide decomposition but also ensures exclusive 1,4-selectivity, avoiding the formation of 1,2-addition impurities. The reaction proceeds under ambient temperature and blue light, removing the need for energy-intensive heating or cryogenic cooling. This温和 (mild) condition profile is particularly advantageous for cost reduction in electronic chemical manufacturing, where thermal sensitivity of substrates is a common concern.
Mechanistic Insights into Cu-Catalyzed Radical 1,4-Addition
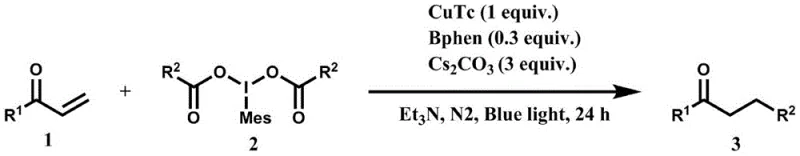
The catalytic cycle initiates with the coordination of the copper(I) catalyst, such as copper thiophene-2-carboxylate, with a phenanthroline ligand to form an active complex. Upon irradiation with blue light, this complex enters an excited state capable of reducing the hypervalent iodine(III) reagent through a single-electron transfer (SET) process. This reduction triggers the homolytic cleavage of the iodine-carbon bond, releasing a carbon-centered radical and generating a copper(II) species. The generated radical serves as a potent nucleophile that attacks the beta-position of the alpha,beta-unsaturated ketone, which has been activated by the base to form an enolate-like intermediate. This precise mechanistic control ensures that the reaction follows a Michael addition pathway rather than direct carbonyl attack.
Impurity control is inherently built into this mechanism due to the specific reactivity of the radical species. Unlike ionic nucleophiles which may attack multiple electrophilic sites, the radical intermediate is selectively guided by the electronic properties of the enone system. The subsequent recombination or termination steps regenerate the catalyst or form stable byproducts that are easily removed during purification. This high level of chemoselectivity is vital for producing high-purity pharmaceutical intermediates, as it minimizes the formation of structural isomers that are difficult to separate. The use of cesium carbonate as a base further stabilizes the transition state, ensuring consistent reaction kinetics across different substrate classes ranging from aromatic to aliphatic enones.
How to Synthesize Ketone Derivatives Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that combines the enone substrate, the hypervalent iodine reagent, the copper catalyst, ligand, and base in a solvent like triethylamine. The reaction mixture is stirred under an inert argon atmosphere at room temperature while being exposed to blue light for a period of 12 to 24 hours. Following the completion of the reaction, monitored by TLC, the solvent is removed under reduced pressure, and the crude product is purified via column chromatography. This streamlined workflow reduces the number of unit operations required, directly contributing to reducing lead time for high-purity pharmaceutical intermediates.
- Combine alpha,beta-unsaturated enone, hypervalent iodine reagent, copper catalyst, ligand, and base in a suitable solvent under inert atmosphere.
- Stir the reaction mixture at room temperature under blue light irradiation for 12 to 24 hours to facilitate the radical coupling.
- Remove the solvent via rotary evaporation and purify the crude residue using column chromatography to isolate the target ketone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial benefits by replacing expensive and hazardous reagents with cost-effective alternatives. The elimination of pyrophoric Grignard reagents and unstable peroxides significantly lowers the cost of goods sold (COGS) by reducing the need for specialized safety infrastructure and waste disposal protocols. The use of earth-abundant copper catalysts instead of precious metals like palladium or rhodium further drives down raw material costs. Additionally, the broad substrate scope means that a single standardized protocol can be applied to synthesize a wide library of derivatives, simplifying inventory management and enhancing supply chain reliability for complex polymer additives or agrochemical intermediates.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with inexpensive copper salts drastically reduces the catalyst cost per kilogram of product. Furthermore, the mild reaction conditions eliminate the need for energy-intensive heating or cooling systems, leading to lower utility costs. The simplified workup procedure, which avoids complex quenching steps required for reactive organometallics, reduces labor hours and solvent consumption, resulting in substantial cost savings overall.
- Enhanced Supply Chain Reliability: The reagents used in this process, such as hypervalent iodine derivatives and copper salts, are commercially available and stable for long-term storage. This stability mitigates the risk of supply disruptions caused by the degradation of sensitive reagents during transport. The robustness of the reaction conditions also allows for flexible scheduling and batch production, ensuring consistent delivery timelines for critical API intermediates without the bottlenecks associated with hazardous material handling.
- Scalability and Environmental Compliance: The absence of heavy metal waste and toxic byproducts aligns with increasingly stringent environmental regulations. The process generates minimal hazardous waste, simplifying effluent treatment and reducing compliance costs. The scalability is demonstrated by the ability to run reactions at room temperature without exothermic spikes, making the transition from laboratory scale to commercial scale-up of complex pharmaceutical intermediates seamless and safe.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. Understanding these details helps R&D and procurement teams evaluate the feasibility of adopting this technology for their specific production needs. The answers are derived directly from the experimental data and mechanistic understanding provided in the patent documentation.
Q: What are the safety advantages of this hypervalent iodine method over traditional oxidation?
A: Unlike traditional methods that rely on unstable peroxides like hydrogen peroxide or tert-butyl hydroperoxide which pose decomposition risks, this method utilizes stable hypervalent iodine reagents and mild copper catalysis, significantly reducing explosion hazards and thermal runaway risks during scale-up.
Q: How does this method improve regioselectivity compared to Grignard reagents?
A: Traditional Grignard reagents often suffer from competitive 1,2-addition, especially with smaller R groups, leading to alcohol byproducts. This radical-mediated 1,4-addition strategy ensures high selectivity for the beta-position, delivering pure ketone skeletons essential for complex drug molecule construction.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process operates at room temperature using inexpensive copper catalysts and avoids cryogenic conditions or hazardous pyrophoric reagents. The simple workup involving solvent removal and chromatography makes it highly amenable to commercial scale-up for producing high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketone Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes like the hypervalent iodine-mediated coupling described in CN115650837A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with advanced photoreactors and rigorous QC labs capable of maintaining stringent purity specifications required for global pharmaceutical markets. We are committed to translating this academic breakthrough into a robust industrial process that delivers high-quality ketone intermediates consistently.
We invite you to collaborate with us to leverage this technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule. Please contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can optimize your supply chain and accelerate the development of your novel therapeutic candidates using this cutting-edge chemistry.
