Transforming Biological Waste into High-Value API Intermediates via Advanced Steroid Conversion
Transforming Biological Waste into High-Value API Intermediates via Advanced Steroid Conversion
The pharmaceutical industry constantly seeks sustainable and efficient pathways to produce critical active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is documented in patent CN112341512A, which outlines a robust method for preparing chenodeoxycholic acid (CDCA) utilizing seal cholic acid (PCA) as the primary raw material. This innovation addresses a dual challenge: it provides a high-value application for seal cholic acid, which is often a residual component in biological processing, and simultaneously mitigates the chronic global supply shortage of chenodeoxycholic acid. By transforming a biological byproduct into a essential steroid medicine precursor, this technology not only enhances resource utilization but also offers a strategic solution for reducing dependency on traditional extraction methods that are often limited by raw material availability. The technical depth of this patent suggests a viable pathway for industrial manufacturers to diversify their supply chains while adhering to greener chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of chenodeoxycholic acid has relied heavily on direct extraction from animal bile, specifically from sources like duck bile. However, this conventional approach faces inherent limitations regarding yield and resource efficiency. In standard extraction processes, seal cholic acid (PCA) is frequently co-extracted but remains difficult to separate or utilize effectively, often ending up as residual biliary paste waste. This residue is typically downgraded to low-value applications such as feed additives or disposed of as solid waste, representing a significant loss of potential chemical value. Furthermore, the direct extraction of CDCA is constrained by the natural concentration of the compound in biological sources, leading to supply bottlenecks that cannot easily meet the surging global demand for liver and gallstone treatments. The reliance on finite biological pools without effective conversion technologies creates a fragile supply chain vulnerable to fluctuations in agricultural outputs and seasonal variations.
The Novel Approach
The methodology presented in patent CN112341512A introduces a transformative chemical conversion strategy that turns the liability of PCA waste into a strategic asset. Instead of discarding seal cholic acid, this novel approach subjects it to a precise four-step synthetic sequence involving protection, halogenation, elimination, and hydrogenation. This chemical intervention allows manufacturers to access a vast, previously underutilized reservoir of raw materials. By chemically modifying the side chain of the steroid nucleus, the process effectively converts the trihydroxy structure of PCA into the dihydroxy structure required for CDCA. This shift from extraction to semi-synthesis significantly broadens the feasible raw material base, ensuring a more stable and continuous supply of this critical intermediate. The ability to synthesize CDCA from abundant PCA derivatives fundamentally alters the economic landscape of steroid manufacturing, offering a scalable alternative to nature-dependent extraction.
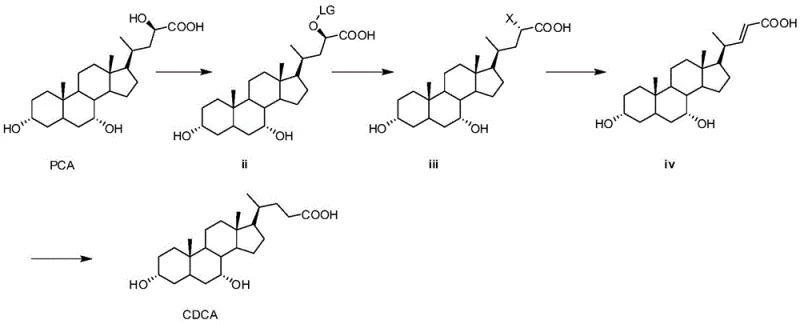
Mechanistic Insights into Steroid Side-Chain Modification
The core of this synthesis lies in the precise manipulation of the steroid side chain through a leaving group strategy. The process initiates with the protection of the alpha-hydroxy group at the C-23 position of the seal cholic acid. By reacting PCA with a leaving group reagent, such as methylsulfonyl chloride or p-toluenesulfonyl chloride, under alkaline conditions using organic bases like DBU or triethylamine, the hydroxyl group is converted into a superior leaving group (LG). This activation is crucial as it primes the molecule for subsequent nucleophilic substitution. The reaction is typically conducted at low temperatures (0-5°C) to maintain stereochemical integrity and prevent degradation of the sensitive steroid backbone. This step ensures that the subsequent halogenation proceeds with high regioselectivity, targeting specifically the activated position without affecting the other hydroxyl groups on the steroid rings.
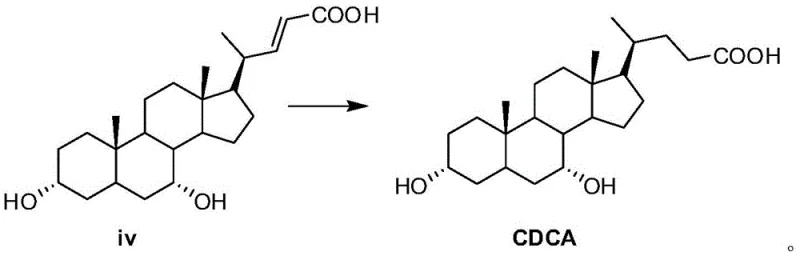
Following protection, the molecule undergoes halogenation where the leaving group is displaced by a halogen atom, preferably bromine, using reagents like lithium bromide in refluxing acetone. This generates a halo-intermediate which is then subjected to an elimination reaction under basic conditions using polar aprotic solvents like hexamethylphosphoramide. This elimination step is pivotal as it introduces a double bond into the side chain, creating an alkene intermediate (compound iv). The final transformation involves catalytic hydrogenation using palladium on charcoal. This step reduces the carbon-carbon double bond to a single bond, saturating the side chain and yielding the final chenodeoxycholic acid structure. The mechanistic elegance of this route lies in its modularity; each step is designed to maximize yield and purity, with the final hydrogenation serving as a polishing step that removes unsaturation impurities, resulting in a product with HPLC purity often exceeding 97%.
How to Synthesize Chenodeoxycholic Acid Efficiently
Implementing this synthesis requires careful control of reaction parameters to ensure optimal conversion rates and product quality. The process begins with the activation of the raw material, followed by sequential functional group transformations that progressively build the target molecular architecture. Operators must pay close attention to temperature controls during the protection phase and solvent choices during the elimination phase to avoid side reactions. The final hydrogenation step is critical for achieving the saturation profile required for biological activity. While the general workflow is established, specific operational details regarding reagent ratios, workup procedures, and purification protocols are essential for successful scale-up. For a comprehensive guide on the standardized synthesis steps, please refer to the detailed protocol below.
- Protect the alpha-hydroxy group of seal cholic acid using a sulfonyl leaving group reagent under alkaline conditions to form compound ii.
- Perform a halogenation reaction on compound ii using a halide salt in acetone to substitute the leaving group with a halogen, yielding compound iii.
- Execute an elimination reaction on compound iii under basic conditions to generate the alkene intermediate, compound iv.
- Conduct a catalytic hydrogenation of compound iv using palladium on charcoal to reduce the double bond and obtain high-purity chenodeoxycholic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers compelling advantages rooted in raw material economics and process stability. The primary driver for cost optimization is the utilization of seal cholic acid, a material that is historically undervalued and often treated as waste. By integrating this feedstock into the production line, manufacturers can significantly lower the baseline cost of goods sold compared to relying solely on high-demand, directly extracted CDCA. This shift reduces exposure to volatile market prices associated with traditional bile sources. Furthermore, the chemical steps utilize widely available industrial reagents such as acetone, methylene chloride, and standard halide salts, avoiding the need for exotic or prohibitively expensive catalysts in the early stages. This accessibility of inputs contributes to a more predictable and manageable cost structure for long-term manufacturing contracts.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the valorization of waste streams. Since seal cholic acid is a byproduct of existing bile processing, its acquisition cost is inherently lower than that of purified CDCA. Eliminating the need for complex separation of PCA from CDCA in the initial extraction phase simplifies the upstream supply chain. Additionally, the avoidance of transition metal catalysts in the initial protection and substitution steps reduces the burden on downstream purification and heavy metal removal processes. This streamlined workflow translates to substantial operational savings, as fewer unit operations are required to achieve pharmaceutical-grade purity, thereby enhancing the overall margin potential for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Diversifying the raw material base is a critical strategy for mitigating supply risk. By enabling the production of CDCA from seal cholic acid, manufacturers are no longer solely dependent on the fluctuating yields of direct CDCA extraction from biological sources. This flexibility ensures a more consistent supply volume, which is vital for meeting the steady demand of the global gallstone medication market. The robustness of the chemical synthesis route means that production can be ramped up based on chemical capacity rather than biological availability. This reliability is particularly attractive for long-term supply agreements where continuity of supply is a key performance indicator for pharmaceutical buyers seeking to secure their API pipelines against biological variability.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard reactor setups and common solvents that are easily managed in large-scale facilities. The conversion of biological waste into high-value medicine aligns with increasingly stringent environmental regulations regarding waste disposal. By repurposing PCA, the process reduces the environmental footprint associated with biological waste treatment. The final hydrogenation step uses heterogeneous catalysis (Pd/C), which allows for easy catalyst recovery and recycling, further supporting green chemistry initiatives. This alignment with sustainability goals not only aids in regulatory compliance but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for environmentally conscious global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of chenodeoxycholic acid from seal cholic acid. These insights are derived directly from the technical specifications and experimental data provided in the patent literature. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for commercial production. The answers reflect the balance between chemical efficiency and practical manufacturability inherent in the disclosed method.
Q: What is the primary advantage of using seal cholic acid (PCA) for CDCA production?
A: The primary advantage is the valorization of biological waste. PCA is often a byproduct of duck bile processing that is discarded or underutilized. Converting it to CDCA expands the raw material source, alleviates supply shortages, and reduces environmental pollution associated with biological waste disposal.
Q: How does this method ensure high purity of the final Chenodeoxycholic Acid?
A: The method employs a controlled multi-step synthesis including specific protection and elimination steps that minimize side reactions. The final product undergoes recrystallization, achieving HPLC purity levels exceeding 97%, which meets stringent pharmaceutical standards for API intermediates.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the patent explicitly addresses industrial production capabilities. The reaction conditions utilize common solvents like acetone and methylene chloride, and standard reagents such as lithium bromide and palladium on charcoal, making the process highly adaptable for large-scale commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chenodeoxycholic Acid Supplier
The technological pathway described in patent CN112341512A represents a significant opportunity for optimizing the production of high-purity chenodeoxycholic acid. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative synthetic routes into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are efficiently translated into industrial outputs. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of CDCA meets the exacting standards required for pharmaceutical applications. Our commitment to quality ensures that the complex stereochemistry of the steroid nucleus is preserved throughout the manufacturing process.
We invite procurement leaders and technical directors to collaborate with us on optimizing their supply chains for steroid intermediates. By leveraging our capabilities, you can secure a stable source of high-quality CDCA while benefiting from the cost efficiencies of this novel synthesis method. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing prowess can support your long-term strategic goals in the pharmaceutical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
